MOJ
eISSN: 2381-182X


Research Article Volume 2 Issue 2
University of Alaska, USA
Correspondence: Matthew P Davenport, University of Alaska, Fairbanks; Fisheries Industrial Technology Center, 118 Trident Way Kodiak AK, 99617, USA
Received: December 17, 2015 | Published: March 23, 2016
Citation: Davenport MP, Smiley S. Determination of reaction progress in the enzymatic hydrolysis of seafood processing byproducts: a simple alternative to the degree of hydrolysis. MOJ Food Process Technol. 2016;2(2):67-72. DOI: 10.15406/mojfpt.2016.02.00033
In many areas of food processing it is desirable to reduce the length of protein molecules. The two most commonly used methods employ either inorganic acids or proteolytic enzymes to cleave peptide bonds between adjacent amino acids. Proteolytic enzymes can reduce the viscosity of aqueous protein mixtures as well as the volume of insolubles. In the Alaskan seafood industry, liquification of processing byproducts by protein hydrolysis would be convenient if there was a simple and reliable method to gauge the progress of the reaction. Laboratory methods to quantify the degree of hydrolysis are expensive, time consuming and demand operators trained in analytical techniques. The biuret protein method uniquely shifts color from blue to purple in the presence of proteins (longer chains of amino acids) and from blue to pink in the presence of hydrolysates or the peptide results of proteolytic action.
The biuret color shift can be used for the quantitative determination of the rate of hydrolysis without the need for expensive equipment. Our goal in this research has been to develop a rapid and simple method to ascertain the progress of hydrolytic enzymatic reactions involving the enzyme Alcalase and the byproducts of seafood processing in Alaska. If allowed to run fully to completion, protein hydrolysis, by either acidification or enzyme reaction, results in the formation of high concentrations of free amino acids imparting bitterness to the hydrolysate. The bitter taste is off putting to fish chickens and livestock reducing feed consumption and limiting growth. A rapid, inexpensive and simple technique to monitor the progress of hydrolytic reactions could be used to control the development of bitter free amino acids. Production of consistently acceptable protein hydrolysates with low levels of free amino acids offers a cost effective method for small remote processors to convert the byproducts of their seafood processing into acceptable and defined feed ingredients.
The economic impact of the seafood industry is significant in the state of Alaska, producing over 65% of wild fish consumed in the U.S.1 Salmon harvested in Alaska in 2014 totaled more than 156 million fish with an ex-vessel value greater than $575 million.2 Much of the salmon harvested in Alaska is processed close to natal salmon streams in small remote facilities across the southeastern, southern and western coasts of the state.
Salmon processing byproducts depends critically on the product form being processed. While heads and viscera are common to virtually all forms of processing, other parts such as backbones and skins are produced only when filleting the fish.3 Because the byproducts are created during the processing of human food, they are subject to considerable regulation underlining the fact that their native quality is very high. These processing byproducts contain potentially valuable components4 and utilization of these byproducts of seafood processing promise to meaningfully increase the realized value of the harvested fish.
The kinds of co-products developed from the waste stream in seafood processing depends on investment. In the huge processing plants shoreside of the Bering Sea, waste streams are handled through wet reduction operations such as Atlas Stord waste processing plants that cost upwards of $20 million. However, small, remote, and seasonal salmon operations cannot justify those kinds of investments and need a less expensive solution. Transforming the seafood processing waste stream into valuable co products has warranted many years of study.5 One promising method, for those processing operations with lower waste stream volumes, involves hydrolyzing the material thereby making it more manageable. Efficient hydrolyzing operations allow bone and fish oil to be separated from the protein mass and that protein converted into a shelf stable product that can be cost effectively co dried. Hydrolysis involves minimal equipment and is much cheaper than available wet reduction operations.
The co-products made from byproducts include fish oil, fish bone meal, fish protein meal and other value added fish feed ingredients.4 Their value depends on the investment and thus the quality of the co product. Fish protein meals of fair to average quality (FAQ) made via wet-reduction from whole anchovetta in Peru, currently command a value of greater than $1,650 per metric ton.6 Fish oil designated for the aquacultural feeds industry is currently valued at greater than $2,100 per metric ton.7 The market for feed ingredients such as palatants and attractants is much less characterized. Refined, human consumable salmon oil would return a significantly higher value but this also requires the highest investment in equipment and facilities.
Although the market value of fish protein hydrolysates will be less that that of wet reduction protein meals never-the-less they still have value. In addition, effective handling of processing byproducts reduces incurred costs associated with managing the plant’s waste stream to meet water quality regulations. Hydrolysates with consistent properties command the greater value but these require controlled conditions, specifically the application of enzymes and of temperature to the byproduct stream.8 To control this, a rapid, simple method for measuring the progress of the enzymatic reaction in the commercial processing plant environment, would prove very helpful.
The utilization of enzyme technology in seafood processing allows for the ease of oil separation from a protein rich liquid9 as well as the production of palatants and feed attractants for incorporation into dietary formulations.10 These materials may be made from relatively low value fish processing waste; increasing returns from a greater portion of the harvested fish.
Aquaculture contributed 36% of the total seafood products in the world in 2006 and is expected to increase with time.11 The increase will heighten the demand for feed ingredients including fish meal.12 Global fish meal production is dependent on climatic conditions, especially off the coast of Peru, and has actually decreased at times.12 In 2004/2005 approximately 144,000 metric tons (mt) of fish meal were produced globally, while in 2008/2009 that figure had dropped to some 97,500 mt internationally.13 Demand for value added aquafeed ingredients to enhance growth performance is increasing. Researchers have shown that an exclusively plant based diet for aqua cultured carnivorous fish does not provide the same weight gains as is found when the plant based diet is augmented with fish protein.10 Enzyme technologies provide one way to meet the increasing need for fish based feed ingredients.
Measuring reaction progress in a protein hydrolysis system, to control the presence of unpalatable bitter components, is necessary.14 By monitoring hydrolysis, the processor can manage the production of these unpleasant flavors and create a standardized, high quality product.15
The most common method to evaluate and monitor the enzyme/protein interaction is by determining the degree of hydrolysis; and there are multiple methods for doing this. These methods require expensive, dangerous chemicals as well as dedicated equipment along with highly trained personal to conduct these tests.16-22 This is costly, time consuming and may not be completed rapidly enough to provide onsite quality control during production. The goal of this work was to develop a rapid, accurate, simple method for monitoring the progress of hydrolysis for the industrial application of enzyme technology in seafood processing in the state of Alaska.
To this end we have adapted the biuret reaction23 through the implementation of advanced colorimetric information of significant confusion is the fact that the term biuret describes a family of organic compounds with the functional group -(HN-CO-)2N- as well as a specific chemical compound known as carbamylurea.23 Neither of these are involved in the biuret reaction or our modifications of the standard biuret test.
Materials
Fresh pollock (Theragra chalcogramma), were obtained from a commercial fish processing plant in Kodiak, Alaska, USA. Fish were immediately returned to the laboratory and filleted. Fresh pollock fillets were frozen and stored at -20°C until needed. The enzyme used in this study was commercial alcalase (Novozymes Inc., Denmark) 2.4L and was provided by a local processor. All chemicals used were of regent grade unless otherwise noted.
Preparation of fish protein hydrolysates
Frozen pollock was thawed over night at 4°C and ground through a 0.5 cm die (VillaWare Power Grinder 320; Cleveland, OH). The ground pollock was mixed 1:2 (w/v) with DDI water and homogenized in a Waring blender (Torrington, CT) for two bursts of 30 seconds at full speed and allowed to equilibrate to 50°C for 10 minutes in a water bath. After equilibration, the alcalase agent was added at 2uL/g and incubated for 0, 15, 30, 45 or 60 minutes. To inactivate the enzyme, the solutions were heated in a water bath at >90°C for 10 minutes.
Indicators of hydrolysis
TCA soluble peptides: The degree of hydrolysis was determined by the method of Hoyle & Merritt.17 10 ml of pollock/enzyme slurry was added to an equal volume of 20% trichloroacetic acid (TCA) to yield a final concentration of 10% TCA prior to heat inactivation. This slurry was then centrifuged at 2400 x g (Thermo Scientific; Waltham, MA) and the supernatant decanted for measurement of soluble nitrogen content using the Leco TruSpec-N nitrogen analyzer (St. Joseph, MI) standardized with ethylene-diamine-tetra-acetic acid (EDTA) according the manufactures recommendations. The degree of hydrolysis was calculated using Hoyle & Merritt’s equation:17 [TCA soluble %N/Total %N]*100.
Absorbance at A280: The spectro photometric absorbance of the supernatants was measured at 280 nm (Varian Cary 50; Palo Alto, CA) by monitoring the increase of tryptophan in the soluble fraction. Supernatants were diluted 1:15 v/v to achieve absorbance measurements within the linear range of the machine (abs < 2.0).
Wet balance: The wet pellet weight was determined after centrifugation at 2400 x g (Thermo Scientific; Waltham, MA) and the supernatant decanted. Pellet weight was determined by difference.
Solids content: The solids content of the supernatants was determined by drying the samples for 24 hrs at 105°C (Thermo Scientific; Waltham, MA) in pre weighed sample pans according to published AOAC methods.24 The solids content was determined by difference in weights assuming all losses were water.
Biuret test color: A biuret reagent: 0.3% copper sulfate, 3.0% NaOH, 0.6% sodium potassium tartrate,25 was used to determine the degree of reaction progress. Biuret reagent (4 ml) was added to the supernatants (1 ml) and allowed to incubate at room temperature (~22°C) for at least 20 minutes. The Hunter color (L*, *a, *b) of the solutions was measured with a Minolta chromameter (Minolta CR-300; Toyko, Japan) operated with a white background card behind the test tubes. The biuret reaction progress was calculated as Hunter color [(*a)*(*b)].
Statistics
Linear correlation comparisons were conducted in Excel (Microsoft Corp., Seattle WA). KYPlot (2.0 beta 15 (32 Bit); Tokyo, Japan) was used to determine differences between treatments via Analysis of Variance followed by Tukey’s test.
Solids content
The solids content of the soluble phase shows a steady increase during the time course of the alcalase hydrolysis (Table 1). Compared to the control, the increase is most simply seen as an increase in soluble proteins fragments or peptides. Other methods have been suggested as rapid measurements of hydrolysis that depend on the change in the solids content of the soluble phase. These methods include refractometry Brixo or the change in freezing point as potential rapid measurements of hydrolysis reaction progress.26 The Brix refractometry method requires larger changes in solids content than those seen in this study.27 The small change in solids content seen in these experiments will not provide the accuracy needed to determine the extent of the hydrolysis reaction with any of these alternative methods.
|
Biuret |
TCA Soluble Peptides |
OD280 |
Solids |
Supernatant |
Untreated |
|
|
|
|
|
0 |
-293.66a |
5.94a |
0.87a |
1.93a |
56.69a |
15 |
-219.85a |
4.90a |
0.88a |
2.07a |
58.41a |
30 |
-300.51a |
5.01a |
0.90a |
2.07a |
61.40a |
45 |
-330.07a |
5.16a |
0.88a |
2.04a |
60.24a |
60 |
-309.94a |
5.481a |
0.87a |
2.09a |
59.69a |
Alcalase |
|
|
|
|
|
0 |
-44.41b |
5.73ab |
1.53b |
4.49b |
77.27b |
15 |
-13.25bc |
9.29b |
1.70c |
4.68bc |
80.95bc |
30 |
-3.47bc |
10.58bc |
1.78cd |
4.81bc |
82.89c |
45 |
6.880c |
11.22c |
1.91d |
4.98c |
83.60c |
60 |
16.72c |
11.64c |
1.95d |
5.07c |
83.72c |
Table 1 Values are averages and SD listed for at least three determinations. Alcalase treatments are 2uL/g. Biuret color determined by a*b (Biuret), TCA soluble peptides, absorbance at 280nm (OD 280), solids content of the supernatant after centrifugation (Solids) and the weight of the supernatant after centrifugation (Supernatant). Significant differences (p<0.05) within columns are indicated by different letters
OD 280
The absorbance at 280 nm of the soluble phase is shown in Table 1. Absorbance at this UV wavelength has been associated with the presence of the phenolic amino acids tryptophan, tyrosine and phenylalanine.28 The presence of other phenolic compounds may interfere, however the presence of significant concentrations of other phenolic compounds is unlikely. Figure 2 shows a steady increase in absorbance at 280 nm over the time course of Alcalase hydrolysis. When compared to the control, a relatively constant absorbance at 280 nm is observed. This means that the increase in absorbance is almost completely due to the hydrolyzing of fish proteins into soluble protein fragments and peptides and any endogenous proteolytic activity in pollock byproducts is not observable under these conditions.
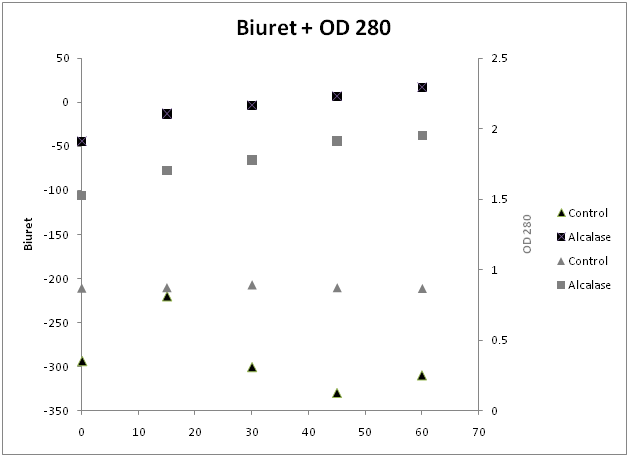
Figure 1 Biuret color (left y-axis, black) and OD 280 (right y-axis, grey). Markers are Alcalase treated (■) samples and untreated control (▲). Values are averages of at least three replicates.
Wet balance
The increase in supernatant weight shows the increase in soluble material during the course of hydrolysis (Table 1). This increase is directly related to the activity of the enzyme on the system. The lack of a parallel increase in the control indicates that any endogenous proteases either are not active at 50°C or have limited activity, and did not result in a measurable effect. The increase in solubility follows previously reported studies on salmon protein hydrolysis.20 Protein solubility is an important property because it partially dictates the functional properties, and thus the applications, of any potential protein based resultant products.20
TCA Soluble peptides
Previous studies on Alcalase have shown it to be an endoprotease.29,30 According to the commercial supplier of alcalase, DSM, the function of this protease limits the extent of the reaction to between 15-25%.26 Under the conditions studied here, the extent of the enzymatic reaction was at a maximum of 11.6% as measured by TCA soluble peptides. When using a theoretical maximum of 15%,26 after 1 hour the digestion was 77% complete (Figure 1). The time limit of this study, providing 11.6% digestion indicates that both functional properties20 of the hydrolysates and radical scavenging properties at this level of DH31 would likely be present.
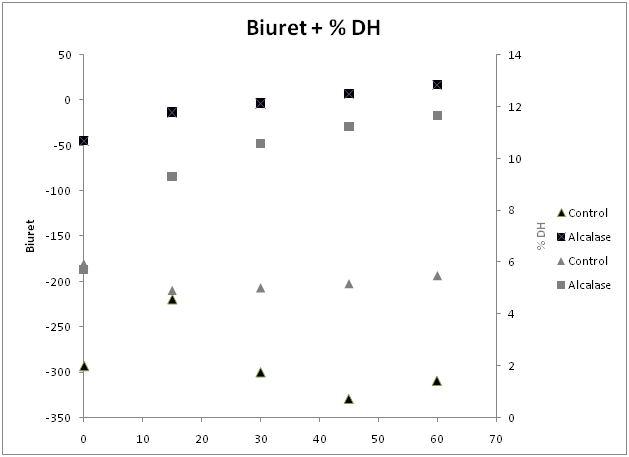
Figure 2 Biuret color (left y-axis, black) and degree of hydrolysis (right y-axis, grey). Markers are Alcalase treated (■) samples and untreated control (▲). Values are averages of at least three replicates.
Biuret color
Figure 3 shows an increase in color, quantified as the product of chromameter values *a and *b, during the hydrolysis reaction versus the control. When these results are compared to the other methods of ascertaining hydrolysis reaction progress used in this study, the biuret color had a linear correlation (R2) with all other methods greater than 0.9 (Table 2). This high degree of correlation shows that the biuret method was an effective substitute for these other, generally more traditional methods. Additionally it is cheap, rapid and simple to determine.
Our biuret color method specifically determined the amount of soluble protein fragments and peptides produced during the enzymatic hydrolysis of pollock flesh. The TCA soluble protein fragments and peptides of the controls were found to be between 5-6% and did not show an increase in biuret color.
Preliminary studies with other exogenous enzymes showed similar results; the pink biuret color increased during the time course of hydrolysis. The use of exogenous enzymes on fish proteins has been widely studied and recommended as a method of byproduct processing for value addition.4,20,32
R2 vs |
TCA Soluble Peptides |
OD280 |
Solids |
Supernatant |
0.975 |
0.974 |
0.956 |
0.959 |
Table 2 R2 linear correlation values of Biuret color vs TCA soluble peptides (%DH), absorbance at 280nm (OD 280), solids content of the supernatant after centrifugation (Solids) and the weight of the supernatant after centrifugation (Supernatant). The R2 linear regression comparison between Biuret color and the other methods used was performed in Excel with the biuret results on the y axis and the correlative metho
Rapid determination of hydrolysis utilizing the biuret reagent
These methods were based on an assumption that the increased solubility of previously insoluble proteins was due to the increasing concentrations of soluble protein fragments and peptides produced by the enzymatic digestion of the substrate material. This is a valid assumption; however a direct, rapid, and accurate measurement of this change remains a challenge. The most commonly cited accurate measurement is the increase in the dry solids content of the soluble fraction. To determine this solid content according to AOAC methods24 requires drying the soluble fraction at 105°C for 24 hours. There are rapid moisture content determination methods that require 1-2 hours per sample to complete. These provide the accuracy desired, however the time required can make them unworkable because the hydrolysis step may be completed in substantially less time.
The use of Bradford protein reagents to measure the degree of hydrolysis largely depends on a minimum length of the protein chain (BIO-RAD).33 This generally does not allow the rapid determination of protein content in the soluble phase because hydrolytic reactions can lead to protein fragments of varied lengths depending on the recognition of a specific cleavage site by the enzyme, enzyme concentration and other reaction conditions. This principle remains true with the simple protein quantification method relying on the biuret reaction.25 However the biuret reaction is unique in that it undergoes a visible color shift in the presence of the peptide products of hydrolysate reactions.
The biuret reaction shows a color shift from blue to purple during standard protein quantification. In the presence of hydrolyzed protein fragments and peptides the color shift is from blue to pink. This reaction can be monitored with a Minolta chromameter (Figure 1) & (Figure 2). Good correlations between this biuret method and other measurements of hydrolysis show that it is sufficiently accurate to be used as a rapid method for the determination of the reaction progress in enzymatic hydrolysis (Table 2). The advantages of this biuret method include increased sample through-put, minimal equipment and reagents that are environmentally safe in the quantities used.
The biuret method also presents other advantages. The production of minimally hydrolyzed protein fragments would allow greater utilization of the currently discarded processing byproducts as high quality protein feed ingredients; protein feed ingredients currently not widely available. Limited hydrolysis will liquefy seafood processing byproducts allowing for fish oil separation as well as fish bone removal while maintaining the high quality of the protein fragments retained.
Determination of reaction progress in enzymatic hydrolysis using this modified biuret method allows seafood processing facilities a simple, rapid and inexpensive method to control the hydrolytic reaction. The startup cost for this test is minimal, including only the reagents, a colorimeter and test tubes. Personnel are easily trained to use the method, because it requires only the mixing of the hydrolysate samples with one testing liquid. The utility and ease of this analytical procedure provides seafood processing plants in remote locations throughout Alaska a useful technology that can be safely conducted on site.34
None.
The author declares no conflict of interest.

©2016 Davenport, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.