Journal of
eISSN: 2373-4310


Research Article Volume 5 Issue 3
1Department of Food Science and Nutrition, King Saud University, Saudi Arabia
2Cardiff School of Health Sciences, Cardiff Metropolitan University, UK
Correspondence: Alhaj OA, Department of Food Science and Nutrition, College of Food and Agricultural Sciences, King Saud University, P.O. Box 2460, Riyadh 11451, Saudi Arabia
Received: October 18, 2016 | Published: December 2, 2016
Citation: Alhaj OA, Irshad I, Kanekanian AD. Hypocholesterolemic and angiotensin i converting enzyme-inhibiting activity of trypsin-hydrolysed bovine casein. J Nutr Health Food Eng. 2016;5(4):653-657. DOI: 10.15406/jnhfe.2016.05.00177
Nowadays, the need for new dietary containing some bioactive peptides with specific amino acid sequences increased to express biological functions. We investigated the hypocholesterolemic effect and ACE-inhibiting activity of bovine casein hydrolysates with different molecular weights. The bovine casein was hydrolysed with trypsin for different amounts of time to release bioactive peptides from the casein. Then, crude casein hydrolysate was ultrafiltered using membranes with two different molecular-weight cut offs (1kDa and 10kDa) to determine the hypocholesterolemic effects and ACE-inhibiting activities of peptides with different molecular weights. The results showed that a higher level of hydrolysis (more active bioactive peptides) was associated with greater hypocholesterolemic and ACE-inhibiting effects in all the tested samples. The unhydrolysed crude casein reduced the cholesterol level in ortho-phthaldialdehyde (OPA) assays by 39.5%, while the 10kDa and 1kDa permeates of casein hydrolysed with trypsin for 4h reduced the cholesterol level by 50.7 and 69.6%, respectively. The unhydrolysed crude casein and the 10kDa and 1kDa permeates of casein hydrolysed by trypsin for 4h inhibited ACE activity by 18.9%, 29.7%, and 51.4%, respectively.
Keywords: bovine casein hydrolysate, trypsin enzyme, bioactive peptides, hypocholesterolemic, ace inhibitory
Coronary heart disease is one of the leading causes of death in the United States, Japan, and the European Union.1 Nearly 30% of the population of Britain suffers from moderately high cholesterol levels. An effective approach to reduce cholesterol levels would likely have a significant effect on coronary heart disease-related mortalities. The use of drugs is an expensive exercise and often entails unwanted side effects. Therefore, some researchers have sought dietary solutions that could be used in lieu of drugs. It was reported that food products containing angiotensin converting enzyme (ACE) peptides played a role in treating mild hypertension.2 Similarly, the consumption of a diet low in fat and saturated fatty acids has shown effectiveness in reducing blood cholesterol levels. Poor consumer satisfaction with low-fat diets has, however, resulted in further efforts to find other dietary solutions that could effectively reduce cholesterol levels.3 Various food commodities have been examined, including milk proteins, which have shown antithrombotic activities in animal trials.4 Casein is more susceptible than whey proteins to proteolysis because of its open and flexible structure.5 The degree of hydrolysis and the enzyme used for hydrolysis are crucial in determining the functional products of the hydrolysis.6 Controlled enzymatic hydrolysis reportedly improved the functional properties of casein.7 The ability of hydrolysed casein to reduce cholesterol levels has been hypothesised to be due to the direct interaction of Arg and Tyr with cholesterol, forming cholesterol apoprotein complexes.8 On the other hand, ACE inhibitors, including milk peptides, decrease the conversion of angiotensin I to angiotensin II, which leads to a decrease in bradykinin metabolism and, consequently, a decrease in arterial blood pressure. We aimed to investigate the hypocholesterolemic effect and ACE-inhibiting activity of bovine casein hydrolysates with different molecular weights.
Materials
Bovine sodium caseinate, trypsin, 0-phthalaldehyde reagent, trifluroacetic acid (TFA), cholesterol, potassium hydroxide, n-hexane, ß- cyclodextrin, sulphuric acid, sodium hydroxide, hydrochloric acid, boric acid, sodium chloride, ACE, Hip-His-Leu, and ethyl acetate were purchased from Sigma Aldrich (U.K.). HPLC water, ethanol, and acetonitrile (ACN) were purchased from Fisher Scientific (U.K.). Ultrafiltration cells and Diaflo Ultrafiltration Membranes of 10kDa and 1kDa were purchased from Amicon Inc. The bicinchoninic acid (BCA) kit was purchased from Pierce (USA).
Bovine casein hydrolysis using proteolytic enzymes
Bovine casein solution was hydrolysed by trypsin for 4h at a ratio of 1:100. Throughout the experiment, the solution temperature and pH were maintained at 37°C and 7.5, respectively. The trypsin was inactivated by thermal treatment in a water bath for 5–6min at 90°C. All samples were kept at -22°C in a freezer prior to further treatment.
Determination of the level of protein hydrolysis
The ortho-phthaldialdehyde (OPA) method a rapid, sensitive, and convenient spectrophotometric assay—was used to determine the degree of hydrolysis. The absorbance of samples was measured spectrophotometrically at 340nm as described by Church et al.,9 and modified by Alhaj et al.,10
The degree of hydrolysis was determined using the following equation:
where S is the hydrolysed sample reading formed by trypsin, C is the unhydrolysed sample reading as a control, and D is the difference in absorbance between the sample reading after 4 h (taken as 100% hydrolysis) and the unhydrolysed sample reading at 0 h (taken as 0% hydrolysis).
Ultrafiltration of bovine casein hydrolysate and standardisation of protein concentration
Crude samples of casein hydrolysates were ultrafiltered using an ultrafiltration unit (Amicon) fitted with Diaflo Ultrafiltration Membranes of 1kDa and 10kDa. The ultrafiltration cell was operated under a pressure of 55 psi imposed by nitrogen gas. The collected crude permeates (1kDa and 10kDa) were freeze-dried using a freeze dryer (Savant MicroModulyo) and stored at -22°C. The protein concentrations of the crude casein and permeates were determined using the bicinchoninic acid (BCA) protein assay kit. All samples were standardised to a concentration of 10mg/mL using double-distilled water.
Reverse-phase high-performance liquid chromatography (RP-HPLC) profile of the casein peptides
The collected permeates of hydrolysed and unhydrolysed casein were fractionated by RP-HPLC using an Agilent Technologies 1200 Series system with a C18 column (250mm×7.75mm, 300Å, ACE, Hichrom LTD, UK) at a controlled temperature of 21.5°C. The peptides were eluted by gradient elution with solvent A for 3min, followed by a linear gradient of solvent B from 0% to 60% for 80min, and finally 100% solvent B for 10min. Solvent A comprised 95% water and 5% ACN containing 0.1% TFA; solvent B comprised 85% ACN and 15% water containing 0.1% TFA. The peptides were eluted at a flow rate of 1ml/min. Absorbance was measured at 220nm.
Evaluation of the cholesterol-reducing effects of the bioactive peptides derived from the bovine casein hydrolysate fractions
A method described by Rudel and Morris11 and modified by Mishra and Prasad12 was followed to determine the ability of the crude casein and the casein hydrolysate to reduce the cholesterol level. Cholesterol solution was prepared by dissolving cholesterol in β-cyclodextrin at a concentration of 1.5mg/85µL. An aliquot of 85µL cholesterol solution was transferred into each test tube and 100µL of each sample was added accordingly. After incubating samples for 1.5h in water bath at 40°C, 1.5mL of 95% ethanol followed by 1mL 33% KOH were added. n-hexane was added to the mixture, then 1.25mL of hexane top layer was transferred into new clean and dry test tubes then evaporated in water bath at 60°C under nitrogen gas. Dried residues were dissolved in 4mL o-phthalaldehyde reagent (OPA), and then 2mL of concentrated sulphuric acid was gently transferred. Absorbance was measured at 550nm using a spectrophotometer.
The percentage cholesterol reduction was calculated using the following equation:
Where, C is the average cholesterol reading for the unhydrolysed sample as a control, and S is the average cholesterol reading for the hydrolysed samples. The samples were: Blank (OPA + sulphuric acid), Control (dried residues of unhydrolysed sample + OPA + sulphuric acid), and Sample (dried residues of hydrolysed sample+OPA+sulphuric acid). All experiments were conducted in triplicate and analysed for standard deviation and p value using QI Macros statistical software. All data was assumed to be normally distributed and results were deemed significant when p<0.05 throughout.
Determination of the ACE-inhibiting activity of the casein hydrolysates
The method described by Pihlanto-Leppala13 was followed to determine ACE inhibition. An aliquot of 300µL of Hip-His-Leu added to each test tube, followed by the 50µL of sample at a concentration of 10mg/mL. Reaction was initiated by adding 6µL of 1mU ACE and incubated for 30min at 37°C. Reaction was stopped by adding 250µL of 1M HCL followed by 1.7mL of ethyl acetate. After mixing, 1.1mL of upper ethyl acetate layer was transferred into clean dried test tubes, and then evaporated under nitrogen gas in water bath at 60°C. Residues were dissolved in 1mL deionised water, then absorbance was measured at 228nm using a spectrophotometer. The ACE inhibition was measured using the following equation:
% ACE inhibition S−CD ×100
Where C is the average absorbance reading of the unhydrolysed sample as a control, S is the average absorbance reading for the samples, and D is the difference in the average absorbance reading between the unhydrolysed and hydrolysed casein samples. The tested samples were: Blank (deionised water), Control: (dried residues of the 0h hydrolysis sample+deionised water), and Sample (dried residues of the 0.5h, 1h, and 4h hydrolysis samples+deionised water). All experiments were conducted in triplicate and analysed for standard deviation (SD) and p value using the QI Macros statistical software. All data was assumed to be normally distributed and results were deemed significant when p<0.02 throughout.
Tryptic hydrolysis and the RP-HPLC profile of casein
The overall amount of tryptic hydrolysis in the case in samples increased steadily over time. For the purpose of calculating the level of hydrolysis, the amount of hydrolysis measured at 4h was taken as 100% hydrolysis Alhaj et al.,10 even though not all of the protein in the sample was necessarily hydrolysed after 4h. Thus, the trypsin produced 73.3% hydrolysis in 30min and 83.33% hydrolysis in 1h. Trypsin has specificity for Arg and Lys Antal et al.,14 which account for about 12% of casein Belitz et al.15 The profile of the casein peptides was also determined by RP-HPLC to verify the hydrolysis capability of the trypsin. The chromatogram of unhydrolysed casein shown in Figure 1(A) has an elution of ca.14peaks at 0h incubation. After 50min, all of the peptides except for two were eluted, indicating strong hydrophobicity. The chromatogram of casein after 30min hydrolysis shown in Figure 1(B) has ca. 51peaks. Likewise, the chromatogram after 1h hydrolysis shown in Figure 1(C) has ca. 51 peaks, although there is more hydrophilicity evident after 1h hydrolysis than after 30min hydrolysis. Similarly, the chromatogram after 4h hydrolysis shown in Figure 1(D) has ca. 61 peaks, which indicates extensive hydrolysis. Thus, increased hydrolysis time was associated with the release of more peaks from the native protein. Compared with that of the peptides eluted from 30min hydrolysis and 1h hydrolysis, the retention time of the peptides eluted from 4h hydrolysis was decreased, because all of the peptides were eluted before 67min. All of the above results reflect that the peaks with the maximum retention time, which are hydrophobic, were released at 0h hydrolysis that the progress of the hydrolysis led to the release of more peptides with less retention time, which are more hydrophilic, and that the most hydrophilic peptides were released at 4h hydrolysis. Zhong et al.,16 determined the difference in hydrophobicity among Arg, Lys, Phe, Tyr, and Leu. Arg and Lys were found to be less hydrophobic than Phe, Tyr, and Leu. The hydrophobicity determined for Arg and Lys was 3.1 and 6.2, respectively, whereas that determined for Phe, Tyr, and Leu was 11.1, 12, and 11.1, respectively. Based on these results, the peptides released from casein by trypsin hydrolysis tended to be hydrophilic.
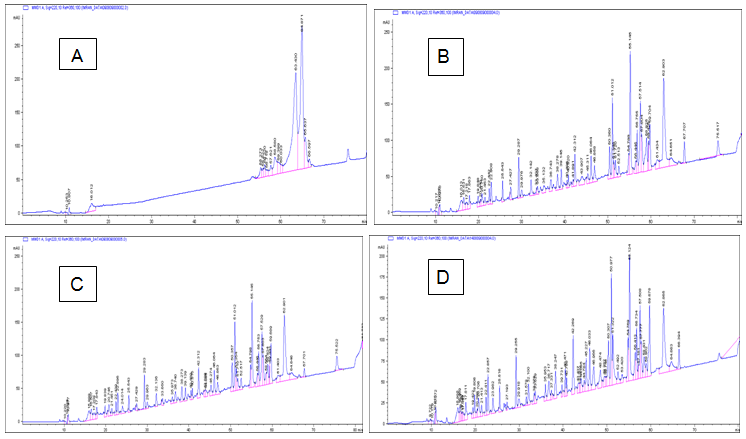
Figure 1 Qualitative RP-HPLC separation of bovine casein hydrolysed by trypsin for 0 h (A), 0.5 h (B), 1 h (C), and 4 h (D).
Cholesterol-reducing activity of the casein hydrolysate formed by trypsin
Figure 2 shows the cholesterol-reducing activity of the casein hydrolysates in the 1KDa and 10KDa permeates. There was a steady increase in the cholesterol-reducing activity over time, with significant differences among the crude casein, the 1KDa permeate, and the 10KDa permeate. Unhydrolysed crude casein showed 23.2% cholesterol-reducing activity after 30min hydrolysis, which increased to 39.5% after 4h hydrolysis due to the release of bioactive peptides by the enzymatic activities of contaminating bacteria in the raw milk Otte et al.,17 or by the endogenous enzymes of the milk Gobbetti et al.18 On the other hand, the 10KDa permeate showed 44.9% cholesterol-reducing activity after 30min hydrolysis, which is almost a twofold increase relative to that of the unhydrolysed crude casein. After 4h hydrolysis, the 10kDa permeate showed 50.7% cholesterol-reducing activity, which is significantly (P<0.05) higher than that of the unhydrolysed crude casein. These results are in agreement with the findings of Sugano et al.,19 who showed that the hypocholesterolemic peptides of soybean protein had molecular masses of 1–10kDa. In that case, the hypocholesterolemic properties of the peptides were attributed to the inhibition of cholesterol absorption and increases in the faecal excretion of steroidsmine & Shahidi.20 The 1kDa permeate exhibited 55.1% cholesterol-reducing activity after 30min hydrolysis, which is significantly (P<0.05) higher than that of the crude unhydrolysed casein (23.2%) and that of the 10kDa permeate (44.9%) after the same amount of time. After 4h hydrolysis, the cholesterol-reducing activity of theKDa permeate increased to 69.6%, which is also significantly (P<0.02) and higher than crude unhydrolysed casein and that of the 10kDa permeate after the same amount of time. Moreover, the cholesterol-reducing activity of the 1kDa permeate after 30min hydrolysis was higher than that of the unhydrolysed crude casein (39.5%) and the 10kDa permeate (50.7%) after 4 h hydrolysis. These results show that peptides with 1kDa molecular mass have more cholesterol-reducing activity than peptides with 10kDa molecular mass (P<0.05) and unhydrolysed crude casein (P<0.02). Our results are in agreement with those of Alhaj et al.,10 who reported that peptides with molecular mass less than 1200 Da are superior to large peptides in reducing cholesterol. Permeates of 1–10kDa are superior to unhydrolysed crude casein in reducing cholesterol, because small peptides are more easily absorbed into the intestine than large peptides, which makes them more effective in cholesterol reduction compared with the larger peptides Shah.21 The cholesterol-reducing activity of the 1kDa permeate is attributed to the high Arg and Lys content resulting from the trypsin hydrolysis Antal et al.,15 The interaction between cholesterol and Arg was suggested to occur by hydrogen bonding between the hydroxyl groups of cholesterol and the guanidine groups of Arg residues Li et al.,8 Likewise, the ε-amino groups of Lys are able to form hydrogen bonds with the hydroxyl groups of cholesterol. Thus, Arg can establish two hydrogen bonds per side chain, while Lys can establish one. Therefore, the bonding between cholesterol and Lys might not be as stable as that between cholesterol and Arg.
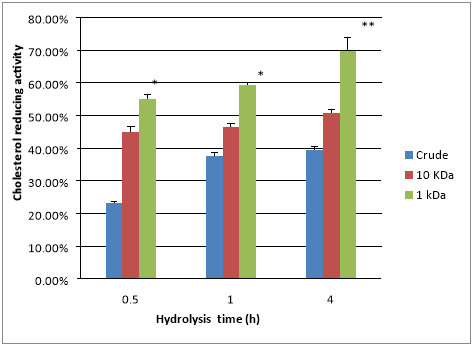
Figure 2 Cholesterol-reducing activity of unhydrolysed crude casein and of 10 kDa permeate and 1 kDa permeate of sodium caseinate (10 mg/mL) hydrolysed by trypsin at 37 °C for up to 4 h at pH 7.5 with an enzyme to substrate ratio of 1:100. Error bars indicate the standard deviation. n = 3, ** (P<0.02), * (P<0.05).
ACE-inhibiting activity of crude hydrolysates formed by trypsin
The ACE-inhibiting activities differed significantly among unhydrolysed crude casein, the 10KDa permeate, and the 1kDa permeate (Figure 3). The unhydrolysed crude casein showed 13.5% ACE-inhibiting activity at 0.5h, which increased to 19% after 4h of hydrolysis because of the production of ACE-inhibiting peptides by the enzymatic activities of contaminating bacteria in the raw milk Otte et al.,17 or by the endogenous enzymes of the milk Gobbetti et al.,18 The 0.5h and 4h tryptic hydrolysates of the 10KDa permeate showed 25.7% and 29.7% ACE inhibition, respectively. The 0.5h and 4h tryptic hydrolysates of the 1kDa permeate showed 34.4% and 51.4% ACE inhibition, respectively. These results clearly reflect that the ultrafiltration treatment of the crude hydrolysate isolated the peptides with higher ACE-inhibiting activity. It has been reported that the ultrafiltration of the thermolysin hydrolysis products of bonito increased the ACE-inhibiting activity by two fold Fujita et al.,22 Our results show that the 10KDa permeate exhibited better ACE inhibition than the unhydrolysed crude casein, while the 1kDa permeate exhibited better ACE inhibition than the 10kDa permeate. In a similar study, sodium caseinate was hydrolysed using trypsin and Bifidobacterium animalis subsp. Lactis (Bb-12) separately Alhaj.23 The fractions with the lowest molecular weights exhibited the greatest ACE inhibition, and the molecular masses of the ACE-inhibiting peptides were less than 1kDa. The reason for the ACE inhibition might be attributed to the purification of specific peptides that have exposed Arg and Lys residues. Antal et al.,14 expressed the similar view that trypsin has a cleaving specificity towards Arg and Lys. So it could be assumed that among the released terminal amino acids, Arg and Lys were involved in the ACE inhibition. It has been reported that positively charged amino acids at the ultimate C-terminal position such as Arg (guanidine group) and Lys (ε-amino group) appear to effectively inhibit ACE Li et al.,24
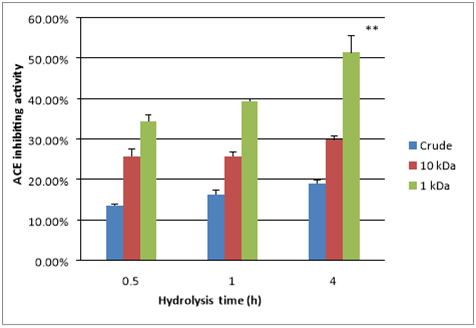
Figure 3 ACE-inhibiting activity of unhydrolysed crude casein and of 10 kDa permeate and 1 kDa permeate of sodium caseinate (10 mg/mL) hydrolysed by trypsin at 37 °C for 4 h at pH 7.5 with an enzyme to substrate ratio of 1:100. Error bars indicate the standard deviation. n = 3 **, (P<0.002).
Chobert et al.,25 found that the ACE-inhibiting activity of tryptic hydrolysates of ovine β-lactoglobulin increased with the degree of hydrolysis. In another study, Qula milk casein was hydrolysed for 60, 120, 180, 240, 300, and 360min using alcalase. The 240min (4h) hydrolysate showed the greatest ACE inhibition. The hydrolysates were fractionated using ultrafiltration, and two novel ACE-inhibiting peptides with respective molecular masses of 550Da and 566.4Da were identified Mao et al.,26 These results clearly reflect that hydrolysis improved and enhanced the ACE-inhibiting activity and that the degree of hydrolysis has a positive correlation with ACE inhibition. Moreover, the use of ultrafiltration isolated the fractions that exhibited higher ACE inhibition activity than the crude hydrolysates.
Our results show that unhydrolysed crude casein reduces cholesterol and ACE-inhibiting activity by up to 39.5% and 19%, respectively. Tryptic hydrolysis improves and enhances the cholesterol-reducing and ACE-inhibiting activity to up 69.6% and 51.4%, respectively. Moreover, the use of ultrafiltration fitted with 1KDa and 10KDa cut off membranes resulted in more functional properties than did the use of crude unhydrolysed casein. The peptides with molecular mass less than 1KDa had the maximum potential for cholesterol reduction and ACE-inhibiting activity. The stability of the bioactive proteolytic fractions in the pilot-scale processing and manufacturing of products need to be evaluated.
None.
Author declares that there is no conflict of interest.

©2016 Alhaj, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.