Journal of
eISSN: 2469 - 2786


Research Article Volume 12 Issue 3
1Department of Microbiology, Adekunle Ajasin University, Nigeria
2College of Natural & Applied Sciences, Wesley University, Nigeria
Correspondence: Dr. Oludare Temitope Osuntokun, Department of Microbiology, Adekunle Ajasin University, Akungba-Akoko, Ondo State, Nigeria
Received: November 20, 2024 | Published: December 16, 2024
Citation: Olorundare SD, Osuntokun OT, Olatujoye CF, et al. Partially/synergistic efficacy of Ficus Exasperata & Syzygium aromaticum extracts against selected dermatophytes and non-dermatophytes fungi isolates. J Bacteriol Mycol Open Access. 2024;12(3):109-117. DOI: 10.15406/jbmoa.2024.12.00383
Dermatophytes are fungal infections which affect the nails, hair, and skin. They are of three major genera, which are Trichophyton, Epidemophyton and Microsporium. Dermatophyte is a pandemic; as it occurs throughout the world. This study determined the individual and synergistic effects of Ficus exasparata and Syzygium aromaticum extracts on both dermatophytes and non-dermatophytes. Upon isolation and culturing following conventional microbiological procedures, ten (10) fungal organisms were identified namely; Mucor pusillus, Candida guilliermondii, Trichophyton rubrum, Epidermophyton floccosum, Trichophyton schoenleinii, Aspergillus fumigatus, Aspergillus flavus, Fusarium avenaceum, Absidia corymbifera and Trichophyton verrucosum. The antifungal susceptibility test of the extracts was carried out using the agar well diffusion method, while the Minimum Inhibitory Concentration (MIC) was evaluated using tube dilution method. The antifungal effects of Ficus exasparata extract was more effective agaisnt Trichophyton schoenleiniiat 50 mg/ml compared to the other fungi with a zone of inhibition of 25.0mm while the antifungal effect of Syzygium aromaticum extracts (ethanol, methanol and N-hexane) was more effective against Candida guilliermondii, Trichophyton rubrum and Trichophyton rubrum, with inhibition zones of 35.0 mm, 40.0 mm and 24.0 mm respectively. The combined aqueous extracts of F. exaperata and ethanol extract of S. aromaticum had a zone of inhibition of 25.0 mm against Aspergillus fumigatus. The synergized methanol extract of F. exaperata and S. aromaticum had a high zone of inhibition of 21.0 mm against both Aspergillus fumigatus and Candida guilliermondi, while the synergized N-hexane extract had the highest zone of inhibition of 25.0 mm against Candida guilliermondi. The synergized ethanol extract of F. exasperata and methanol extract of S.aromaticum had the highest zone of inhibition (20.0 mm) against Trichophyton verrucosum. The synergistic effect of F. exaseprata and S. aromaticum extract has proved to be more potent compared to their individual extracts on dermatophytic infections. Further investigation on the antimicrobial effectiveness of Ficus exasperata and Syzygium aromaticum against fungal infections is recommended.
Keywords: sandpaper, clove, synergistic, dermatophytes, extract
Fungi are unicellular heterotrophic spore-producing organisms that derive nutrient by decomposing organic matters or by forming symbiotic relationship with other organisms. Fungi can be beneficial and some pathogenic. Naturally, they are not pathogenic,but can be one due to certain reasons such as immune-compromised system, excessive use of antibiotics etc. Dermatophytic fungi are fungal species which causes cutaneous mycosis, which are not deadly. Medicinal plants are believed to be important sources of new chemical substances with therapeutic effects. Thus, this research work is carried out to assess the effect of Ficus exasperata, syzygiumaromaticum and their synergistic effect against both dermatophytic and non dermatophytic fungi.
Mycosis infection is a common and diverse group of diseases caused by various species of fungi. These infections can affect different parts of the body, including the skin, nails,respiratory tract, and internal organs.1 Fungal infections (Mycosis) are among the most difficult diseases to manage in humans. Reports indicate high rates of morbidity and mortality caused by fungal infections.2
Despite their irreversible impact on human health, fungal pathogens have been mostly neglected by both the public and public health officials.3 Dermatophytes have been routinely placed in the class Deuteromycetes or "Imperfect Fungi". The dermatophytes' sexual state is helpful for epidemiological research and for identifying particular species. Thus, dermatophyte species can be divided into three genera according to variations in conidial morphology: Microsporum, Epidermophyton, and Trichophyton.4 Dermatophytoses are characterized by superficial invasion by fungal hyphae in the skin, hair, and nails causing sub acute or chronic infections.5
Although dermatophyte infections are restricted to areas of the epidermis, they can be invasive and cause serious widespread infections in immunocompromised patients.6 Major risk factors for the development of invasive fungal infections include, among others, HIV treatment in AIDS patients, cytotoxic chemotherapy in cancer patients, immunosuppressive therapy where innate defenses have been breached and the presence of catheters and other indwelling devices.7,8 Currently, mucormycosis also known as black fungus diseases have been acquired as secondary infections in COVID-19 patients.9
Epidermophyton are Large, multicellular, club-shaped, thin-walled macroconidia that are grouped in bunches define this genus; microconidia are not generated. Features of the genus are derived from E. floccosum. There are two species of Epidermophytonbased on anamorph morphology, (E. floccosum and E. stockdaleae). E. floccosumis the only pathogenic ''anthropophilic'' species in this genus that is found globally responsible for the majority of tinea cruris infections.9
Microsporum are microconidia and macroconidia in this genus. Macroconidia are spindle-shaped, multiseptate, and have a thin or thick echinulate cell wall. They can also be abundant or rare. However, the echinulations on the macroconidial cell wall serve as this genus' primary characteristic that sets it apart. Pyriform microconidia measure roughly 2-3μm. Microsporum audouiniiis the type species. On the basis of anamorph morphology, there are approximately 18 species of Microsporum. In the Mediterranean region, tineacorporis and tineacapitis are most frequently caused by M. canis.10
Trichophyton are genus produces microconidia “2-3 μm pear-shaped” and macroconidia “cigar-shaped” with smooth walls. Based on anamorph shape, Trichophyton is divided into 25 species and T. tonsurans is the type species.10,11 In Central and North Europe, T. rubrum is the most prevalent dermatophyte within the past 20–30 years. According to reports, in the United States and Canada, the most commonly isolated organism in infecting children with tineacapitis is Trichophytontonsurans, while the isolated organism in tineapedis and tineacruris is T. mentagrophytesvar. interdigitale, a member of the T. mentagrophytes.10
As eukaryotic pathogens, fungi share many similarities with their host cells, which impairs the development of antifungal compounds.2 Cutaneous fungal parasites have survived several generations of therapeutic regimens, and the increasing invasive fungal infections along with the emerging resistance of pathogens and disadvantages with the existing antifungal drugs, demand the development of new antifungal drugs in clinical practice.12
Men are more likely than women to contract this fungal infection, which generally affects men between the ages of 20 and 40. This infection usually starting in the interdigital clefts then spreads to ankles, dorsum, soles, legs, and toenails, a condition known as tineaunguium. Patients with diabetes are thought to have a 50% increased risk of developing a fungal infection, such as tineapedis.13 Tineapedis is differentiated into interdigital type, squamous-hyperkeratotic type and vesiculous-dyshidrotic type. The most prevalent clinical manifestation is the interdigital, which mostly affects the gaps between the fourth and fifth toes and manifests as maceration, fissuring and peeling.
Squamous-hyperkeratotic type (hyperkeratosis and acanthosis) where the pinkish skin of the heels, sides, and soles of the foot is covered in tiny silvery scales (moccasin foot), and vesiculous-dyshidrotic type. The etiology of the complex infection, which is a combination dermatophyte and bacterial infection, is polymicrobial infection and is clinically more severe.14 The dermatophyte that cause tineapedis were identified as T. rubrum, T. mentagrophytes, M. Canis, E. Floccosum, T. verrucosum and T.violaceum with decreasing frequency.15
Clove (Syzygium aromaticum) is one of the most ancient and valuable spices of the orient. Portuguese first discovered in the sixteenth century the origin of clove plants is Molucca islands. But the use of clove in ayurvedic and homoeopathy medicine was well known before that time. The major part of the world’s consumption of the clove spice is in the home kitchens. Cloves have been used for their antiseptic and analgestic effects and studied for use in platelet aggregation inhibition, antithrombotic activity and chemo-protective and antipyretic effects. Like many culinary spices, cloves help relax the smooth muscle lining of the digestive tract. The oils of the cloves have been known to stimulant and it may increase blood circulation. The present paper reviews health benefits from the use of clove, covering its chemical constituents, phyto-pharmacology, nutritional and medicinal value16–18 Ficusexasperata, commonly called forest sand paper tree / plant, is one of the 800 species of terrestrial plants in the family Moraceae. F. exasperata inhabits the secondary rainforest of West Africa and extensively spread in all eco-regions of Nigeria. This plant is locally called Ewe Ipin (Yoruba), Opoto (Calabar), Anwulinwa (Igbo) and Ijikpi (Igala). F. exasperata has been ethno-botanically reported to have varied therapeutic uses such as treatment / management of hypertension, epilepsy, arthritis. The leaf is used to scratchskin parts affected by ringworm, while the grounded leaves applied topically are used to treat boils. The viscid non-milky sap of this plant is used to stop bleeding, treat sores eye trouble and stomach pain (Figures 1, 2).19
Study site
This project work was carried out in the Microbiology Laboratory of Adekunle Ajasin University, Akungba Akoko, Ondo State. Samples were collected from different salons in Akungba-Akoko with coordinate of 7.470°N 5.7379°E Latitude of 7.467 and Longitude 5.733.
Sample collection
Samples were collected from equipment used in the following salons within Akungba Akoko: Jeff-Time salon (JT), Abacha hair salon (AH) and New World Unisex salon (NW), and aslo from people who shew clinical symptoms of dermatophytic infections. After collection, the samples were tagged and taken to the laboratory for microbiological analysis.
Isolation of the test organisms
Pour plate
The plates were carefully labeled at the bottom and one-tenth of one mili-liter (0.1ml) of each selected dilution was poured into each plate asceptically. The agar was then poured into the plates and swelled gently to ensure uniform distribution. The media were allowed to set on a flat top work bench, after which the plates were incubated at room temperature for 3-7 days (Plates 1–8).
Swabbing method
The labelled swab-sticks used to collect samples from infected individual were brought to the laboratory for subsequent microbiological analysis. The agar was prepared and poured asceptically into the plates. Upon solidification of the agar, the swab-sticks were swabbed upon the plates gently. After swabbing, the plates were covered and incubated at room temperature for 3-7 days.
Sub-culturing
Upon getting distinct colonies from the plates, another media was prepared following the manufacturer’s instruction. After incubation, the media was poured into a sterile plate and allowed to cool. After the plate was set, a small portion of the colony was cut from the previous plate and placed aseptically on the new media.
Identification of various isolated samples
Fungal staining
A drop of Lactophenol cotton blue (stain) was placed on a clean, grease free microscope slide. A part of the colony was teased out using the innoculating needle and placed on the stain and teased out gently to avoid dislodging the internal features of the fungi. The cover slip was then placed gently on it.The slide was then viewed under the microscope, using objective lens of x40. The result of the microscopy was then compared with those in Compendium of Soil Fungi volume 2 (28).
Standardization of extracts
Half (0.5)ml of the extracts was measured and transferred into a flask containing fifty (50) ml of sterilized water. The flask was shaken to ensure uniform distribution of the extract. The flask was then labeled 100mg/ml. from the flask, ten (10) ml of the diluted extract was measured and filled into a bottle containing ten (10) of sterilized water, the bottle was labeled 50mg\ml. From the bottle labeled 50mg/ml, ten (10) ml of the diluted extract was measured and transferred into another bottle containing ten (10) of sterilized water, the bottle was labeled 25mg\ml. From the bottle labeled 25mg/ml, ten (10) ml of the diluted extract was measured and transferred into another bottle containing ten (10) of sterilized water, the bottle was labeled 12.5mg\ml.This process was repeated for all extract used.
Synergizing of extract
Ten (10) ml of Ficus exasperata extract was added to five (5) ml of Syzygium aromaticum extract and left for twenty-four hours.
Antimicrobial suceptibility test (Agar well diffusion method)
Innoculation of isolates into broth
The media was prepared following the manufacturer’s instruction, the media was then filtered to remove the solidifying agent (agar) from it and this resulted into broth. The broth was filled into eleven (11) test-tubes and sterilized using an Autoclave at 121°C for 15 minutes. After sterilization, the broth was allowed to cool to a temperature of 35°C, then the isolate were innoculated into it using the innoculating needle. The test-tube was then incubated at 27°C for 24 hours.
Antifungal suceptibility test
After the isolates wasaseptically introduced into the plate, two drops of the extracts were introduced into the bored hole well. Also a control (Clotrimazole) was added to the hole bored at the middle of the plate. The extracts were allowed to diffuse for about 30 minutes, then the plates were incubated for a period of 18-24 hours.
Reading of result
After the incubation period, any clear zone around the well was measured and recored. Those zones are known as zone of inhibition.
This studydetermined the individual and synergistic effects of Ficus exasperata and Syzygium aromaticum extract on both dermatophytes and non-dermatophytes. Swabbing was made on different fomites such as comb, brushes and clippers used in some selected salons.Skin swab was also conducted on infected individuals and taken to the Microbiology laboratory, Adekunle Ajasin University, Akungba-Akoko for investigation using standard microbiological procedures. Upon isolation and microscopic examination of all thirty one (31) isolates cultured, ten (10) distinct fungal species were identified, namely: Mucor pusillus, Candida guilliermondii, Trichophyton rubrum, Epidermophyton floccosum, Trichophyton schoenleinii, Aspergillus fumigatus, Aspergillus flavus, Fusarium avenaceum, Absidia corymbifera and Trichophyton verrucosum. Four (4) of these test fungi were dermatophytes namely: Trichophyton rubrum, Epidermophyton floccosum, Trichophyton schoenleiniiand Trichophyton verrucosum, while the remaining six (6): Mucor pusillus, Candida guilliermondii Aspergillus fumigatus, Aspergillus flavus, Fusarium avenaceum and Absidia corymbifera were non-dermatophytes. The antifungal susceptibility test was carried out using agar well diffusion method to compare the effects of the individual extracts of Ficus exasperata and Syzygium aromaticum and their synergistic effects against the identified organisms (Figure 3, Tables 1–4).
|
Isolate code |
Number |
Percentage (%) |
Probable organism |
|
ACC 1 |
3 |
9.677 |
Trichophyton verrucosum |
|
SS1 |
8 |
25.806 |
Aspergillus flavus |
|
ACC 2 |
1 |
3.226 |
Aspergillus fumigatus |
|
SS 2 |
1 |
3.226 |
Absidia corymbifera |
|
NW 1 |
1 |
3.226 |
Fusarium avenaceum |
|
SS 3 |
1 |
3.226 |
Trichophyton schoenleinii |
|
NW 2 |
2 |
6.452 |
Epidermophyton floccosum |
|
NW 3 |
3 |
9.677 |
Trichophyton rubrum |
|
NW 4 |
1 |
3.226 |
Candida guilliermondii |
|
SS 4 |
10 |
32.258 |
Mucor pusillus |
Table 1 Frequency of occurence of the organism from samples collected
|
Isolate code |
Color |
Reverse |
Margin |
Elevation |
Opacity |
Surface |
Shape |
|
ACC 1 |
White |
Creamy brown |
Filamentous |
Flat |
Translucent |
Cotton like |
Circular |
|
SS1 |
Lemonish green |
Light brown |
Filamentous |
Raised |
Opaque |
Cotton like |
Irregular |
|
ACC 2 |
Lemonish yellow |
Creany |
Filamentous |
Flat |
Opaque |
Rough |
Circular |
|
SS 2 |
White |
Creamy |
Even |
Flat |
Opaque |
Glistening |
Irregular |
|
NW 1 |
Whire |
Brown |
Filamentous |
Raised |
Opaque |
Rough |
Circular |
|
SS 3 |
White |
Creamy |
Even |
Raised |
Opaque |
Glistening |
Circular |
|
NW 2 |
White |
Yellow |
Filamentous |
Flat |
Opaque |
Smooth |
Circular |
|
NW 3 |
White |
Creamy |
Filamentous |
Flat |
Opaque |
Cotton like |
Irregular |
|
NW 4 |
Black |
Black |
Wavy |
Raised |
Opaque |
Rough |
Irregular |
|
SS 4 |
White |
Creamy |
Filamentous |
Flat |
Opaque |
Cotton like |
Fillamentous |
Table 2 Morphological characteristic of isolates
KEY: ACC: Abacha clipper; SS: Skin sample; NW: New world
|
Isolate Code |
Hyphae structure |
Sporangiophore or conidiophore |
Septa |
Type of asexual spores |
Type of sexual spores |
Distinct character |
|
ACC 1 |
Branched |
Absent |
Septate |
Microsonidia, macroconidia |
Ascospores |
Large, multiseptate macroconidia. Often found in association with cattles. |
|
SS1 |
Branched |
Present |
Septate |
Conidia |
Ascospore |
Vesicle with radiating sterigmata. Produces aflatoxin |
|
ACC 2 |
Branched |
Present |
Septate |
Conidia |
Ascospore |
Vesicle with radiating sterigmata, roughened conidia. Conidiophores with flash-shaped vesicles. |
|
SS 2 |
Branched |
Present |
Aseptate |
Sporangiospores |
Zygospores |
Non-septate hyphea, pyriform sporangia. Produces sporangiophores with sporangia at the tips |
|
NW 1 |
Branched |
Present |
Septate |
Macroconidia |
Ascospores |
Curved multiseptate macroconidia. Banana shaped macroconidia. Often found in soil and decaying matter. |
|
SS 3 |
Branched |
Absent |
Septate |
Microsonidia, macroconidia |
Ascospores |
Spiral\helical hyphea. Less common. |
|
NW 2 |
Branched |
Absent |
Septate |
Macroconidia |
Ascospore |
Large, multiseptate macroconidia |
|
NW 3 |
Branched |
Absent |
Septate |
Microsonidia, macroconidia |
Ascospore |
Spiral\helical hyphea. Red pigment on PDA |
|
NW 4 |
Pseudohyphae |
Absent |
Septate |
Blastospores |
Ascospores |
Pseudohyphae. Can be both yeast and filamentous. Opprtunistic pathogen |
|
SS 4 |
Branched |
Present |
Aseptate |
Sporangiospore |
Zygospores |
Non-septate hyphea, pyriform sporangia. Often found in decaying organic matter |
Table 3 Microscopic features of the organisms
|
Isolate Code |
Probable Organisms |
|
ACC 1 |
Trichophyton verrucosum |
|
SS1 |
Aspergillus flavus |
|
ACC 2 |
Aspergillus fumigatus |
|
SS 2 |
Absidia corymbifera |
|
NW 1 |
Fusarium avenaceum |
|
SS 3 |
Trichophyton schoenleinii |
|
NW 2 |
Epidermophyton floccosum |
|
NW 3 |
Trichophyton rubrum |
|
NW 4 |
Candida guilliermondii |
|
SS 4 |
Mucor pusillus |
Table 4 The probable names of the fungal isolates in the study
Figures 4, 5 and 6 reveal the antifungal effects of Ficus exasparata extracts against the fungal organisms with clotrimazole as control. Trichophyton schoenleinii was susceptible to all the extracts tested against it, with highest zone of inhibition of 38.00mm by the N-hexane extract at 25mg/ml and lowest zone of inhibition of 9.00mm by the ethanolic extractat 100mg/ml.
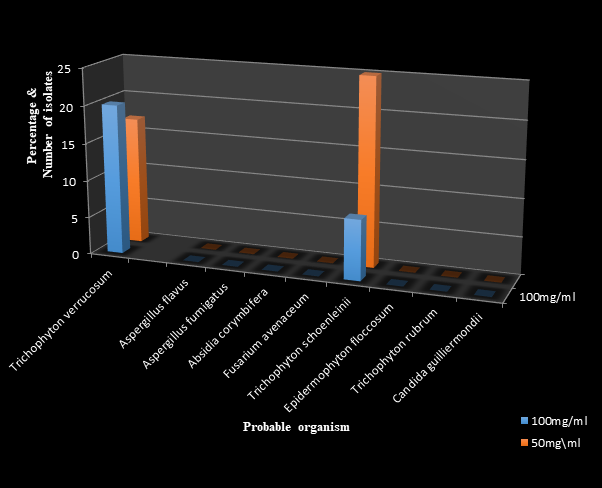
Figure 4 The antifungal effects of standardized Ficus exasperata ethanol extract measured in milimeters (mm).
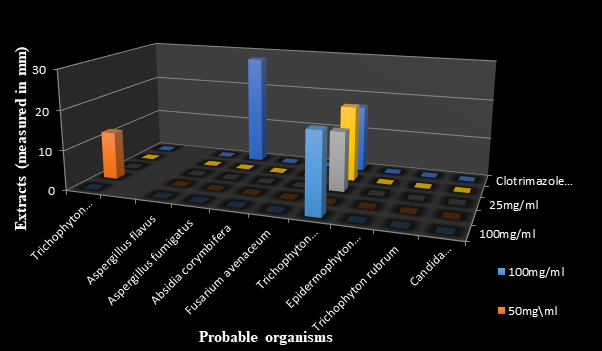
Figure 5 The antifungal effects of standardized Ficus exasperata methanolic extract measured in millimeters (mm).
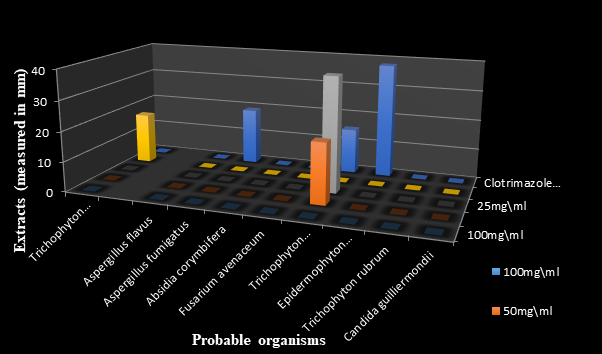
Figure 6 The antifungal effects of standardized Ficus exasperata N-hexane extract measured in millimeter (mm).
Figures 7, 8 and 9 show the antifungal effects of Syzygium aromaticum extracts against the fungal organisms with clotrimazole as control. The methanolic extract had the highest zone of inhibition of 30.00mm againstTrichophyton rubrum with its lowest zone of inhibition at 0.00mm with the ethanolic extract even at 100mg/ml.
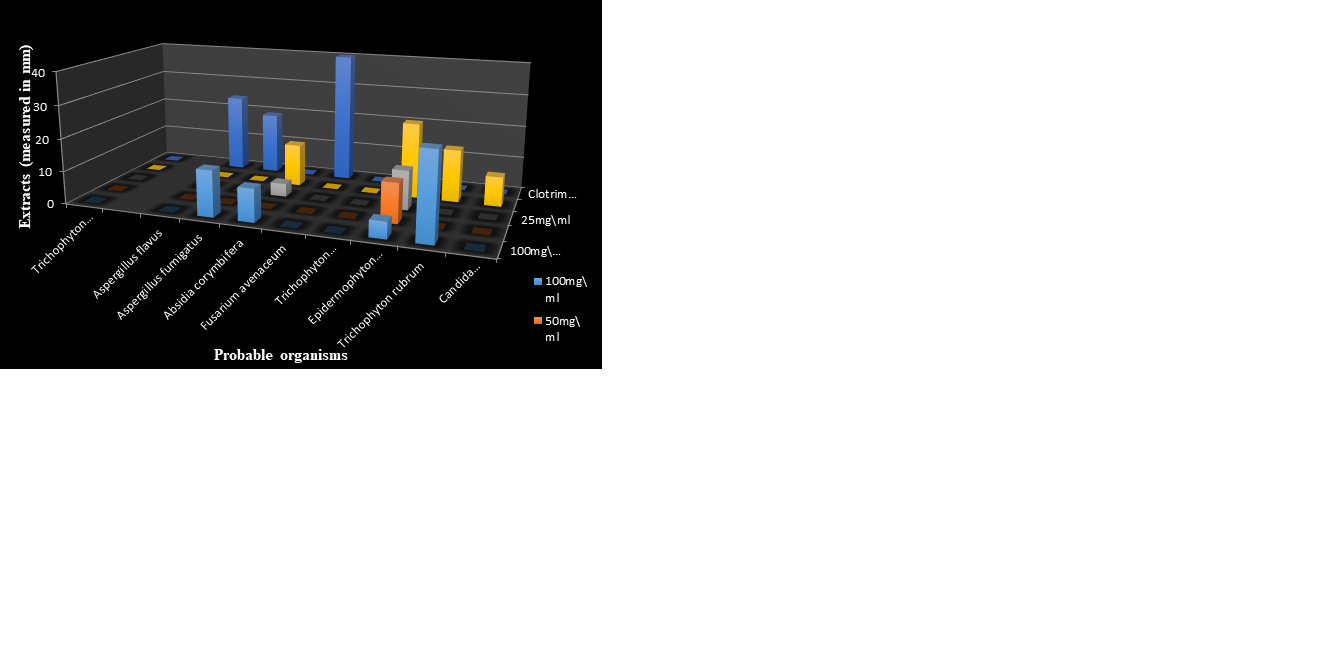
Figure 7 The antifungal effects of standardized Syzygium aromaticum N-hexane extract measured in milimeter (mm).

Figure 8 The antifungal effects of standardized Syzygium aromaticum ethanolic extract measured in milimeter (mm).
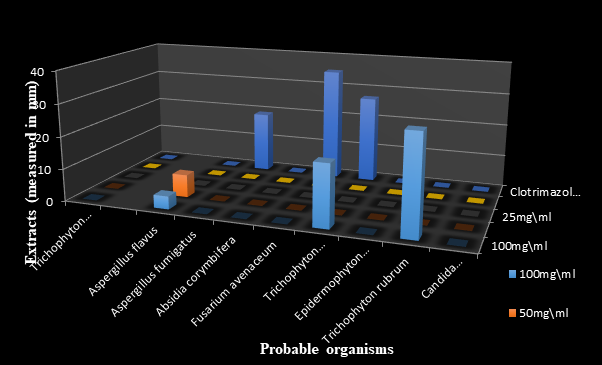
Figure 9 The antifungal effects of standardized Syzygium aromaticum methanolic extract measured in milimeter.
Figure 10 reveals the synergistic effects of Ficus exasperata and Syzygium aromaticum extracts at 100mg/ml. The synergized aqueous extract of F. exaperata and ethanolic extract of S.aromaticum had a zone of inhibition of 25.00mm againstAspergillus fumigatus. The synergized methanolic extract of F. exaperata and S.aromaticum had a high zone of inhibition of 21.00mmagainst both Aspergillus fumigatus and Candida guilliermondii, while the synergized N-hexane extract had the highest zone of inhibition of 25.00mm againstCandida guilliermondii. The synergized ethanolic extract of F. exasperata and methanolic extract of S.aromaticum had the highest zone of inhibition of 20.00mm againstTrichophyton verrucosum. Among all the isolates,Trichophyton verrucosum was susceptible to all the synergized extracts tested against it.
Different reports abound on the incidence of dermatophytic infection globally, with low mortality in several countries of the world including Nigeria and her neighoring countries.
Syzygium aromaticum (Cloves) have antimicrobial peoperties, and can even inhibit both dermatophyte and non-dematophyte moderately. Ficus Exasperata (Sand-paper) has some medicinal properties which include, but not limited to: antihypertension, antiulcer. Sandpaper also have antimicrobial potential and has always been used in combating dermatophytic infection in our local environment and it has been pretty effective. These antimicrobial properties are often due to the phytochemicals present in them. But inthis research work, the antifungal effect of Syzygium aromaticum is less effective compared to that of Ficus exasperata.
In this research work few dermatophytes were found even from salons around the test area which is Akungba-Akoko. Several non-dermatophyte were also found some of which are spoilage organism and secondary causative agent of dermatophytic infections. The presence of Aspergilus spp, Candida sp. among others is in line with the work of.20,21 The presence of these non-dermatophyte show a synergistic relationship between non-dermatophye and dermatophyte, which has been similarly reported.22
Dermatophyte and non-dermatophytes varies in term of mode of nutrition: dermatophyte obtain their nutrient by breaking down keratin, which is aprotein found in skin, hair and nails as reported by.19 While non-dermatophyte obtain their nutrient through: parasitism, commensalism etc. according to,23 dermatophyte are often resistance to topical antifungal and this is on the increase.
The morphological characteristic of the organism which includes the color (white, creamy, greenish-yellow etc), elevation (flat, raised, convex etc), surface (rough, smooth, glistening etc), margin(wavy, filamentous, even etc), shape (regular, circular, irregular punctiform), reverse color (brown, black, creamy etc) and opacity (transparent, opaque or translucent) are in line with the work of many researchers and some renowned Mycologist. These morphological characteristic can some times be used to classify some fungi species which may have similar morphology. Some of the organism have branched and unbranched hyphea, which helps them with there nutrient uptake, interaction with host cell if they are pathogenic, it also helps them adapt to specific environment. These features also plays a critical role in determining the organism pathogenecity, how they would react to drugs.
Also the presence, absence or posseseion of false crosswall contribute tohow the fungi reacts to drugs. Several journals on Antifungal Resistance and Fungal cell wallhas shown that aseptate hyphae have reduced drug penetration, they also have enhanced virulence factors and alsoincrease antifungal resistance. While septate hyphae have reduced virulence factors, repond to drugs easily. Organism like Candida guillermondii which have pseudoseptate hyphae are moderately virulence, have intermediate response to antifungal24 also show that some dermatophyte are resistance to Terbinafine especially those that caused Tinea capitis, Tinea cruris, Tinea corporis and Tinea pedis.
David,24 in his work reported that Aspergillus funmigatus which is the most common cause of invasive, chronic and allergic aspergillosis which collectively affect ten million people is resistance to the triazole group of oral antifungal drug. David, 2021 shows that Candida spp such as Candida guillermondii among others shows lees susceptibiity or resistance in rare cases to fluconazole. The presence of Aspergillus flavus among the isolates implies that infectious organism which can cause skin, nail and even respiratory infection. And this can be as a result of poor sanitation, hygine, contaminated equipment and use of clipper or comb from infected individual both client and staffs.25
The presence of A. flavus can cause allergic reaction which can trigger some reaction such as itching, redness etc. It can also, which is a rare but serious infection which can spread to the internal organs.25 A. flavus can also produce a mycotoxin called aflatoxin, which can damage the liver and some vital organs in the body.26
The presence of Mucor pusilius amongsamples gotten from salon maybe as a result of poor ventilation and poor sanitation of the equipment used. The organism can also be used in the production of biofuel , enzymes, antibiotics (pusilin) which is in line with the work of Ibrahim, et al24 and Al-Shammari.25 The presence of the rare Candida spp:Candida guillermondii among the isolate indicate the presence of opportunistic fungi which affect immunocompromised individuals and also a spoilage organism, which spoils dairy products, fruit juice and other food products. Candida guillermondii take part in the production of biofuel.
Trichophyton rubrum has morphological characteristic of white surface, fillamentous margin and a flat elevation. Trichophyton rubrum found among isolate from salon can pose a threat of infection to both client and staff of the salon as it can cause body ringworm and this can cause a damage to the reputation of the salon., this can be prevented by proper sterilization of clipper, comb, brush.25,26 Epidermophyton floccosum: a dermatophyte which causes whichTinea corporis was isolated from human skin, this can be as a result of poor hygine of the individual. Sharing of personal items such as towel, comb, hair brush can facilitate its spread.
The use of partially sterilized equipment in the salon where samples were collected contributed to the high occurrence of Mucor pusilius and Aspergillius flavus among the isolate and which can lead to the client being infectedand this can be controlled by individual having his or her personal beautifying gadget and the proper sterilization of commonly used equipment in salon.Poor personal hygine may be the cause of Trichophyton rubrum and Trichophyton verrocossum having a frequent occurrence among the dermatophytes, and these organism causes Tinea faciei, tinea barbae etc. These organism can also be acquired from sharing of object like comb, hair brush, towel etc. and thus personal hygine plays a vital role in curbing the spread of these organism.27
Personal hygine is one of the ways through which nsome dermatophytic infections can be avoided, also sharing of personal thing like clothes, underwear, comb can also prevent the spread of sermatophyte. Non-dermatophyte can also be prevented in the same way, as they can form relatonship with dermatophyte to cause infection. Alons should always sterilizile any equipment which they use, before and after use and if possible they should avoid client with obvious dermatophytic infection. By doing this th frequency of both dermatophye and non-dermatophyte would reduce. The extract of Ficus exasperata has more effect on the test organism with Trichophyton schoenlenill having the largest zone of inhibition at 25mg\ml of the N-hexane extract and its smallest zone of inhibition at 100mg\ml of the ethanolic extract. While the N-hexane extract of Syzygium aromaticum had effect on Trichophyton rubrum with the largest zone of inhibition at its 100mg\ml. this would imply that the 100mg\ml is effective only on Trichophyton rubrum, while the other organism are resistance to the extract The synergistic effect of both extract (Methanol extaract of both plant and Ethanol extract of Ficus exasperata and methanol extract of Syzygium aromaticum) has effect on almost all the organism: and this implies that the synergistic effect of F.exsperata and S. aromaticum extract is more effective compare to the individaual extract.28
This study has tested the individual effect of Ficus exasperata and Syzygium aromaticum extract and also their synergistic effect and the result shows that the synergistic effefct is more effective compare to the individual effect. The extract therefore can be exploited for the production of antifungal oilment or topical creams.
Based on the study, the synergistic effect of Ficus exasperata and Syzygium aromaticum possess richer antifungal properties. However, more scientific studies would be required to isolate the bioactive compound in these medicinal plants, and also to standardize them.
Self-funded research, authorship, and/or publication of this article: Financial assistance was not provided by any Research Foundation.
The laboratory staff of Adekunle Ajasin University, Department of Microbiology, Faculty of Science, Akungba Akoko, Ondo State, Nigeria.
The authors declare that there are no conflicts of interest.

©2024 Olorundare, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.