MOJ
eISSN: 2381-182X


Chlorellaand Spirulina are the two of the most well-known microalgae genus. Both microalgae genus have a significant content of proteins, vitamins, pigments, fatty acids, sterols, among others, which make their production/application by the food industry quite interesting. Chlorella genus is a eukaryotic microorganism, whereas Spirulina genus (cyanobacteria) is a prokaryotic microorganism. The aim of this review was to provide an overview on Chlorella and Spirulina microalgae, particularly as an alternative source of functional foods, nutraceuticals, and food supplements, in which the following compound groups were addressed: (I) Long-Chain Polyunsaturated Fatty Acids; (II) Phenolic Compounds; (III) Volatile Compounds; (IV) Sterols; (V) Proteins, Amino Acids, Peptides; (VI) Vitamins; (VII) Polysaccharides; (VIII) Pigments and (IX) Food. Chlorella and Spirulina microalgae and their derivatives are concluded not to be widely commercially exploited. However, they are remarkable sources of functional foods, nutraceuticals and food supplements.
Keywords: chlorella, spirulina, functional foods, nutraceuticals, food supplements
LCPUFAs, long-chain polyunsaturated fatty acids; ω6, omega-6 family; GLA, γ-linolenic acid; AA, arachidonic acid; ω3, omega-3 family; EPA, eicosapentaenoic acid; DHA, docosahexaenoic acid; AL, linoleic acid; AAL, alpha-linolenic acids; vitamin A, beta-carotene; vitamin K, phylloquinone; vitamin E, alpha-tocopherol; B1, thiamine; B2, riboflavin; B3, niacin; B5, pantothenic acid; B6, pyridoxine; B9, folic acid; B12, cobalamin; B7, biotin; FAO, food and agriculture organization; PCs, phenolic compounds; SW, subcritical water; SPE/SFE, solid-phase/supercritical-fluid; VOCs, volatile compounds; AA-CSIA, stable isotope analysis; FDA, american food and drug administration; EFSA, european food safety authority; GRAS, generally recognized as safe; PCA, p-coumaric acid; NC, narigenin chalcone; N, naringenin; LQ, liquiritigenin; D, daidzein; A, apigenin; G, genistein; L, luteolin; DK, dihydrokaempferol; DQ, dihydroquercetin; Q, quercetin
Chlorella and Spirulina are two of the most well-known microalgae genus. Chlorella is unicellular and Spirulina is a filamentous cyanobacterium, multicellular. Both are live in fresh water and have bioactive compounds like protein, vitamins, pigments, long chain polyunsaturated fatty acids, sterols and other compounds that make those microalgae very interesting from the health benefits point of view.
The name Chlorella was derived from the Greek chloros and from the Latin ella, which mean green and small, respectively. Chlorella microalgae have been present on earth since the pre-Cambrian period-2.5 billion years ago. Japan is currently the world leader in Chlorella microalgae consumption.1-3 The name Spirulina was based on its spiral shaped. However, linear shaped Arthospira microalgae have been identified.4 Spirulina microalgae are commonly called blue-green algae-cyanobacteria; Arthospira Platensis and Arthospira Maxima are cultivated worldwide.
Phytoplanktons such as microalgae have a nutritional value. In this sense, the genus Chlorella and Spirulina are two of the most prominent microalgae due to their high production of (I) Long-Chain Polyunsaturated Fatty Acids; (II) Phenolic Compounds; (III) Volatile Compounds; (IV) Sterols; (V) Proteins, Amino Acids, Peptides; (VI) Vitamins; (VII) Polysaccharides; (VIII) Pigments and (IX) Food.
Therefore, there are numerous exceptional compounds of interest that can be produced by microalgae, in particular Chlorella and Spirulina microalgae. These biocompounds can be further applied as functional, nutraceuticals and supplements feed.
Long-chain polyunsaturated fatty acids
Microalgae consist of approximately 30% lipids making them very interesting as food supplementary by humans. In this sense, microalgae are a source of long-chain polyunsaturated fatty acids (LCPUFAs) especially of the omega-6 family (ω6) such as γ-linolenic acid (GLA) and arachidonic acid (AA), and omega-3 family (ω3) eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Omega 3 and 6 families are essential fatty acids - they cannot be synthesized de novo by humans; and therefore, must ingest from food.19,20 Thus, as detailed below, omega 3 and 6 families are intimately related to health maintenance and disease prevention, in which microalgae are a promising source of ω3 and ω6.
GLA - ω6 fatty acid is an essential fatty acid presenting anti-inflammatory properties. Although the human organism is capable of producing very long chain fatty acids from linoleic (AL) and alpha-linolenic acids (AAL), its synthesis is affected by several factors, which makes the ingestion of these fatty acids essential for the maintenance of a healthy condition.19,20
Arachidonic acid is present in the membranes of body cells, and is the precursor of eicosanoid production through the metabolic pathway of the arachidonic acid cascade. It is one of the essential fatty acids, which need to be obtained via feeding by most mammals. Some of them have little, if any, ability to convert linoleic acid to arachidonic acid, and so it is essential that they get it in the diet.21
During breastfeeding, arachidonic acid can be found in breast milk and the placenta. This acid is responsible for the development of babies, some studies indicate that the low intake of arachidonic acid from the diet of premature babies, can cause health problems for these children. For those who practice physical exercise, arachidonic acid is also important, it can be one of the keys to muscle development. Studies have shown that this acid has increased performance levels.22 Lower levels of AA can contribute to neurological diseases such as Alzheimer23 and Autism.24,25
Fatigue, poor memory, dry skin, heart conditions, suicidal behavior (depression) and schizophrenia can be related to deficiency of ω3.
EPA (eicosapentaenoic acid), (ω3 fatty acid) has an anti-inflammatory action in our body, since it acts as a precursor for prostaglandin-3, thromboxane-3, and leukotriene-5 group, substances that are part of our defense against inflammation by helping to neutralize the pro-inflammatory activity of other similar molecules.
One of the key benefits of EPA is to aid heart health and blood circulation, preventing clots from forming in the blood and reducing the risks of thrombosis and cerebrovascular accident (stroke). Individuals who have inflammatory diseases, such as lupus and rheumatoid arthritis, may benefit even more from the EPA use.26 DHA is essential for fetal development and helps to form the retina, in addition, DHA has antioxidant action and is the most beneficial fatty acid for brain health, since it favors cognition and connections between neurons, benefiting the memory, attention, reasoning, imagination, judgment and various other aspects related to our mind.
In fact, a study that provided 900 mg of algae DHA for six months for a group of people suggests that DHA supplementation at this level may support the memory of healthy adults aged 55 or older (based on a clinical study using 900 mg DHA per day for six months in healthy adults with a mild memory complaint). In addition, there are research that links DHA to increased production of anti-inflammatory and neuroprotective substances preventing the formation of deleterious substances in the brain, which would decrease the risks of having neurodegenerative diseases such as Alzheimer's and Parkinson's. The metabolic pathway of polyunsaturated fatty acids is shown in Figure 1. Fatty acids of families ω-6 and ω-3 can be produced by the body from linoleic and alpha-linolenic acids, by the action of enzymes elongase and desaturase. The elongase acts added two carbon atoms in the initial part of the chain, and the desaturases act by oxidizing two carbons in the chain, creating a double bond in the cis-configuration. Research has pointed out that ideal ratio between ω3/ω6. Western diets have an omega 6 to omega 3 ratio of about 10 to 20:1. Several recommendations have been established by authors and health agencies from different countries on the ideal ratio of omega 6 and omega 3. Countries such as Germany and Sweden have established the ratio 5:1, while in Japan this recommendation is more rigorous, being 2:1. The Food and Agriculture Organization (FAO) is less demanding and recommends the ratio of 5-10:1.27
Regarding recent reports on the production of LCPUFAs from microalgae, Ferreira et al.30 evaluated the fatty acid profile of Chlorella homosphaera, Chlorella sp. and Chlorella minutissima, when cultivated in a heterotrophic mode (BG 11 or basal media) with 0, 5 and 10 g of glucose/L. They found that the highest concentration of lipids in the dry biomass of (I) Chlorella homosphaera, (II) Chlorella sp. and (III) Chlorella minutissima strains were (I) 22.4% w/w (BG 11-10 g of glucose/L); (II) 21% w/w (Basal-5 g of glucose/L) and (III) 21.5% w/w (Basal-10 g of glucose/L), respectively. Whereas, the highest concentration of total PUFA was (I) 35.25% w/w (Basal-0 g of glucose/L), (II) 24.35% w/w (Basal-10 g of glucose/L) and (III) 25.65% w/w (Basal-10 g of glucose/L). Similarly, the highest production of ω3-ω6 were (I) 35.05% w/w (Basal-0 g of glucose/L), (II) 24.05% w/w (Basal-0 g of glucose/L) and (III) 24.95% w/w (Basal-10 g of glucose/L). The authors concluded that heterotrophic cultivation of Chlorella is an effective strategy for the production of PUFAs, particularly the essential fatty acids. These biomolecules can be applied in the food production. In this sense, some commercially available products (human nutrition) produced with Chlorella and Spirulina microalgae are shown in Table 1.
Microalgae Genus |
Main Producers |
Products |
Production (Ton/Year) |
Chlorella |
Taiwan Chlorella Manufacturing Co. (Taiwan) |
Tablets, powders, nectar, noodles |
2,000 |
Klotze (Germany) |
Powders |
||
Spirulina |
Hainan Simai Pharmacy Co. (China) |
Powders, extracts |
3,000 |
Earthrise Nutritionals (USA) |
Tablets, powders, extracts |
||
Cyanotech Corp. (USA) |
Tablets, powders, beverages, extracts |
||
Myanmar Spirulina Factory |
Tablets, chips, pasta and liquid extract |
Table 1 Chlorella and Spirulina commercialized for human nutrition31
Therefore, Chlorella and Spirulina are very interesting alternative sources of LCPUFAs, which can be applied to a variety of nutraceutical and pharmaceutical purposes.
Phenolic Compounds
Phenolic compounds (PCs) also known as polyphenolics are biocompounds chemically composed of one or more phenolic rings that can be halogenated. These halogenations are responsible for the different biological activities.7,8 PCs are considered one of the most important classes of natural antioxidants. They protect both the cells and other natural body chemicals from the damage caused by free radicals. These are reactive atoms that contribute to tissue damage in the body. Free radicals, for example, oxidize low-density lipoprotein - LDL cholesterol, which can lead to its adhesion to arteries and lead to heart disease.32,33
PCs are indicators of stress microalgae metabolism. PCs are secondary metabolites that are not directly involved in biological processes as photosynthesis, cell division and reproduction.34 PCs are also related to the chemical protective mechanisms of microalgae as against biotic factors such as, settlement of bacteria, ultraviolet radiation and metal contamination.35-38
Considering the importance of PCs to be used as supplementary food evaluated the subcritical water (SW) technology to optimize the extraction of phenolic compounds from Chlorella sp. microalgae to be further applied as an antioxidant was evaluated.39 It was found compounds as caffeic acid, ferulic acid and p-coumaric acid. It is worth noting that the extraction is commonly carried out by organic solvents, a non-environment-friendly compound, that is, SW is an interesting alternative. The production of PCs from Spirulina maxima also was studied. The authors have found mainly pinostrobin, gallic acid, cinnamic acid, p-OH-benzoic acid, chlorogenic acid and vanillin acid, showing concentrations of 33.61, 19.82, 15.77, 14.18, 13.70 and 7.12% (based in the relative area), respectively.40
A new extraction technique based on a combination of solid-phase/supercritical-fluid (SPE/SFE) to obtain phenolic compounds from Spirulina platensis was studied.41 They have found 3,4-dihydroxybenzaldehyde (853.85 ng/g of dried cell) and p-hydroxybenzoic acid (604.94 ng/g of dried cell) as main compounds; and protocatechuic acid (137.98 ng/g of dried cell), vanillic (254.29 ng/g of dried cell), syringic (3.45 ng/g of dried cell), caffeic acid (169.52 ng/g of dried cell), chlorogenic acid (72.11 ng/g of dried cell) and 4-hydroxybenzaldehyde (107.35 ng/g of dried cell) as minority compounds.
Some studies have demonstrated the ability of microalgae to produce polyphenols, for example Chlorella pyrenoidosa, produce p-coumaric acid (the precursor of the flavonoid synthesis)-Figure 2.42
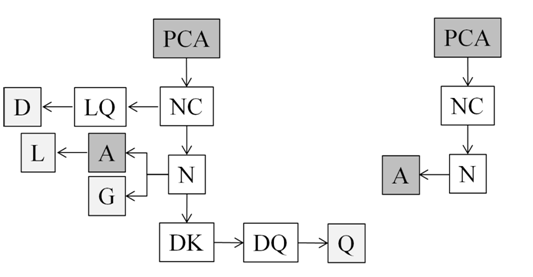
Figure 2 Pathway for flavonoid biosynthesis in Chlorella (A) and Spirulina (B) microalgae. (Enzyme abbreviations: PCA: P-Coumaric Acid; NC: Narigenin Chalcone; N: Naringenin; LQ: Liquiritigenin; D: Daidzein; A: Apigenin; G: Genistein; L: Luteolin; DK: Dihydrokaempferol; DQ: Dihydroquercetin; Q: Quercetin).42
The phenolic compounds biosynthesized by Chlorella and Spirulina microalgae were compared.42 They found that Chlorella microalgae have more content of phoroglucinol, which is used by the pharmaceutical industry - gastrointestinal disorders43 and also apigenin, which induces autophagy in leukemia cells (Table 2).44 On the other hand, Spirulina microalgae have more content of p-coumaric acid, which has antioxidant properties45 and ferulic acid that also has antioxidant properties and is commonly used as
Phenolic Compound |
Chlorella (ng/g) |
Spirulina (ng/g) |
Phloroglucinol |
74,000 |
51,000 |
p-Coumaric acid |
540 |
920 |
Ferulic acid |
0.63 |
0.97 |
Apigenin |
9.9 |
6 |
Table 2 Content of phenolic compounds in Chlorella and Spirulina microalgae42
Thus, Chlorella and Spirulina microalgae are rich sources of phenolic compounds that have antioxidants properties, among others interesting nutritional and health properties.
Volatile compounds (VOCs)
In the last years the interest in microalgaevolatile compounds (VOCs) has increased, mainly due to the diverse structural features and interesting biological activities of VOCs. However, VOCs can cause musty, fishy, and mud-like odor and produce harmful toxins, which is particularly important in water quality.47-53
Other interests concerning to VOCs have been drawing attention due to their biological activities54,55 which have been identified as carbonyls, alkenes, saturated and unsaturated aliphatic alcohols, aldehydes, ketones, esters, thioesters, sulfides terpenes, fatty acids, isoprenylated and brominated hydroquinones; and phycotene. VOCs present antibacterial, antifungal, antiviral and anticancer activities.48,56,57
The steam-volatile metabolites of Chlorella vulgaris, which was grown in fresh water culture, have been studied.58 It was found that more than 105 VOCs were produced, even so only 30 compounds were identified, for example hydrocarbons, acids, alcohols, esters, aldehydes and ketones-33.76, 23.93, 15.62, 8.02, 3.24 and 2.71% of the total VOCs, respectively. More details are shown in Table 3. It was also studied the phytotoxic effect of the VOCs. The authors found an acid phytotoxic effect significantly higher in comparison to the other fractions, due to mainly linoleic acid.
Class |
Compounds |
Composition (%) |
Hydrocarbons |
α-pinene |
2.11 |
β-pinene |
7.63 |
|
dodecane |
1.76 |
|
hexadecene |
15.3 |
|
heptadecane |
0.74 |
|
heptadecene |
2.11 |
|
octadecane |
3.21 |
|
tetracosane |
0.35 |
|
heptadecene |
0.55 |
|
Acids |
odecanoic |
0.2 |
tetradecanoic |
0.9 |
|
hexadecanoic |
8.73 |
|
octadecanoic |
2.2 |
|
octadec-9-enoic |
0.73 |
|
octadec-9,12-dienoic |
10.2 |
|
octadec-9,12,15-trenoic |
0.97 |
|
Alcohols |
hexadecanol |
10.8 |
octadecanol |
2.3 |
|
nonadecanol |
2.01 |
|
phytol |
0.51 |
|
Esters |
methyl-hexadecanoate |
2.2 |
methyl-octadecanoate |
3.52 |
|
methyl-octadec-9-enoate |
0.98 |
|
methyl-octadec-9,12-dienoate |
1.32 |
|
Aldehydes |
hexanal |
0.14 |
nonadecanal |
0.9 |
|
hexadecanal |
2.2 |
|
Ketones |
decanone |
0.7 |
hexadecanone |
1.7 |
|
α-ionone |
0.13 |
|
β-ionone |
0.18 |
|
U* |
12.72 |
Table 3 Composition of VOCs found in Chlorella vulgaris58
Recently, was determined the chemical profile of VOCs (Spirulina strains) and then, applied those VOCs in food products.59 As result, they obtained mainly (93.19%) hydrocarbons (medium length alkanes and alkenes) and in small quantities, odorous compounds, such as 2-methylisoborneol, 2-pentylfuran, 𝛽-cyclocitral, and 𝛽-ionone, as can be shown in Table 4.
Class |
Compounds |
Composition (%) |
Hydrocarbons |
2-pentylfuran |
3.03 |
tetradecane |
0.27 |
|
pentadecane |
6.48 |
|
hexadecane |
5.12 |
|
6,9-heptadecadiene |
0.62 |
|
heptadecene |
77.67 |
|
Alcohols |
2,6-dimetrylcyclohexanol |
2.41 |
2-ethyl-1-hexanol |
0.38 |
|
2-methylisoborneol |
0.98 |
|
Ketones |
1,1,3-trimetryl-2-cyclohexanone |
0.38 |
β-ionone |
1.59 |
|
Esters |
diisobutyric acid 1-tert-buty-l-2-methyl-1,3-propanediyl ester |
0.45 |
*A |
β-cyclocitral |
0.65 |
Table 4 Composition of VOCs produced by Spirulina platensis59
One of the most important sensory properties of food is the flavour. Chlorella and Spirulina microalgae have shown great potential to be applied in food due to their rich composition of VOCs, which present biological activities (VOCs). In addition, microalgae VOCs, usually are not related to musty, fishy, and mud-like odor.
Sterols
Sterols are lipids containing a ring formation in their chemical structures. Sterols play a fundamental role in the membrane integrity of microalgae.60 Over the last years, there is an increasing interest in the microalgae sterols, particularly due to their advantages of production and; nutritional and pharmaceutical importance.10
Sterols composition of red and brown microalgae are quite predictable however, the sterol composition of the green algae is unpredictable.61 Green microalgae such as Chlorella microalgae are shown to biosynthesized a variety of sterols.62
The sterols composition of Chlorella pringsheimii and Chlorella fusca were investigated.63 They found brassicasterol, ergostenol, poriferasterol and clionasterol in Chlorella pringsheimii, and ergosternol and chondrillasterol in Chlorella fusca.
Among sterols, phytosterol, which is structurally similar to cholesterol, is one of the most promising sterols. It can be used in healthy diets, in particular those that aim to reduce coronary heart disease.64 According to the International Union of Pure and Applied Chemistry (IUPAC) the chemical structure of phytosterol can be illustrated as shown in Figure 3.
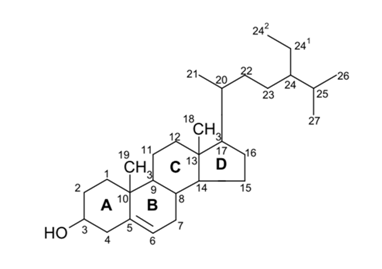
Figure 3 Chemical structure of phytosterol (campesterol is obtained removing 242. Removing 241 and 242 is yield cholesterol. Double bound between C22 and C23 it is obtained stigmasterol. Hydrogenating C5 and C6 stigmastanol are produced. Hydrogenating C5 and C6 and removing 242, campestanol is obtained. Taking away 242, double bound between C22 and C23 and inverting the stereochemistry at C24 it can obtain brassilcasterol).10
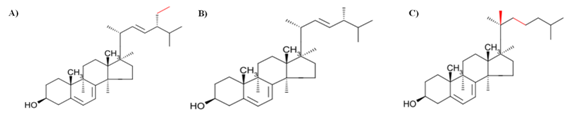
Figure 4 Chemical structure of the main phytosterols from microalgae: (A) 7-dehydroporiferasterol, (B) Ergosterol and (C) 7-oxocholesterol.10
The bioactivity functions of microalgae-derived phytosterol and their potential application in functional food and pharmaceutical industry, were described, such as immunomodulatory, anti-inflammatory, anti-hypercholesterolemic, antioxidant, anticancer and antidiabetic.10
Ergosterol and 7-dehydroporiferasterol are the major's sterols found in microalgae, both represent 45% of the total sterols.65 In this sense, some sterols from Chlorella vulgaris such as ergosterol, 7-dehydroporiferasterol peroxide, 7-dehydroporiferasterol, ergosterol peroxide and 7-oxocholesterol were identified, in which all of them showed effective anti-inflammatory and anticancer activities.66
Cholesterol and sitosterol were found in Spirulina maxima.67 The authors studied the correlation between sterols and antimicrobial activity. Similarly, 3 sterols produced by Spirulina platensis were identified: campesterol, stigmasterol and β-sitosterol; 8.7, 25.4 and 29.4% respectively.68
Concluding, the interest in microalgae sterols is increasing due to their higher concentration, when compared to plant sterols. Chlorella and Spirulina microalgae are sterols producers. Additionally, phytosterol (biosynthesized by microalgae), which cannot be synthesized by humans and have been made part of the human diet, could easily to be applied in nutraceutical and pharmaceutical industry.
Proteins, amino Acids and peptides
Microalgae proteins have been receiving attention by the food industry, mainly due to their great potential as an essential amino acid source.3,69
Microalgae such as Chlorella sp. and Spirulina sp. contain high protein contents-more than 50% dry weight-.68,59 In this sense, microalgae have higher nutritional quality when compared to conventional plants, especially soy. Microalgae also have other nutritive compounds such as carbohydrates, lipids, vitamins, polysaccharides, pigments, minerals and thus they are often used as functional foods, nutraceuticals, and food supplements.3,70,71
Spirulinasp. microalgae are composed of phycobiliproteins, which is a group of proteins related to the photosynthesis system (phycobiliproteins show pigment properties). Phycobiliproteins are well-known due to their hepatoprotective, anti-inflammatory, immunomodulatory, anticancer and antioxidant properties.13,34,72 Moreover, phycobiliproteins can also be applied as labels for antibodies, receptors, and other biological molecules that classify cells activated by fluorescence and are used in immunoblotting experiments and microscopy or fluorescence diagnosis.73
Chlorella sp.microalgaeare safe sources of proteins - dietary supplementation. Microalgae proteins have shown significant results in reducing blood pressure, lowering cholesterol and glycemia levels, accelerating wound healing and improving immune functions.74,75
The large-scale microalgae cultivation started in the 1960s in Japan, in which Chlorella microalgae were used as a food additive. In the 1970s and 1980s, the industrial production of microalgae expanded to USA, China, Taiwan, Australia, India, Israel and Germany. In the last years, the production of Spirulina and Chlorella microalgae has increased, and currently it is practiced by most countries. The annual production is about 7,500 tons of dry biomass (5,000 and 2,500 tons of Spirulina and Chlorella microalgae, respectively).76
Regarding the application of microalgae as a source of essential amino acids, the following essential amino acids were identified: lysine, leucine, isoleucine, tyrosine, tryptophan and valine, in which essential amino acids comprised approximately 35% of total amino acids.73 These amino acids contribute to the high nutritional quality of microalgae, making them highly suitable for use as supplements or as nutraceuticals.73,77,78 A new application of microalgae amino acids was developed.79 This application allows investigators to link consumers to food sources by tracing essential amino acids from producers to consumers. They used stable isotope analysis (AA-CSIA), to evaluate nutrient sources, especially essential amino acids, from Chlorella and Spirulina microalgae, as shown in Table 5.
Microalgae |
Isoleucine |
Leucine |
Phenylalanine |
Valine |
Chlorella |
26.74 |
31.05 |
26.35 |
28.07 |
Spirulina |
31.29 |
34.42 |
35.17 |
35.62 |
Table 5 Essential amino acids found in Chlorella and Spirulina microalgae79
Another interesting approach in microalgae proteins is related to bioactive peptides from microalgae, which can exert hormonal effects in the physiological stages of the human body.80 Peptides extracted from microalgae have antioxidant, anticoagulant, antihypertensive, immunomodulatory, antimicrobial and cholesterol lowering functions.81 The purified peptides from Chlorella vulgaris species was described as a great potential in the protection of DNA against oxidative damage of cells.82 The peptides extracted from Chlorella vulgaris species can be used in the prevention of diseases such as atherosclerosis, cancer and coronary diseases.
In this sense, proteins, amino acids and peptides from microalgae seems to be a very interesting alternative, in comparison to the traditional sources of proteins, amino acids and peptides, which can be applied to the human dietary requirements.
Vitamins
Chlorella and Spirulina microalgae are vitamin producers that are used in animal and human metabolisms,83 in which the vitamins that are commonly produced by microalgae are: vitamin A (in the form of beta-carotene), vitamin C, vitamin E and vitamin B such as thiamine (B1), riboflavin (B2), niacin (B3), pantothenic acid (B5), pyridoxine (B6), folic acid (B9) and cobalamin (B12).12 These vitamins are used to nourish the body, detoxify and normalize intestinal function, as well as stimulate the immune system and regenerate cells.12
Table 6 summarizes the vitamins content in Spirulina and Chlorella microalgae. When compared to Chlorella microalgae, Spirulina microalgae are richer sources of vitamin E, vitamin B1 and vitamin B7. Vitamin E protects the membrane lipids from oxidative damage and also prevents diseases such as coronary, atherosclerosis and multiple sclerosis, whereas thiamin (vitamin B1) presents anti-inflammatory properties while biotin (vitamin B7) presents maintaining hair, nails and skin healthy properties.84
Vitamins |
Chlorella |
Spirulina |
Vitamin A |
30.77 mg |
0.34 mg |
Vitamin C |
10.4 mg |
10.1 mg |
Thiamin (vitamin B1) |
1.7 mg |
2.4 mg |
Riboflavin (vitamin B2) |
4.3 mg |
3.7 mg |
Niacin (vitamin B3) |
23.8 mg |
12.8 mg |
Pantothenic acid (vitamin B5) |
1.1 mg |
- |
Pyridoxine (vitamin B6) |
1.4 mg |
0.4 mg |
Folate (vitamin B9) |
94 µg |
94 µg |
Cobalamin (vitamin B12) |
0.1 ug |
0 ug |
Vitamin E (alpha-tocopherol) |
1.5 mg |
5.0 mg |
Vitamin K (phylloquinone) |
- |
25.5 µg |
Table 6 Vitamin content in Chlorella and Spirulina microalgae per 100g of dry cells
Chlorella microalgae biosynthesis vitamin A. In fact, vitamin A is the most abundant vitamin produced by microalgae. Vitamin A is involved in immune function, vision, reproduction and cellular communication.84,85 Niacin (vitamin B3) is also abundantly biosynthesized by microalgae. Vitamin B3 is important for the metabolism of fats, cholesterol synthesis, DNA synthesis, regulation of glucose (blood sugar), reduce cholesterol and cardiovascular disease.85,86
Vitamin B12 (cyanocobalamin) is present in microalgae at low concentration. Vitamin B12 from microalgae in Chlorella sp. has better bioavailability than Spirulina microalgae.87 Vitamin B12 deficiency can cause neurological and psychiatric problems, due to its importance for neurological function and also for a proper red blood cell formation and DNA synthesis.
Chlorella and Spirulina microalgae have an interesting concentration of folic acid (Vitamin B9), which is necessary for the formation of cells and maintenance of metabolism, preservation of skin and mucous membranes and for the normal development of bones and teeth.69,88
Ingestion of small amounts of microalgae (biomass) may aid to reach all vitamin requirements (animal feed and human nutrition). Microalgae vitamins can enhance the nutritional value of algae cells - applied as a nutritional supplement. Nevertheless, it is important to note that the vitamin contents (microalgae) significantly change with environmental factors (cultivation), harvesting strategy and the method of drying the cells.12,88
Polysaccharides
Polysaccharides are polymeric carbohydrate structures that are widely used by industries, in particular food industry. Regarding the search for new natural antioxidants, the use of microorganisms such as Chlorella and Spirulina microalgae has been widely studied due to their high concentration of polysaccharides.13,14,89,90
In this sense, the polysaccharidesfrom Chlorella and Spirulina microalgae improve the enzymatic activity of the cell nucleus and synthesis of DNA repair, besides being a beneficial species to the immune system.14,32
Moreover, polysaccharides from Spirulina platensis and Chlorella pyrenoidosa microalgae have significant antioxidant properties, tumor activities, and immunomodulatory properties, as well as their great potential for the removal of superoxide and hydroxyl peroxide radicals.89,91
Two high molecular weight polysaccharides that were named immulina and immurella were identified.92 The immulina and immurella polysaccharides were isolated from Spirulina platensis and Chlorella pyrenoidosa, respectively. These polysaccharides presented higher activity against cancer, when compared to the fungal polysaccharides such as schizoplyllan, lentinan and krestin. The composition of immulina and immurella is shown in Table 7.
Immulina Polysaccharide (% mole) |
Immurella Polysaccharide (% mole) |
||
Rhamnose |
35.4 |
Arabinose |
31.6 |
Glucuronic acid |
9.7 |
Galactose |
26.8 |
Fructose |
7.7 |
Rhamnose |
12.4 |
Galactose |
7.1 |
Glucose |
5.4 |
2-methyl-Rhamnose |
5.9 |
3-methul-Arabinose |
3.0 |
Xylose |
5.5 |
3-methyl-Mannose |
2.5 |
3-methyl-Rhamnose |
4.2 |
Xylose |
2.4 |
2-methyl-Xylose |
4.2 |
4-methyl-Arabinose |
2.4 |
4-methyl-Rhamnose |
3.9 |
Mannose |
2.3 |
Glucose |
3.6 |
Ribose |
1.9 |
Mannose |
2.4 |
2,4-dimethyl-Arabinose |
1.3 |
Galacturonic acid |
2.0 |
3-methyl-Galactose |
1.2 |
3-methyl-Galactose |
2.0 |
3-methyl-Xylose |
0.9 |
Arabinose |
1.8 |
3-methyl-Rhamnose |
0.9 |
Amino sugar |
1.5 |
3,5-dimethyl-Hexose |
0.7 |
2-3-dimethyl-Fucose |
1.2 |
6-methyl-Galactose |
0.5 |
N-acetyl-glucosamine |
0.9 |
Glycerol |
0.5 |
2-methyl-Glucose |
0.5 |
2-keto-3-deoxy-Octulonosic acid |
0.4 |
Glycerol |
0.4 |
2,3,6-trimethyl-Mannose |
0.4 |
|
|
3,6-dimethyl-Mannose |
0.4 |
|
|
2,3-dimethyl-Mannose |
0.4 |
|
|
2-methyl-Galactose |
0.4 |
|
|
N-acetyl-Galactosamine |
0.3 |
|
|
N-acetyl-Glucosamine |
0.3 |
|
|
Amino sugar |
0.3 |
Table 7 Composition of polysaccharides from Spirulina platensis (immulina) and Chlorella pyrenoidosa (immurella)92
Rhamnose was found as the main compound ≈52.3% of total polysaccharides produced by Spirulina microalgae.93 Similarly, the extracts of Spirulina microalga polysaccharides were characterized, in which rhamnose represented ≈49.7% of total polysaccharides.94
Regarding Chlorella polysaccharides, two polysaccharides (69,658 Da and 109,406 Da) that were produced by Chlorella pyrenoidosa strains were identified. Both polysaccharides were composed mainly of rhamnose and mannose; 37.8 and 15.2%, respectively.91
Therefore, recently, microalgae polysaccharides have been used as alternative source of polysaccharides by the food industry, nutraceuticals, cosmetics and mainly pharmaceutical industry, since microalgae polysaccharides have antiviral activity against the virus Herpes simplex virus (type 1 and 2).14,91,95,96
Pigments, carotenoids and phycobiliproteins
Currently, rapid advances in biotechnology processes have been taking place worldwide. Regarding the production of specific biocompounds such as pigments, microbial processes, in particular those obtained from microalgae, have advantages over higher plants extraction (e.g. Marigold flowers), for instance easier cultivation, higher concentration, year-round production, potential to use agro-industrial wastewater as nutrient source, specific pigments as astaxanthin are rarely synthesized by higher plants (vascular plants). In addition, microalgae do not require arable land.15,16
Pigments are fundamental for photosynthetic algae metabolism. In other metabolisms such as human metabolism, pigments can act as antioxidant, anti-carcinogen, anti-inflammatory, anti-obesity, anti-angiogenic and neuroprotective.
The major classes of microalgae pigments are carotenoids and phycobiliproteins, in which carotenoids are sub-classified in two groups: carotenes and xanthophylls (oxygenated derivatives of carotenes). In addition, carotenoids are mainly used by the food industry as dietary supplements, fortified foods and food dyes and animal feed. Whereas phycobiliproteins are a group of colored proteins with linear tetrapyrrole prosthetic groups (bilins). Usually, phycobiliproteins are sub-classified into three groups: phycoerythrin, phycocyanin and allophycocyanin. The main potential applications of phycobiliproteins are: antioxidant, anti-inflammatory, anti-tumor, immunomodulating, radical scavenging, antiviral and antifungal.15,16,97,98
Carotenoids are hydrophobic compounds which have color orange, red and yellow. Usually, they share C40 backbone structure of isoprene units, in which more than 600 different carotenoids have been derived from C40 backbone structure. The bioproduction of carotenoids is biochemically related to photo-synthetic metabolism, for instance lutein can transfer absorbed energy to chlorophylls. However, pigments have others biochemistry function such as astaxanthin and canthaxanthin, which play a role in cell protective mechanisms. Currently, the major carotenoids produced at industrial scale are β-carotene, astaxanthin, lutein, lycopene, and canthaxanthin. Nevertheless, other pigments such as fucoxanthin (microalgae) have good potential to be produced at industrial scale in the short-term future (Figure 5).16,99
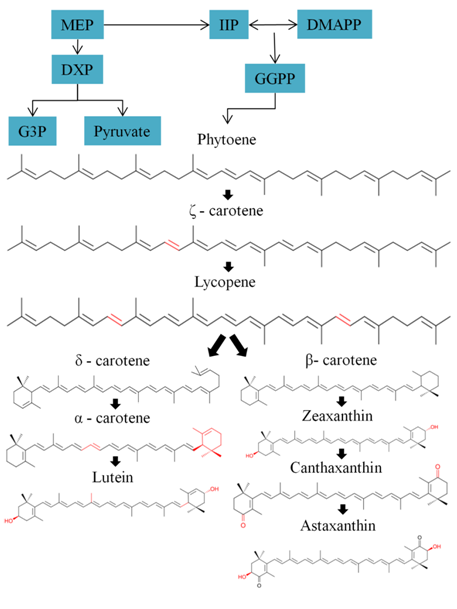
Figure 5 Primary steps of the biosynthetic pathway of carotenoids (non-mevalonate pathway) in most green microalgae species-G3P glyceraldehyde 3-phosphate; DXP 1-deoxy-D-xylulose-5-phosphate; MEP 2C-methyl-D-erythritol-4-phosphate; IPP Isopentenyl pyrophosphate; DMAPP dimethylallyl pyrophosphate; GGPP geranylgeranyl diphosphate.16
On the other hand, phycobiliproteins are watersoluble pigment proteins (Figure 6). Phycobiliproteins are classified based on their spectral properties. Phycoerythrin (λA max = 540570 nm; λF max = 575590 nm), phycocyanin (λA max= 610620 nm; λF max: 645653 nm) and allophycocyanin (λA max = 650655 nm; λF max = 657660 nm).100
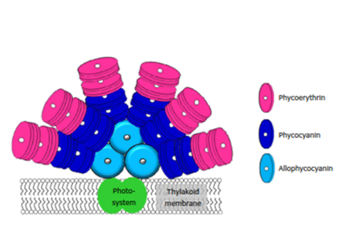
Figure 6 Schematic diagram of phycobilisome situated on the thylakoid membrane.100
Phycobiliproteins can be used in the production of fermented milk products, ice creams, soft drinks, sweet cake decoration, milk shakes, etc.100
The optimum conditions of phycobiliproteins production are related to environmental stress conditions, in particular light. Light, carbon and nitrogen sources stimulate the synthesis of phycobiliproteins. Compared to carotenoids, the biosynthesis of phycobiliproteins is more complex due to transcription, translational and posttranslational pathways and synthesis of amino acids, proteins and phycobilins.97
The concentration of lutein produced by Chlorella salina,in which≈ 5.06 mg of lutein per gram of dry alga was found.98 Regarding recovery and purity of microalgae pigments, Li et al.101 reported that the final purity of lutein by Chlorella vulgaris was94% and the yield was 88%, respectively.
The production of C-Phycocyanin by Spirulina sp. was described.18 C-Phycocyanin is sensitive pigment (light, pH, temperature and oxygen). Thus, strategies are needed to make the industrial applications of C-Phycocyanin feasible. The authors concluded that high concentration of sugars enhanced the stability of C-Phycocyanin, which leads to applications as confectionery and pastry.
Despite of (I) great market (market value of lutein is expected to reach US $309 million by 2018, market value of β-carotene is expected to reach US $334 million by 2018), (II) high added-value (average price of 5% astaxanthin is about US $1,900/kg) and (III) many studies on the production of pigments by microalgae, many challenges still remain, in particular harvesting and extraction processes.16,99
Microalgae as food
Microalgae in the human diet: The use of microalgae as food products has been increasing due to concerns regarding health and safety issues, for instance replacing synthetic dyes (synthetic β-carotene has been related to lung cancer and cardiovascular diseases). In this sense, according to American Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) food products based on Spirulina sp. are classified as GRAS (Generally Recognized As Safe).15-18 In addition, clinical studies indicated that the Spirulina consumption could lead to the reduction of cholesterol, protection against some types of cancers, enhance immune response, increase of intestinal lactobacilli (probiotics), protection against radiation (sunscreen lotion) and alternative treatment for obesity.102,103
Spirulina sp. has been used for a long time by people as consumable food in Mexico (Aztecs and other Mesoamericans) and Chad (Africa).15,104 Currently, the market for microalgae pigments has increased, in particular for human consumption.16
Many food products can be produced by the use of microalgae or their biocompounds, for instance isotonic beverages, cereal bars, instant soups, pudding, cake powder mix and biscuits.15 In this sense, Spirulina platensis can be used as a source of protein for malnourished people, in which chocolate biscuits fortified with Spirulina platensis presented higher protein content and higher digestibility.103 Similarly, the protein content (bread) increased due to the addition of dried Spirulina platensis (bread composition).105. Moreover, it was observed that the concentrations of 11 amino acids, significantly, increased, in which 4 out of them, are essential amino acid acids (threonine, methionine, leucine and isoleucine).105
An interesting approach using microalgae is the different compositions of wheat flour, cassava flour and Spirulina platensis biomass in the development of doughnuts, in which the presence of microalgae biomass enhanced the nutritional quality of cassava doughnut in terms of the protein, mineral, fiber and lipid.17
22 different Indian recipes with dried Spirulina were studied and all recipes were approved in terms of sensory evaluation. They also affect positively hyperglycemia and hyperlipidemia.106
Elaborated fresh spaghetti enriched with Chlorella vulgaris and Spirulina maxima was compared to standard semolina spaghetti (chemical composition, optimal cooking time, cooking losses, swelling index and water absorption, etc.).9 The authors concluded that presence of microalgae enhanced the nutritional and sensorial quality of pasta. In addition, no changes in cooking and textural properties were observed.
Microalgae in animal nutrition: Microalgae can also be used for animal nutrition, in which ≈30% of algae produced worldwide is currently used in animal nutrition102 for instance, fed hens with Chlorella microalgae.107 The authors observed that the content of linolenic acid and docosahexaenoic acid increased in the egg yolk. Similar results were found by using Spirulina microalgae instead of Chlorella microalgae.108
In this sense, the effect of dietary supplementation (light lambs) with microalgae rich in docosahexaenoic acid was investigated.109 The intramuscular fat was the most affected parameters. The authors concluded that the microalgae enhanced the quality (nutritionally) of lamb muscle, in particular the relations total polyunsaturated fatty acids/total saturated fatty acids; ω3/ω6 fatty acids. Whereas the diet supplementation with Spirulina platensis in the rabbit with high blood serum cholesterol levels decreased the cholesterol levels and also increased high-density lipoprotein cholesterol.110 Similarly, adult rabbits that were fed with Spirulina platensis showed an increasing of the digestibility of crude proteins.111
A subtle improvement in the body weight growth of piglets that were fed with Spirulina platensis was observed.112
When fattening calves were fed with the suspension of Chlorella and Scenedesmus microalgae, it was found that microalgae enhanced the digestibility of crude fiber, which leads to reduced total feeding cost, compared with animals fed with sesame seed oil.101
It was already proved that the use of Chlorella and Spirulina microalgae can be applied as a food source, in which it is intrinsically associated to many positive effects on health. The consumption (human or animal diets) of Chlorella and Spirulina microalgae has been occurring for a long time, particularly in Mexico and Chad (Africa), however, it is restricted in some countries, for instance Brazil. In this sense, the food industry can use microalgae in formulations of doughnuts, spaghetti, biscuits, etc., which show a remarkable potential, especially in nutraceutical terms.
Microalgae in fish feed: Fishmeal is traditionally used in fish feed and in the recent years, due to the increasing in fish production, microalgae appear as an economic and environmental-friendly alternative and also because they contain almost all nutrients that is needed for fish.
The effect of dietary Chlorella on the growth performance and physiological parameter (blood parameters and digestive enzyme) of Gibel carp (Carassius auratus gibelio) was evaluated. The addition of 0.8-1.2% of Chlorella showed better growth, higher contents of lyzosyme (blood parameter) that affect protein/lipid metabolism and immunity of gibel carp and also higher amount of digestive protein (amylase, lipase and protease) in comparison to the control (without Chlorella supplementation). Furthermore, the cholesterol of fish fed with Chlorella was lower than that found in the control.113
The effects of Chlorella vulgaris (supplement food) on blood and immunological parameters of Caspian salmon exposed to Viral Nervous Necrosis virus and they observed that the presence of Chlorella in fish fed diet could act as a natural immunuestimulant.114
Another study showed the replacement of fish meal by Chlorella meal supplemented by dietary cellulase to crucian cap Carassius auratus and after evaluated the growth performance, digestive enzymatic activities, histology and myogenic genes’ expression they conclude that Chlorella meal could totally replace the fish meal.115
Spirulina can be also applied in fish feed providing an increased growth rate of kenyi cichlids on Spirulina-based diet in comparison to the control (without Spirulina). The Spirulina feeding frequency (once and three times a day) on growth performance and seed production on kenyi cichlids (Maylandia lombardoi) were evaluated. The growth and the seed production on kenvi cichlids fed with Spirulina three times a day were higher compared to fed once a day.116
In recent years, much interest has been focused on the potential of microalgae biotechnology, mainly due to the identification of several substances synthesized by these microorganisms. Microalgae are of great importance, both biologically and economically. Their economic importance is related to the wide range of microalgae applications all over the world, from the food industry to medicine, from immuno-stimulants to biofuels, from cosmetics to agriculture. The immense biodiversity of microalgae and consequent high variability in their biochemical composition, combined with the use of genetic improvement and the establishment of large-scale cultivation technology has allowed certain species (e.g Spirulina microalgae) to be commercially available. The cultivation of microalgae has been carried out aiming at the production of biomass for both food manufacturing and also for obtaining natural compounds with high added value. Among these natural compounds are polyunsaturated fatty acids, carotenoids, phycobilins, polysaccharides, vitamins, sterols and many natural bioactive compounds such as antioxidants, cholesterol reducers, which can be used especially for the production of functional foods.
The São Paulo Research Foundation-FAPESP (Project number 2013/50218-2) and BNDES-FUNTEC/VALE (Project number 2542).
The author declares no conflict of interest.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.