MOJ
eISSN: 2374-6920


Research Article Volume 1 Issue 5
1Department of Head and Neck Surgery
2Department of Experimental Therapeutics
Correspondence: Arumugam Jayakumar, Department of Head and Neck Surgery, The University of Texas MD Anderson Cancer Center, Houston, Texas 77030, USA
Received: November 01, 2014 | Published: November 11, 2014
Citation: Jayakumar A, Wu HK, Briggs K, et al. LEKTI, a physiological inhibitor of multiple serine proteinases, suppresses perineural and lymphovascular invasion of human head and neck cancer cells in a mouse orthotopic model. MOJ Proteomics Bioinform. 2014;1(5):124-128. DOI: 10.15406/mojpb.2014.01.00029
Serine Protease Inhibitor Kazal-type 5 (SPINK5) gene encodes 3 different Lympho-Epithelial Kazal-Type-Inhibitor (LEKTI) isoforms. We identified and cloned LEKTI by its constitutive expression in normal oral mucosa and its loss of expression in matched tumor specimens of patients with head and neck squamous cell carcinoma (HNSCC). Stable re-expression of LEKTI in HNSCC OSC19 cells resulted in reduced migration and invasion and enhanced adhesion on variety of ECM substrates in vitro with a concomitant reduction inexpression of endogenous MMP-14, MMP-8, KLK5, and ADAM8. Here, we sought to determine the consequences of LEKTI re-expression on the in vivo changes in the tumor growth and invasion using an orthotopic model of tongue cancer. In the tongue tumors of mice, lymphovascular invasion or perineural spread was found in 100% of tumors derived from parental cell lines but was almost totally absent in all tumors derived from LEKTI-expressing clones. Our work suggests that loss of LEKTI expression in primary tumors might correlate with aggressive biologic behavior and restoration of LEKTI expression by pharmacologic means might be beneficial for patients with HNSCC.
Keywords: SPINK5, LEKTI, lymphovascular invasion, perineural spread, HNSCC
LEKTI, lympho-epithelial kazal-type-inhibitor; SPINK5, serine protease inhibitor kazal-type 5; HNSCC, head and neck squamous cell carcinoma; MMP, matrix metalloproteinases; ECM, extracellular matrix; DMEM, dulbecco’s modified eagle’s medium
Local tumor invasion and regional lymphatic metastasis occur by the attachment of tumor cells to components of the extracellular matrix (ECM) and by degradation of ECM by proteinase enzymes elaborated into the tumor microenvironment.1–4 These processes are regulated by such proteolytic enzymes as serine proteases, cysteine proteases, and matrix metalloproteinases (MMPs) tightly balanced by their endogenous inhibitors in the tumor microenvironment.5,6 Thus, inhibition of such proteinases can disrupt critical steps of invasion and metastasis. Indeed, several proteinase inhibitors have shown importance in a range of cancer types by the loss of expression correlating with advanced tumor progression.7–9 Moreover, proteinase inhibitors have been demonstrated to hold tumor suppressor functions that oppose tumorigenesis, proliferation, angiogenesis, invasion, and metastasis.10–12
An inhibitor of multiple serine proteinases, lymphoepithelial kazal-type inhibitor (LEKTI), was identified and cloned in our laboratory on the basis of its constitutive expression in normal oral mucosa and loss of its expression in matched head and neck squamous cell carcinoma (HNSCC) specimens and multiple HNSCC lines.13 It was also shown by several investigators that LEKTI protein was encoded by SPINK5 gene and mutations in SPINK5has been linked to the inherited disorder known as Netherton Syndrome.14–24
We produced recombinant full length LEKTI and several of its fragments using baculovirus expression system and established that recombinant human LEKTI inhibits a battery of serine proteinases in vitro including plasmin, trypsin, cathepsin G, human KLKs, and elastase, enzymes implicated in the activation of MMPs.25–29 We also showed that pro-LEKTI is processed into four cleavage products ranging in size from 37 to 60kDa in human oral keratinocytes.30
In a recent study, we stably over expressed LEKTI in HNSCC cells and evaluated the effects of restored LEKTI on cellular proliferation, morphology, adhesion, invasion and expression of key MMPs involved in tumor progression. We demonstrated that stable expression of LEKTI in OSC-19 cells resulted in markedly decreased levels of expression of genes encoding MMP-9, MMP-14, KLK5, and ADAM8. Furthermore, LEKTI over expressing cells displayed striking morphological changes are more adhesive and less invasive. These results demonstrate a novel negative regulatory role for LEKTI in modulating the production of key MMPs involved in ECM degradation and suggest that loss of LEKTI in HNSCC tumor cells could have a pivotal role in HNSCC progression.
In the present work, we sought to determine the consequences of LEKTI re-expression on the in vivo changes in the tumor growth and invasion using an orthotopic model of tongue cancer. In the tongue tumors of mice, lymphovascular invasion or perineural spread was found in 100% of tumors derived from vector or parental cell lines but was almost totally absent in all tumors derived from LEKTI-expressing clones (p=0.008 by Fisher’s exact test).
Materials
A human oral squamous cell carcinoma cell strain, OSC19, was a gift from Dr. Theresa Whiteside. Female athymic nude mice aged 6-8 weeks were purchased from Harlan Sprague-Dawley, Inc., Indianapolis, IN; SDS-polyacrylamide gels and pre-stained markers were procured from Bio-Rad Laboratories, Hercules, CA; nitrocellulose membrane from Schleicher & Schull BioScience, Keene, NH; horseradish peroxidase-conjugated goat-anti-mouse IgG (H+L) were obtained from Jackson Immuno Research Laboratories, West Grove, PA; lipofectamine 2000 and pcDNA3.1 (-) were procured from Invitrogen, Carlsbad, CA; ECL kit was obtained from Amersham Bioscience Corporation, Piscataway, NJ; Kodak X-AR5 films were purchased from Eastman Kodak, Rochester, NY; hematoxylin and eosin (H&E) were purchased from Sigma-Aldrich, St. Louis, Mo.
Isolation of OSC-19 LEKTI stable clones
OSC19 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum, 2mM glutamine, and antibiotics. The pro-LEKTI expression plasmid encodes the entire full-length LEKTI polypeptide and has a hexahistidine tag at its C-terminus. OSC-19 cells were stably transfected with the human LEKTI cDNA cloned into the pcDNA3.1 vector or a control vector without LEKTI insert after G418 (0.5 mg/ml) selection.OSC19, OSC19-LEKTI-expressing clone 11, or OSC19-LEKTI-expressing clone 17were cultured at 37 °C in humidified incubator with 5% CO2 and 95% air.
Orthotopic model of tongue cancer
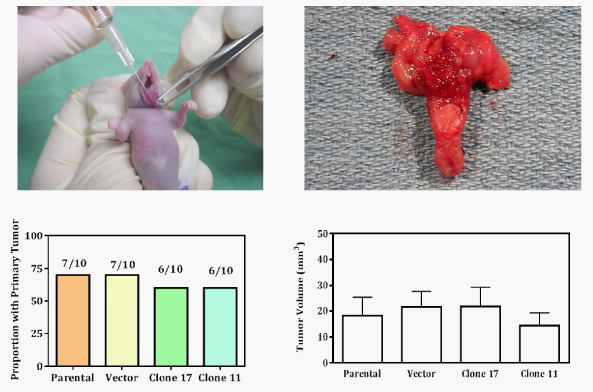
Tongue tumors in mice were generated by modifications of orthotopic xenograph models previously described by Kawashir.31 Mice were cared for in accordance with the Institutional Animal Care and Use Committee and the Department of Veterinary Medicine of M. D. Anderson Cancer Center. Inoculation with OSC-19 parental, vector, LEKTI-expressing clone 11, or LEKTI-expressing clone 17 cells was randomly allocated in forty mice. In preparation for inoculation, cells were trypsinized and resuspended in DMEM at 8x106cells/mL. Animals were marked, weighed, and anesthetized by intraperitoneal injection of ketamine cocktail at 10µl/g of body weight. Inoculation was performed by injecting 25µl of cell suspension to deliver 200,000 cells into the submucosa of the dorsal tongue at the circumvallate line. Mice were maintained on a gel-consistency diet and monitored for weight loss. The animals were euthanized 21days after inoculation and the tongue, cervical lymph nodes, and lungs were removed by microsurgical dissection. Specimens were formalin-fixed, paraffin-embedded, serially sectioned at 200µm levels, and stained with hematoxylin and eosin (H&E). Histologic slides were independently reviewed by two investigators unaware of which inoculation group the specimens belonged.
Summary of gross findings
Inoculating the tongue submucosa of athymic nude mice with OSC-19 cells predictably results in xenograft tumors with locally invasive growth. To examine the changes in invasive growth characteristics of tumors in which LEKTI expression was returned to the microenvironment, we inoculated mice with OSC-19 parental, vector LEKTI-expressing clone 11 or LEKTI-expressing clone 17 cells (Table 1). We reviewed serial sections of histologic specimens for pathologic features of tumor invasion (Table 2). An endophytic growth pattern with or without mucosal ulceration was noted. Grossly normal appearing regional lymph nodes were noted. Gross bone invasion in one of 40 animals was observed. No gross evidence of lung metastases was observed.
Blinded review |
|
Primary tumor |
Regional metastasis |
i. Presence or absence |
i. Presence or absence |
Statistical analysis |
|
i. Comparisons between |
|
|
|
|
Chi Squared Test or Wilcoxon Rank Sum Test |
Table 2 Histological evaluation
OSC-19 parental and vector lines showed 70% and stable clones showed 60% tumorigenicity (Figure 1, bottom left histogram). Moreover, re-expression of LEKTI had no effect on tumor volume (Figure 1, bottom right histogram). However, tumors derived from OSC-19-LEKTI-expressing clone17 demonstrated no perineural invasion compared to perineural invasion in 100% of tumors derived from parental cells (Figure 2, top left histogram). Representative micrographs of typical tumors from parental cells show multiple tumors cells invading into the perineureum of small nerves (Figure 2, top left panel, arrow head), whereas tumors from OSC-19-LEKTI-expressing clone17 show tumor nests in the proximity of nerves without invasion (Figure 2, top right panel, arrow head).
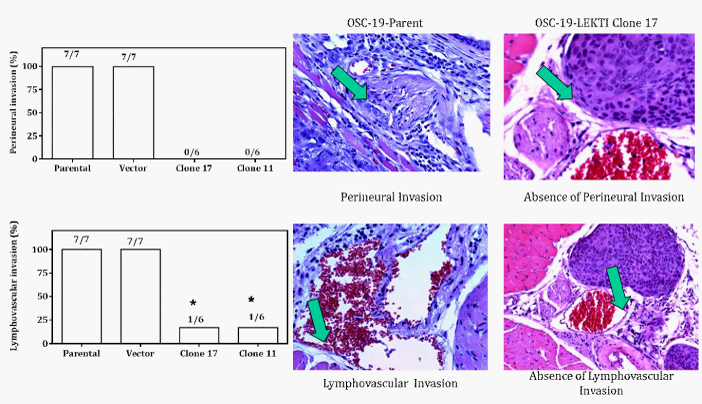
Similarly, tumors derived from OSC-19-LEKTI-expressing clone17 demonstrated 17% lymphovascular invasion compared to lymphovascular invasion in 100% tumors derived from parental cells (Figure 2, bottom left histogram). Representative micrographs of typical tumors from parental cells show multiple tumors cells invading beyond the basement membrane of vascular and lymphatic vessels (Figure 2, bottom left panel, arrow head), whereas tumors from OSC-19-LEKTI-expressing clone17 show tumor nests in the proximity of vessels without invasion (Figure 2, bottom right panel, arrow head). Together, lymphovascular invasion or perineural spread was found in 100% of tumors derived from vector or parental cell lines but was almost totally absent in all tumors derived from LEKTI-expressing clones (p=0.008 by Fisher’s exact test).
Together these findings support a role for LEKTI as a tumor suppressor gene in head and neck squamous cell carcinoma. Indeed, several studies have reported the differential expression of proteinase inhibitors as markers for diagnosis and prognosis in various primary cancer types. For instance, the differential expression of maspin has been associated with good prognostic factors in breast cancer,32 increased survival and longer remission after resection of lung cancer,33 and low malignant potential in ovarian cancer.34 Such studies have proven vital to generating hypotheses of functional implications for further study.
Our findings in a mouse xenograph model of tongue cancer suggest that LEKTI expression results reduce tumor invasion potential in vivo. The prognostic significance of perineural invasion in head and neck squamous cell carcinoma warrants the investigation of mechanisms underlying this process. To date, the understanding of specific mechanisms has been limited by the difficulty in establishing appropriate models. However, analysis of human squamous cell carcinoma specimen by immunohistochemistry has found correlation between Laminin-5 staining and the presence of perineural invasion in head and neck squamous cell carcinoma,35 suggesting that expression of Laminin-5 is an important step in the process of perineural invasion. Thus, mechanisms of disrupting the processing of Laminin-5 in relation to tumor invasion may underlie the effect of LEKTI expression in mouse tumors. Our recent published result showing stable LEKTI re-expression blocks migration and invasion of HNSCC OSC-19 cells36 and immunohistochemical analysis of the graded loss of LEKTI expression along the spectrum of epithelial dysplasia37 suggests a role for the protein as a marker of differentiation that is lost early in tumor progression. Moreover, similar studies to correlate levels of LEKTI expression with perineural invasion in head and neck squamous cell carcinoma tumor specimen are ongoing.
In summary, in a xenograph model of tongue cancer, tumors derived from LEKTI-expressing clones of an invasive head and neck cell line demonstrated limited pathologic features of lymphovascular and perineural invasion. Our findings suggest an important negative regulatory role for LEKTI in modulating extracellular matrix degradation. The loss of LEKTI expression in head and neck squamous cell carcinoma may represent a critical event in tumor progression to an invasive cellular phenotype. Restoration of LEKTI expression by pharmacologic means might be beneficial for patients with HNSCC.
Funding: Supported in part by the NIH-NCI P50 CA097007, NIH R01 DE013954, NIH P30 CA016672, Alando J. Ballantyne Distinguished Chair in Head and Neck Surgery award, Michael A. O’Bannon Endowment for Cancer Research, NIH INRS Award T32 CA060374, and AAO-HNSF Percy Memorial Grant.
A.J, T.D.S, and G.L.C provided intellectual input into the experimental design and execution of the study. A.J and T.D.S wrote the manuscript. K.B. generated LEKTI stable clones. H.K.W. and T.D.S performed most of the experiments. V.R. helped to organize the data.
The author declares no conflict of interest.

©2014 Jayakumar, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.