Journal of
eISSN: 2373-4310


Editorial Volume 4 Issue 6
Dipartimento di "Scienze Biomediche, University of Messina, Italy
Correspondence: Francesco Cacciola, Dipartimento di "Scienze Biomediche, Odontoiatriche e delle Immagini Morfologiche e Funzionali", University of Messina, Via Consolare Valeria, 98125- Messina, Italy
Received: September 16, 2016 | Published: September 19, 2016
Citation: Cacciola F. Analysis of the carotenoid composition in overripe fruits by advanced liquid chromatography techniques. J Nutr Health Food Eng. 2016;4(6):544-545. DOI: 10.15406/jnhfe.2016.04.00153
Carotenoids represent a class of pigments, widely distributed in nature. They are well-known for their colorant and biological properties even though some studies have recently demonstrated some remarkable health-promoting functions.1,2 Carotenoids are classified into two main classes: hydrocarbons, known as carotenes (e.g., b-carotene, lycopene), and oxygenated carotenoids, known as xanthophylls (e.g., b-cryptoxanthin, lutein).3,4 Moreover, during ripening, etherification greatly increases in more stable forms.5
A recent study reported the carotenoid composition and stability in tree overripe fruits, namely hybrid persimmon-apple, banana (pulp and peel) and nectarine for the first time, also evaluating whether post-climacteric biochemical changes were linked to carotenoid degradation in the investigated fruits. These wastes still represent an important source of bioactive, and their use could be diverted towards either in the animal feed production, or to the recovery of purified molecules for nutraceutical purposes. Since they occur in complex mixture for their analyses a very advanced analytical liquid chromatography technique was employed. Such a technique is called ccomprehensive two-dimensional liquid chromatography (LC×LC) and it is based on the combination of two independent separation steps with orthogonal selectivity. In LC×LC, a primary column is connected to one or more secondary column by means of a switching valve as an interface. The function of the interface is to isolate continuous fractions of the first dimension column (1D) effluent and then release them onto the second dimension (2D) column; in this way the entire sample is analysed in both dimensions, and very high values of peak capacities are obtained.6,7 In that study a normal phase × reversed phase (NP×RP) set-up was developed, consisting of a micro-bore (1.0mm I.D.) cyano column for the 1D separation, interfaced to a 2D C18 column packed with fused-core particles (30×4.6mm; 2.7mm), coupled with both photodiode array (PDA) and mass spectrometry (MS) detectors.8
On the one hand, chromatography on the cyano stationary phase allowed a good separation of the carotenoids in seven groups of different polarity in the first dimension, namely hydrocarbons, mono-ol-esters, di-ol-di-esters, di-ol-mono-esters, free-mono-ols, free-di-ols, free-epoxy-di-ols. On the other hand, the secondary C18 column allowed the separation of carotenoids within each class, according to their increasing hydrophobicity and decreasing polarity (for components of the same class, the elution order increases with the number of carbon atoms of the fatty acid chain). In Figure 1 A-B (top and down), typical contour plots of two representative samples namely overripe hybrid persimmon-apple and nectarine at 450nm with peak identification, are shown.
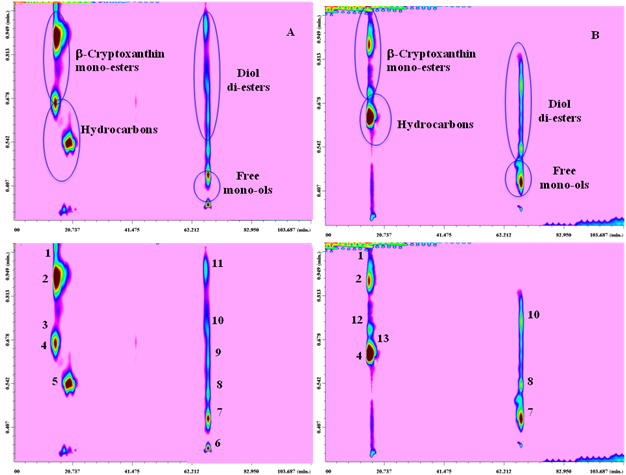
Figure 1 Normal-phase LC × Reversed-phase LC contour plots, indicating the chemical classes of the samples investigated (A and B top) along with peak identification (A and B down) for hybrid persimmon-apple and nectarine, respectively.
1: β-Cryptoxanthin-C16:0, 2: β-Cryptoxanthin-C14:0, 3: β-Cryptoxanthin-C16:1, 4: β-Carotene, 5: Lycopene, 6: n.d., 7: β-Cryptoxanthin, 8: Anteraxanthin-C14:0, 9: Anteraxanthin-C14:0-C14:0, 10: Lutein-C14:0-C14:0, 11: Zeaxanthin-C14:0-C14:0, 12: Z-β-Carotene, 13: β-Cryptoxanthin-C18:1
Identification of the separated compounds was achieved by means of both PDA and MS detection (through APCI ionization). In the case of carotenoids, MS detection represents a powerful tool and the operation of the interface under both positive and negative mode offers the double advantage of improved sensitivity and/or identification power. MS spectra obtained under negative ionization mode do show very intense pseudo-molecular ions [M.]-, which make identification of low-abundant components easier; on the other hand, abundant fragmentation is generally observed under positive APCI ionization, especially for carotenoid esters, whose fragment ions can help in structure elucidation. The complementary information attained allowed to discriminate between compounds showing similar (or nearly identical) UV-absorption properties, arising from the same chromospheres groups. The studied fruits are representative of tropical and temperate zone fruits and are considered as climacteric fruits since these fruits can be ripened after harvest. In that study also the evaluation of post-climacteric biochemical changes due to carotenoid degradation in the investigated fruits was taken into account. The carotenoid profile for the overripe kakimela, reported for the first time, highlighted how the hydrocarbons, b-carotene and lycopene, and the various identified xanthophylls mono- and di-esters were stable in the sample. The stability of the main carotenoid in overripe nectarine,9 b-carotene, was observed in this study together with other minor carotenoid in this fruit like cis b-carotene and the xanthophylls esters.
None.
Author declares that there is no conflict of interest.

©2016 Cacciola. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.