Journal of
eISSN: 2373-633X


Article shortly illustrates a case of a patient with malignant metastatic uveal melanoma, who developed multiple brain metastases after ophtalmectomy. The article focuses on the difference in pre-contrast T1 signal between the original lesion and its brain metastases in current case and tackles the issues of improved melanoma lesion detection with susceptibility weighted imaging (SWI).
Keywords: malignant uveal melanoma, melanoma metastases, melanin T1 hyperintensity, susceptibility weighted imaging, T2* relaxation
MRI, magnetic resonance imaging; WI, weighted imaging; DWI, diffusion weighted imaging; SWI, susceptibility weighted imaging; GD: gadolinium
Malignant melanoma of the uvea is considered to be the most common primary intraocular disease in adults, with the incidence of 5.2-5.7 cases per million per year with a mean age of 53.1 The factors associated with poor systemic prognosis in patients are:
Metastatic sites vary with most common being brain, liver, lung, bone and kidney. The diagnosis is established by means of opthalmoscopy, fluorescin angiography and ultrasound. Imaging differential diagnosis is rather broad and might include retinoblastoma, choroidal hemangioma, choroidal nevi, choroidal detachment, choroidal cysts, neurofibroma, schwannoma of the uvea and even some metastatic tumors. High resolution MRI is an imaging technique of choice for depicting retrobulbar/extraocular tumor extension.
It is known that melanin holds unique properties, which cause paramagnetic effects with shortening of T1 and T2 relaxation times, and therefore resulting in elevated signal in T1-WI and decreased signal in T2-WI images.1 There are other publications on this matter2,3 which also describe the T2* weighted imaging role in improving the overall detection of metastatic melanoma.4
A 62 years old man, 6 month post-ophtalmectomy with histologically confirmed malignant melanotic melanoma of the choroid, presenting in November 2014 with ataxia, dizziness, memory disorders and headache complaints. Prior brain and orbits MR-exams of this patient, done 6 month before, demonstrated broad-based voluminous process confined within the left globe, measuring approximately 14x4 mm on axial scans; no brain lesions werefound at that time (Figure 1A-1C).
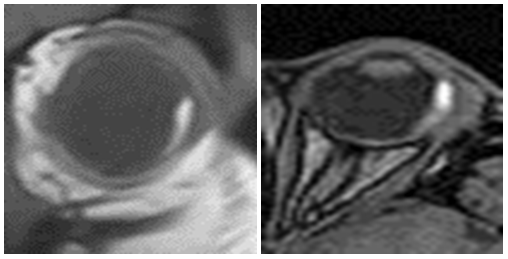
Figure 1B & 1C Which is attributable to paramagnetic melanin properties.
Figure 1 MR-study of the orbits demonstrates intermediately low in homogenous signal in from the broad-base voluminous lesion in the lateral aspect of the left globe in T2-WI sequence.
MR protocol
MR-exam was performed on Seiemens Magnetom Avanto 1.5T using T1, T2, DWI (b-factor = 0, 500, 1000, 1500), SWI and T1post-gadolinium sequences (Figure 2A-2E).
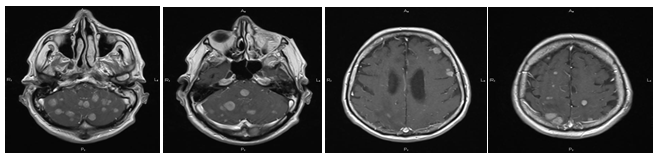
Figure 2E Larger lesions are surrounded by vasogenic edema (dotted arrows).
Figure 2 Brain MRI demonstrates multiple uveal melanoma metastases (arrows) with heterogeneous low signal in T2, DWI, SWI sequences.
Key findings
The patient turned out to have multiple round and oval-shaped space-occupying lesions of the brain, localized in both hemispheres and infratentorial structures, including the pons, larger being surrounded by peripheral vasogenic edema. All of the lesions demonstrated heterogeneouslydecreased signal in T2-WI and DWI (most distinctly at a b-factor of 500) sequences, iso/hypointense signal in pre-contrast T1-WI sequence and marked enhancement in T1 post-gadolinium sequences. It should be noted that SWI sequence was able to demonstrate even the smallest lesions, not visible prior to contrast enhancement, due to their paramagnetic properties, which were most likely pronounced due to the melanin contents (Figure 2).
One of the key puzzles of this case is the difference in T1 signal between uveal lesion and its brain metastases. A study by Gaviania et al.,4 analyzed signal characteristics of 120 newly detected metastatic brain lesions from patients with malignant melanoma and patients with lung cancer. In this study, the biopsy samples of patients with malignant melanoma were graded according to histopathologic findings into:
The brain metastases of the patients, whose biopsies showed heavy melanin were more likely to demonstrate T1 hyperintensity than the metastases in patients with amelanotic or lightly melanotic tumors. It was also mentioned that melanoma metastases were 5 times more likely to demonstrate T2*-related signal intensity loss due to susceptibility effect, rather than lung metastases.4 Another study by Enochs et al.,3 postulated that the T1 hyperintensity of melanin positively correlates with the amount of bound paramagnetic metal (mostly iron and copper), which depends on the scavenging abilities of melanin, with the latter being subject to variations. This approach could possibly provide an explanation for the difference in non-enhanced T1 signal from the original uveal melanoma of the eye (T1-hyperintense) and subsequent brain metastases (T1-iso/hypointense) in the current case, since the presence of melanin in the original tumor was histologically confirmed. It may be suggested that T1 signal difference might be attributed either to difference in melanin contents or difference in melanin metal scavenging abilities. Further research is needed to support or decline this assumption.
Additionally and in support of study by Gaviania et al.,4 it is necessary to mention the role of susceptibility weighted imaging (SWI) in improved detection of melanoma metastases (Figure 3A-3C). Due to the discussed paramagnetic properties of melanotic lesions, the decay of transverse magnetization is caused by a combination of spin-spin relaxation and magnetic field inhomogeneities, which is a T2* relaxation. Gradient echo sequences, like SWI, allow visualization of this effect, since in spin-echo sequences the transverse relaxation caused by magnetic field inhomogeneities is eliminated by the 180° pulse.5 Thus it is worth to routinely add SWI sequence to MR-protocols when examining patients with metastatic melanoma for better lesion detection.
None.
Author declares that there is no conflict of interest.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.