eISSN: 2379-6367


Opinion Volume 9 Issue 5
IFBV-BELHERB BP 98, Luxembourg
Correspondence: Pierre Lutgen, IFBV-BELHERB BP 98, Niederanven, Luxembourg
Received: September 15, 2021 | Published: October 8, 2021
Citation: Lutgen P. Prophylaxis of malaria by Artemisia infusions: The key role of lysine. Pharm Pharmacol Int J. 2021;9(5):210-211. DOI: 10.15406/ppij.2021.09.00346
There are many anecdotic reports or clinical trials describing the prophylactic effect of Artemisia plants against malaria and this has been documented in several peer reviewed papers. But the mechanism of this action has not been elucidated. The prophylactic effect is not due to artemisinin or its derivatives. We made a literature survey to find possible explanations.
Since several years we try to understand why Artemisia infusions show a very efficient prophylaxis against malaria. This has been noticed in clinical trials and anecdotic reports in several African countries. Patrick Ogwang in his seminal work finds that Artemisia annua infusion consumed once a week is effective in preventing multiple episodes of malaria attacks and its mode of action may involve stimulation of white blood cells production especially monocytes.1 We have proposed several hypotheses, but were so far unable to verify them in vivo.2,3 Another hypothesis was based on the oxidative weakening of sporozoites by NO generated by arginine. Scientific papers on this topic are scarce.4,5 Another explanation for the prophylactic effect is resting on the high concentration of fatty acids in medicinal plants, and particularly polyunsaturated fatty acids (PUFA). Preliminary studies had indicated to a research team in Norway a suppressive influence of fish oils on rodent malaria.6
The unsuspected role of amino acids
A recent paper from Portugal has studied the role of amino acids in depth and comes to very convincing conclusions.7 We extract one figure from their paper. Drinking an aqueous solution of arginine-lysine-valine (RKV) several weeks before the infection significantly reduces the liver infection load (Figure 1).
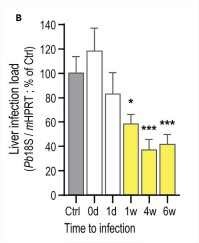
Figure 1 The drinking water (Ctrl) was replaced by RKV-supplemented water on the day of Plasmodium berghei sporozoite injection (0 d), or 1 day, 1 week, 4 weeks, or 6 weeks before infection.
In another figure they clearly show that the efficacy of lysine is much higher than that of valine and arginine. The role of amino acids in malaria infections has mostly been ignored and it is difficult to find papers on this subject.8,9 To gain insight into the mechanism of malarial hemozoin formation, these authors examined the effect of amino acids on beta hematin formation in vitro. Surprisingly some of these amino acids like arginine, histidine, lysine showed a significant inhibition. In contrast beta-hematin formation was enhanced by leucine, isoleucine, valine and methionine.
Breast milk protects neonates against malaria and other diseases during six months. Colostrum is rich in zinc and potassium, but it is also rich in lysine. A systematic review in human milk from 13 countries showed that lysine declined in the two first months of lactation and then remained relatively constant (from 148 to 68 mg/100ml).10 A survey made in 1996 showed that the essential amino acid lysine may be lacking in nutrition for many areas of the world. Average lysine supply was 6 662 mg/day in Western Europe and 2 542 in subSaharan Africa. This is below the FAO/WHO minimal requirement of 2 840 mg/day and might enhance the susceptibility of Africans to Anopheles mosquito bites. And to some extent counterbalance the immunity acquired by multiple malaria infections.11
Artemisia plants are rich in arginine, lysine and valine.12,13 According to Brisibe EA the lysine content in Artemisia annua is 2.17 g/100 g. Checking for other plants we find that Camellia sinensis (green tea) only contains 0.02 g/100 g. Greentea has no antimalarial properties.14 And that the medicinal plants (dry) leaves. Euphorbia thymifolia only contains 0.14 g/100g of lysine, Zinziber officinale 0.14 g/100g and Pavetta indica 0,30 g/100 g15 And for Vernonia amygdalina 0.05 g/100g.were found. All values lower than for Artemisia.16
As noted previously, Artemisia plants are rich in both lysine and zinc. In malaria infections it was noticed that there is a strong synergy between zinc and arginine in the inhibition of hemozoin (Mutaz Akkawi, Al Quds University, Palestine, personal communication). A similar positive synergy has been noticed for zinc and lysine in improving the immune system. In a study in Indonesia, infection rate for several diseases (diarrhea, influenza, pharyngitis…) was lower in zinc group and zinc + lysine group (p<0.065) compared to control group. Zinc + lysine supplementation also increased CD4 count and had better effect compared to zinc supplementation alone.17
In France a comparative study was conducted on the anti-malarial effects of Artemisia annua (containing artemisinin) and Artemisia afra (lacking artemisinin) plant infusions in vitro against the asexual erythrocytic stages of Plasmodium falciparum and the pre-erythrocytic (i. e., liver) stages of various Plasmodium species. Low concentrations of either infusion accounted for significant inhibitory activities across every parasite species and stage studied. These antiplasmodial effects were essentially artemisinin-independent and were additionally confirmed by observations of the parasite apicoplast and mitochondrion. In particular, the infusions significantly incapacitated sporozoites. This seminal work requires further studies on the role of amino-acids and particularly lysine on the liver stages of malaria infections. Publications in this field of research are scarce.But the role of amino acids is complex. Arginine is an amino acid that has been shown in nonclinical studies to be essential in the life cycle of many viruses. Which would imply that Artemisia infusions rich in arginine and other amino acids might enhance Covid infections.18
The amino acid lysine in conjunction or not with arginine plays a key role in malaria prophylaxis. Further in-depth studies and clinical trials on this subject should be encouraged.
None.
Authors declare that there is no conflict of interest.

©2021 Lutgen. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.