Journal of
eISSN: 2373-4310


Research Article Volume 13 Issue 1
1Amity Institute of Organic Agriculture, Amity University, India
2Amity Institute of Herbal Research & Studies, Amity University Uttar Pradesh, Noida, UP, India
Correspondence: Charu Gupta, Amity Institute of Herbal Research & Studies, Amity University Uttar Pradesh, Sec-125, Noida-201313, UP, India
Received: February 24, 2023 | Published: March 8, 2023
Citation: Nag K, Gupta C. Antimicrobial and antioxidant properties of Trachyspermum ammi leaf extracts against some plant pathogens. J Nutr Health Food Eng. 2023;13(1):1-4. DOI: 10.15406/jnhfe.2023.13.00363
Aim: The goal of the current study was to assess the antimicrobial and antioxidant effects of the leaf extracts of Trachyspermum ammi using the agar well diffusion method and phenol estimation technique against some plant pathogens. We have observed from the literature review that very little information is available on the antimicrobial properties of leaf extracts of Trachyspermum ammi. Therefore, this is the first complete study to report on the antimicrobial and antioxidant effects of the leaf extracts of Trachyspermum ammi against common plant pathogens.
Material and methods: A total of three extracts using different solvents (ethyl acetate, ethanol, and 50% aqueous ethanol) are prepared using the soxhlet apparatus and the maceration technique. The antimicrobial activity of the leaf extracts was studied against common plant pathogens like Staphylococcus epidermidis, Bacillus subtilis (NCIM 2920), Staphylococcus aureus (NCIM-5345), Escherichia coli (NCIM 5346) and Pseudomonas aeruginosa. The fungal species used in the present study were Fusarium oxysporum (NCIM 1008), Aspergillus flavus (NCIM 1316), Penicillium citrinum (NCIM 1435), and Rhizopus stolonifera (NCIM 1139). The antioxidant activity was assessed by the Folin-Ciocalteau method using the standard gallic acid solution as a reference.
Results: It was found that the ethyl acetate leaf extract prepared using soxhlet apparatus exhibited a wide range of antimicrobial activity against all groups of bacteria with the highest inhibition zone produced against Escherichia coli (NCIM 5346) and Pseudomonas aeruginosa i.e, 35mm and 30mm respectively. In the case of fungus, both ethanol and ethyl acetate extracts show good percentages of inhibition for Fusarium oxysporum (NCIM 1008) and Penicillium citrinum (NCIM 1435) i.e, 0.77%, 0.77%, and 0.40%,0.42% respectively, whereas exhibited no and partial inhibition for Rhizopus stolonifera (NCIM 1139) and Aspergillus flavus (NCIM 1316) respectively. The leaf extracts show a significant antioxidant activity of 15.52mg/g of GAE.
Conclusion: In the current investigation, the results illustrate that the leaf extracts of Trachyspermum ammi exhibit a wide range of antimicrobial and antioxidant activity and can potentially be used as a natural biopreservative.
Keywords: Trachyspermum ammi (ajwain), antibacterial, antifungal, antioxidant, total phenolic content, biopreservative
Trachyspermum ammi L. is a medicinal plant that belongs to the family Apiaceae comprising 270 genera and species1 Ajwain tends to grow in regions that are dry and barren. This spice is indigenous to Egypt but is also grown in many parts of South & West Asia, including Iran, India, Pakistan, and other countries. Gujarat and Rajasthan are regions within India that are well known for cultivating ajwain.2 It is commonly known as Ajwain in Hindi,3,4 Bishop's weed in English, Yamini in Sanskrit, Lodhar Bengali in Punjabi, Ajma in Gujrati, Kath in Kashmiri, and Omam in Tamil.5
The name ajwain is derived from the Sanskrit words yavanaka or ajomoda. Ajwain is an ancient and well-known Ayurvedic spice.6 It is an annual, aromatic, erect herb bearing white flowers and small brownish fruit with many leafy branches and small feather-like leaves.
Ajwain seed, the most commonly used part of the plant is a popular spice in curries because of its distinct aromatic scent and flavor. Most of the research studies such as antibacterial, anti-mycobacterial, and antiviral effects have been done on the seeds of this plant but very limited studies have been done on its leaves. Thus, the objective of the current study was to investigate the antimicrobial and antioxidant effects of leaf extracts of ajwain genotypes commonly produced in India.7,8
Ajwain (Trachyspermum ammi) is a plant with medicinally useful chemicals that can be used for various therapeutic purposes. Medicinal properties of the plant include antioxidant, antispasmodic, antimicrobial, and antifungal9 reported the active ingredients of the ajwain plant include six major chemical compounds including 49% thymol, 30.8% γ-terpinene, 15.7% p-cymene, 2.1% b-pinene, 0.8% myrcene, and 0.7% limonene. The main component is thymol (35–60%), a strong germicide, antispasmodic, and fungicide agent.10 However, sometimes γ-terpinene and p-cymene exceed the thymol content.11
Globally, an increasingly serious and horrifying issue in modern agriculture has been the resistance of dangerous bacteria to various antimicrobial treatments. Plant extracts containing phytochemicals, which have both antibacterial and antioxidant capabilities, must be used to control this issue because synthetic compounds can be hazardous in nature. Plant secondary metabolites play an important role in plant-insect interactions. Some compounds extracted from Trachyspermum ammi plant have insecticidal activity. In previous studies, the alcoholic seed extract of T. ammi was found to be effective against different developmental stages of Aedes aegypti i.e. larva and pupa and considered an eco-friendly remedy. The mortality rate of larvae was three times faster than the pupa stage12 hence, we can observe T. ammi insecticidal properties as well.
Ajwain is an exceptionally trustworthy plant source because it is abundant in components with potential bioactivity.13 In recent years much attention has been given to non-chemical systems for seed treatment to protect them against many plant pathogens.14 Since our country has favorable conditions for agriculture and allows the production of medicinal plants at relatively low cost, there is the possibility of using them in the fight against plant pathogens, along with other control methods that can prevent the indiscriminate use of antibiotics and pesticides.15 The results of the experimentation showed that leaf extract of ajwain has substantial anti-microbial action along with antioxidant properties. It has the potential to be utilized as a reliable supply of biocides for sustainable agriculture.
The antibacterial and antifungal activity was assessed using a growth inhibitory zone assay with the agar well diffusion technique after the extracts were produced.
Sample preparation
About 500 grams of locally available Trachyspermum ammi plant leaves were collected and oven dried for about 4 days until a constant weight was obtained. They were then crushed and made into a fine powder. The sample was stored at room temperature until further use.
Preparation of extracts
Extraction was carried out with a Soxhlet extractor using ethyl acetate. Its excess solvent was then removed using a vacuum rotatory evaporator. DMSO is added to the concentrated extract and stored at room temperature. Two other leaf extracts were prepared using the maceration technique, where 5gm of leaf powder is added to 50ml of ethanol and 50ml of 50% aqueous ethanol respectively, and kept aside for 3 days. After 3 days the extract was filtered and kept on the water bath for solvent evaporation. Once done, 2ml of DMSO is added and stored until further use.
Bacterial and fungal strains
Five bacterial strains (3 Gram-positive and 2 Gram-negative), mostly plant pathogens, were selected for the study. Gram positives bacteria were Staphylococcus epidermidis, Bacillus subtilis (NCIM 2920), and, Staphylococcus aureus (NCIM-5345), while Gram-negative bacteria were Escherichia coli (NCIM 5346) and Pseudomonas aeruginosa. The fungal species used in the present study were Fusarium oxysporum (NCIM 1008), Aspergillus flavus (NCIM 1316), Penicillium citrinum (NCIM 1435), and Rhizopus stolonifera (NCIM 1139) (Figure 1). The standard bacterial and fungal stock cultures were obtained from the culture collection of Amity University UP, India. The viability tests for each isolate were carried out by resuscitating the organism in nutrient agar medium and Sabouraud’s dextrose agar (SDA) medium respectively. The stock on nutrient agar medium (Hi Media, Mumbai, India) and potato dextrose agar medium were incubated at 37 °C for 24 h (bacteria) and 28 °C for 3-days (fungi) following storage at 4 °C until required for sensitivity testing.16
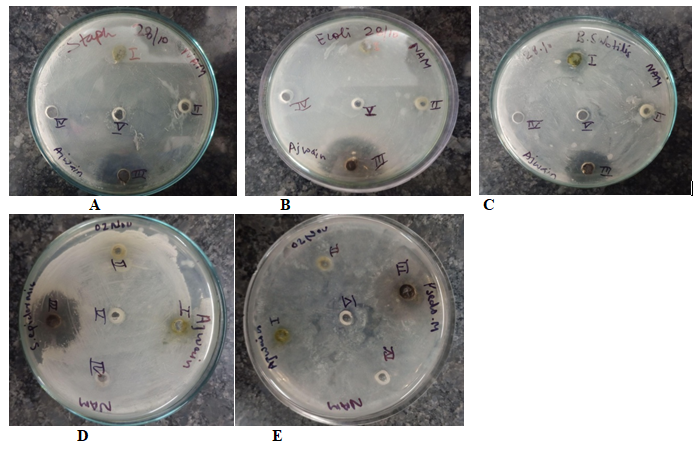
Figure 1 Zone of antibacterial inhibition of ajwain leaf extracts against (A) Staphylococcus aureus (NCIM-5345), (B) Escherichia coli (NCIM 5346), (C) Bacillus subtilis (NCIM 2920), (D) Staphylococcus epidermis, (E) Pseudomonas aeruginosa
.Antibacterial activity testing using agar well diffusion technique
The antibacterial activity of the T ammi. leaf extracts were determined by the agar well-diffusion method.3 A pure isolate of each bacterium was first subcultured in nutrient broth at 37 ºC for 24 h. Standardized inoculum (100 μL, 106 CFU/mL; 0.5 Mac-Farland) of each test bacterium was spread with the help of a sterile spreader onto a sterile agar plate to achieve a confluent growth. The plates were allowed to dry and a sterile cork borer (6.0 mm diameter) was used to bore the wells in the agar. Subsequently, 50 μL of each extract was introduced in wells of an agar plate. The plates were allowed to stand for 1 h for diffusion and then incubated at 37 ºC for 24 h. The inhibition zone diameter (IZD) was observed and measured in mm.
Antifungal assay
For the antifungal activity of T. ammi leaf extracts, sterilized SDA plates and a sterile cork-borer (6-mm diameter) was used to bore wells in the agar. 50 μL of ethanol and ethyl acetate extracts were introduced in separate plates to each peripheral well while a fungal disc was inoculated in the central well. A negative control (DMSO) was used in one peripheral well to compare the activity incubated at 28 °C. After incubation for 3-5 days at 28°C, the inhibition zones are measured in mm and recorded.
Determination of total phenolic content and antioxidant activity
The total phenolic content in the methanolic leaf extract of ajwain was determined spectrophotometrically with the Folin-Ciocalteau reagent.17 First, the crude leaf extract of the sample leaves was prepared by adding 100mg of dried leaf powder to 10 ml of methanol and left overnight. An Aliquot of sample in 0.1 mL was taken, made up 1ml using distilled water. Then 0.5ml folin-ciocalteau reagent and 1ml 20% Na2CO3 were added. The sample was left for 20min. at room temp. then 10ml D.W was added. OD was taken at max.720nm. Here, gallic acid 1mg/1ml with 3X dilution in a 1:5 ratio was used as a standard reference. The results were expressed as gallic acid equivalents (GAE) of dry matter in mg/g.
A total of five bacterial species and four fungal species were used to assess the antimicrobial activity of leaf extracts. The antimicrobial activities of the extracts were determined by the agar well-diffusion method against three Gram-positive bacteria and two Gram-negative bacteria (Table 1). The extracts were active against both Gram-positive and Gram-negative bacteria. They were most effective against Escherichia coli (NCIM 5346) followed by Pseudomonas aeruginosa with inhibition zone diameter (IZD) of 35.0 mm and 30.0 mm respectively. In contrast to the negative control (DMSO), the ethanolic leaf extract exhibited active inhibition against all bacteria with the highest zones against Bacillus subtilis (NCIM 2920) and Staphylococcus epidermidis of 15.0 mm and 12.0 mm respectively) (Figure 2).

Figure 2 Zone of antibacterial inhibition of ajwain leaf extracts against (A) Fusarium oxysporum, (B) Penicillium citrinum, (C) Rhizopus stolonifera
.Bacterial species |
Ethanol |
Ethyl acetate |
50% Ethanol |
DMSO |
Escherichia coli (NCIM 5346) |
10 |
35 |
- |
- |
Bacillus subtilis (NCIM 2920) |
15 |
25 |
- |
- |
Staphylococcus aureus (NCIM-5345) |
10 |
25 |
- |
- |
Pseudomonas aeruginosa |
10 |
30 |
- |
- |
Staphylococcus epidermidis |
12 |
25 |
- |
- |
Table 1 The mean value diameter of inhibitory zones (mm) with ajwain leaf extracts and one negative control (DMSO)
In the current investigation we are able to find out that ethyl acetate extract shows significant growth inhibition for all the bacteria. In contrast, ethanol extract shows little inhibition while 50% ethanol extract exhibits no inhibition at all.
The antifungal effects of extracts have also been investigated and a total of four test fungi were used (Table 2). Amongst fungi, the leaf extracts exhibited antagonistic activity against Fusarium oxysporum (NCIM 1008) and Penicillium citrinum (NCIM 1435) whereas, exhibited no and partial inhibition for Rhizopus stolonifera (NCIM 1139) and Aspergillus flavus (NCIM 1316) respectively. In the case of assessing the antifungal activities, we calculated the percentage of inhibition (PI) index for each fungal species, where:
Percentage of Inhibition (PI) = C-T/C*100
Where C= Radial growth of the pathogen in control
T= radial growth of the pathogen in treatment
In the results we observe partial inhibition (pi) in case of Rhizopus stolonifera.
Fungal species |
C = Growth of pathogen in control (DMSO) in mm |
T = growth of the pathogen in treatment in(mm) |
Percentage of inhibition (PI) |
|||
|
|
Ethanol extract |
Ethyl acetate extract |
Ethanol extract |
Ethyl acetate extract |
|
Fusarium oxysporum (NCIM 1008) |
90 |
20 |
20 |
0.77% |
0.77% |
|
Penicillium citrinum (NCIM 1435) |
70 |
42 |
40 |
0.40% |
0.42% |
|
Rhizopus stolonifera (NCIM 1139) |
90 |
pi |
pi |
pi |
pi |
|
Aspergillus flavus (NCIM 1316) |
90 |
90 |
90 |
0 |
0 |
|
Table 2 The mean value of PI (in %) with the two leaf extracts and one negative control
From the above calculation we can say that both ethanol and ethyl acetate extracts exhibit active inhibition against fungi Fusarium oxysporum (NCIM 1008) and Penicillium citrinum (NCIM 1435) with the percentage of inhibition (PI) 0.77%, 0.77%, and 0.40%,0.42% respectively. In contrast, we can observe that the extracts show no i.e, inactive inhibition against Aspergillus flavus (NCIM 1316) and partial inhibition against Rhizopus stolonifera (NCIM1139). The antifungal property of the leaf extracts can help in selectively eliminating fungal pathogens from a host with minimal toxicity to the host.18
For the calculation of the total phenolic content TPC the following formula was used:
GAE = 20*absorbance of sample/absorbance of standard,
= Xmg/0.1ml /25mg = X *100mg/10ml/25mg,
= X *100 mg/1000 /10ml/25mg,
= X *4 mg/ /10ml /1gm,
= X* 4mg /1gm.
Absorbance of the sample= 0.013
Absorbance of the standard = 0.067
Hence, X = 20*0.013/0.067 = 3.88
Therefore, GAE = X*4mg/g
= 3.88*4 mg/g
= 15.52 mg/g
Reported a strong relationship between total phenolic content and antioxidant activity in fresh fruits, vegetables, and grain products. Depending on the concentration used, phenolic compounds can act as either bactericidal or bacteriostatic agents.19 The concentration of polyphenols in ajwain leaf extracts has been found to influence antioxidant activity.20 Polyphenolic compounds are known to have antioxidant activity and it is likely that the antioxidant activity of the extracts is due to these compounds (Chatterjee et al., 2012). This activity is believed to be mainly due to their redox properties, which play an important role in adsorbing and neutralizing free radicals, quenching singlet, and triplet oxygen, or decomposing peroxides.21The level of the phenolic compounds in the methanolic leaf extract of ajwain was considerable hence, showing great antioxidant properties. These antioxidants provide protection against damage caused by free radicals.18
None.
Authors declare no conflict of interests.
None.

©2023 Nag, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.