Journal of
eISSN: 2469 - 2786


Research Article Volume 10 Issue 1
1Laboratories, Indoor Environmental Consulting and Labs (IECL), Australia
2Consultancy, Indoor Environmental Consulting and Labs, Australia
Correspondence:
Received: March 28, 2022 | Published: April 18, 2022
Citation: Wilkie AD, Venz L, Richards B. Mould surface sampling techniques and collection efficiency on paper-faced gypsum board. J Bacteriol Mycol Open Access. 2022;10(1):1-7 DOI: 10.15406/jbmoa.2022.10.00317
Surface sampling techniques for non-viable fungi in building environments are useful tools for investigators in determining hazards to occupants. However, data regarding capture efficiency in this context is limited. Our data demonstrates that collection efficiency of Bio-Tape surface capture medium on paper-faced gypsum board only captures between half and three-quarters of mould present on the surface. Surface sampling using a dry-swab technique showed similar efficiency of capture to tape lift samples. ‘Surface air’ samples had poor collection efficiency and should be avoided where possible in preference to other sampling options. Finally, we propose a sampling strategy based on non-viable microscopy techniques followed by molecular analysis for validation and speciation of samples of interest. Improvements in sampling and data analysis techniques for mould sampling of buildings will aid in providing meaningful results to help building inspectors evaluate health hazards.
Keywords: mould inspection, fungal sampling, non-viable, surface sampling, collection efficiency, capture efficiency, bio-tape, tape-lift, swab, fungi, building inspection, surface-air sampling
ACGIH, american conference of governmental industrial hygienists; ASTM, american society for testing and materials; CIRS, chronic inflammatory response syndrome; EPA, environmental protection agency; ERMI, environmental relative moldiness index; HERTSMI2, health effects roster of type-specific formers of mycotoxins and inflammagens; IEP, indoor environmental professional; IICRC, international institute of cleaning and restoration certification; NGS, next generation sequencing; PCR, polymerase chain reaction; qPCR, quantitative PCR; RO, reverse osmosis; SD, standard deviation
Mould growth in the indoor environment pose a hazard to health and wellbeing of occupants.1,2 Sampling of buildings for mould is a useful tool in identification of potential hazards to occupants.3,4 Indeed, visual inspection of premises for mould contamination may not capture the entire picture.5 Mould exposure has been linked to adverse health reactions.6–12 and as such effective monitoring of mould is an important task.
The majority of mould sampling to assess buildings for mould contamination in Australia is conducted using non-viable sampling techniques. This involves using spore trap cassettes to collect air samples and several options for testing surface samples. A strong preference for non-viable sampling over other methods has been observed not only in Australia but worldwide due to the low cost and time effective analysis.13 Additionally, it should be noted that non-viable sampling can detect the presence of both viable and non-viable mould – which have been indicated as potential allergens.2,10,14–16 The presence of fungal fragments which have also been indicated as potential contaminants17,18 and inactive fungal material can still release toxins.19
Surface sampling for non-viable mould is typically conducted using tape lift surface samples20–22 (e.g. Zefon Bio-Tape) in a technique first described by Flegel.23 However, other methods to collect non-viable surface mould samples include swab samples, Swiffer cloth dust samples, bulk samples4,24 and so called ‘surface air’ samples. Tape lift samples have become the surface sampling medium of choice for non-viable samples due to the ease of use, repeatability of sampling, ease of processing and analysis and cost effectiveness. Non-viable mould samples are analysed by microscopy, often aided by a staining procedure to improve contrast.25 Several methods for analysis of non-viable surface samples exist22 but reliable quantitative methods are typically considered more useful.26 Meider and Messal26 have recently published data outlining a quantitative method of analysis of tape lift samples.26
Viable, molecular and other techniques for mould analysis
Viable analysis: Viable mould sampling involves collecting samples from a site and culturing the samples on a growth medium (e.g. potato dextrose agar or malt extract agar). The colony morphology and growth characteristics when combined with the microscopic morphology can provide much more accurate identification of mould – to the species level in many cases.27,28 However, such analysis costs more due to the time for analysis, the requirements for more consumables and materials and the skill involved in proper identification of mould species. Analysis also takes considerably more time as the samples must be incubated for several days prior to analysis.4
Flow cytometry: Flow cytometry-based techniques have been investigated as an alternative route for mould identification.29 Flow cytometry utilises various parameters of individual particles such as size and circularity combined with data on the spectral properties of different cells including auto-fluorescence profiles to identify mould.30 This technique has the advantage of eliminating analyst bias in the identification process but is limited by the availability and reliability of software on identifying mould genera. Indeed, many mould species present very divergent spore morphologies and positive identification of species based on data from fluorescence spectra is yet to be achieved.29 Nevertheless, this technique has the potential to be a very fast sampling technique for fungi with innovations such as the portable flow cytometer InstaScope by DetectionTek.31
Molecular techniques: PCR based techniques for the identification of mould have been employed for several years, probably most commonly in the ERMI or HERTSMI-2 assays.32,33 These assays tested for common mould species and made comparisons between indoor and outdoor samples. Recently, Next Generation Sequencing (NGS) has been employed to identify mould in relation to mould related illness such as Chronic Inflammatory Response Syndrome (CIRS).34,35 NGS has the distinct advantage of not having to look for specific targets but have the ability to identify what species are present in a sample.35 This technology has incredible potential for identification of fungi, but the high cost involved is prohibitive in the widespread adoption of these techniques. More readily available DNA based techniques using qPCR systems represent a middle ground in which mould species can be identified with accuracy in a timely fashion and without the prohibitive cost of current NGS technology.
Visual inspection: Visual inspection of buildings for fungal contamination remains a key part of determining the extent of contamination in structures. While visual inspection does not definitively identify mould, trained inspectors should be able to determine fungal growth with decent reliability. A recent paper has applied image analysis with visual mould inspections to quantify mould growth in structures.36 Visual inspections will likely remain a key factor in determining mould contamination of buildings for the foreseeable future.
Why use microscopy when molecular techniques exist?
Microscopic analysis for mould provides a cost-effective method of determining the extent of mould contamination in sampled locations. While it cannot provide information on the species of mould present, it can provide insight into if a potential water damage issue exists, if a hidden mould issue is contributing to poor air quality or if remediation works have significantly reduced mould levels.3,5,22 Microscopic analysis of non-viable samples can also provide insight about the activity of mould in samples through assessing for fungal structures such as conidiophores, whereas a molecular based technique does not.37 The prohibitive costs or time limitations of molecular techniques will likely slow uptake of these techniques - but present a significant improvement in care for sufferers of chronic mould exposure investigations such as for CIRS patients.35
Improvement in the consistency and reliability of data obtained from microscopic analysis of mould is therefore important in improving the consistency of collection and analysis of mould samples in a residential setting. This article aims to improve accuracy and comparability of surface mould samples to help inspectors detect trends in data and isolate issues in premises.
In order to assess the collection efficiency of surface sampling apparatus, experiments were designed to compare the amount of mould collected to the estimated total mould present on the surface. For each experiment, a set area of surface – 4cm2 (the size of a Zefon Bio-Tape readable area) was sampled by one of the techniques listed, followed by several subsequent samplings of the same location. The sum of all sampling results was used to obtain a reasonable approximation of the total mould on the surface. Mould levels of the initial sampling were then compared to the total mould levels and an efficiency calculated.
Paper-faced gypsum board was selected as the material to be tested due to its suitability to foster mould growth, its ubiquitous use in construction of buildings, its availability and cost.38 Pieces of 10mm thick gypsum board were incubated in a high humidity environment to obtain heavily mould contaminated material. Mould genera most commonly detected on the material were Aspergillus spp. and Stachybotrys spp. and associated hyphae.
Bio-tape sampling
Surface samples were collected using Zefon Bio-Tapes by placing the adhesive section on the material and pressing on the entire back of the adhesive slide with firm pressure. It should be noted that error in sampling using tape lifts could be introduced by poor sampling technique such as moving the slide while sampling, inconsistent pressure on the slide during sampling, and attempting to sample rough or uneven surfaces, therefore care must be taken when collecting samples to ensure sampling technique remains consistent.
Swab sampling
Surface samples were collected using dry swabs by wiping all sides of the swab across the set area in three directions (horizontal, vertical and diagonal). Swabs heads were extracted and transferred to sterile tubes for analysis. The swab was suspended in 5ml of RO (reverse osmosis) water and vortexed for 30 seconds. The suspension was then analysed using a Neubauer counting chamber combined with microscopic analysis. Swab samples for non-viable mould has advantages and disadvantages.39 First, a swab sample can be taken practically on any surface – and is very useful for locations in which sampling with a tape lift would not be possible.3,14 Additionally, swabs are very economical and easy to use.
‘Surface air’ sampling techniques
‘Surface air’ samples are an adaptation from a sampling technique classed ‘surface air system’ and bulk dust sampling techniques. ‘Surface air system’ samples are viable samples collected onto agar plates by direct deposition of spores onto the plate using a specialised pump. In contrast, ‘Surface air’ samples are non-viable samples collected from a set surface area onto a spore trap cassette. ‘Surface air’ samples have been used by inspectors to assess surfaces for mould growth in instances where tape lift samples are inappropriate. They are generally collected by using a wall sampling tube and a spore trap cassette collecting air directly adjacent to a surface. Bulk dust sampling involves using a vacuum and a filter cassette to collect dust for analysis, primarily for culture or molecular based techniques.4,14 Academic literature regarding ‘Surface air’ samples is extremely scarce and the use of this technique requires further investigation. This technique may have arisen from misinterpretation of collection of bulk dust sampling techniques or ‘surface air system’ viable sampling.
This article outlines two sampling techniques for ‘surface air’ samples and estimates of their efficacy in order to help improve consistency using this type of mould sampling. The collection efficiency of this sampling technique listed in this article is by no means a definitive efficiency, but rather a guidance value to help give a reasonable estimate of the actual mould levels on a surface.
Technique 1 – using a spore trap cassette and a wall sampling tube a sample is collected from a set area (16x25mm – same as a Bio-Tape sampling slide) by hovering the inlet of the sampling tube 3mm from the surface and slowly moving across the entire area. Airflow was set at 15 litres per minute as the manufacturer recommendation for Air-O-Cell cassettes. Sampling time is set to 2 minutes to prevent overloading of the spore trap.
Technique 2 – using a spore trap cassette and a wall sampling tube a sample is collected from a set area (16x25mm) by gently pressing the inlet of the sampling tube onto the surface in multiple overlapping areas until the entire area has been covered thoroughly. Airflow was set at 15 litres per minute as the manufacturer recommendation for Air-O-Cell cassettes. Sampling time is set to 1 minute to prevent overloading of the spore trap.
Statistical analysis
Unpaired t tests were performed using GraphPad QuickCalcs online software (Graphpad Software Inc.) and Boxplots were generated using Microsoft Excel (Microsoft Inc.). Mean, standard deviation and interquartile ranges were calculated. Separate experiments were performed N number of times as listed in each figure and table.
Bio-Tape collection efficiency
In order to examine the collection efficiency of Zefon Bio-Tape surface samplers the following methodology was used. Multiple heavily mould contaminated sections of paper-faced gypsum board were sampled repeatedly in the exact same location 10 times. The results are shown in Table 1 & Figure 1. Our results show that the capture of mould from the gypsum decreased with each sampling to minimal levels – indicating that the vast majority of mould from the sampled area had been collected. Additionally, the data indicates that the initial Bio-Tape sampler on the paper face of gypsum had an efficiency of approximately 59.5% (Figure 2 & Table 2). Furthermore, using data from multiple consecutive collections showed a collection efficiency from each sampling with a mean of 86.2%, or disregarding outliers a mean of 61.2% and a median of 56.4% (Table 2). This data is presented in Figure 2. It should be noted that a high amount of variation was observed when comparing the efficiency of each sample to the efficiency of the sample taken immediately prior. Taken together these data indicate that the true collection efficiency of Bio-Tape surface samplers lies at approximately 60% for paper faced gypsum board. This value should not be taken as a definitive efficiency of sampling, more a reference point indicating that a tape lift sample on paper-faced gypsum board will likely only collect approximately 50-70% of the mould present on a surface.
|
Tape-lift number |
Exp 1 |
Exp 2 |
Exp 3 |
Exp 4 |
Exp 5 |
Exp 6 |
|
1 |
697779 |
172048 |
333861 |
233198 |
405505 |
22802 |
|
2 |
240971 |
64518 |
121651 |
25004 |
66202 |
18008 |
|
3 |
107789 |
44955 |
26040 |
10494 |
23708 |
6737 |
|
4 |
32000 |
17619 |
12567 |
19174 |
13862 |
4923 |
|
5 |
10883 |
9846 |
5959 |
6478 |
16453 |
1943 |
|
6 |
4275 |
23708 |
5959 |
1555 |
12567 |
2721 |
|
7 |
34519 |
5700 |
3368 |
1943 |
8421 |
1814 |
|
8 |
20729 |
11142 |
2721 |
4016 |
6866 |
2073 |
|
9 |
17101 |
2591 |
1943 |
1425 |
5571 |
1166 |
|
10 |
2202 |
3109 |
2591 |
1684 |
4923 |
1036 |
|
Sum |
1168248 |
355238 |
516662 |
304971 |
564079 |
63222 |
Table 1 Total fungal structures per cm² observed from consecutive tape-lift samples from multiple experiments
Mean = 49540, SD = 11676. N = 6.
|
Tape-lift number |
Exp 1 |
Exp 2 |
Exp 3 |
Exp 4 |
Exp 5 |
Exp 6 |
|
1 |
0.5973 |
0.3607 |
0.4843 |
0.6462 |
0.7647 |
0.7189 |
|
2 |
0.3453 |
0.7898 |
0.3750 |
0.3644 |
0.1072 |
0.1633 |
|
3 |
0.4473 |
0.3741 |
0.6968 |
0.2141 |
0.4197 |
0.3581 |
|
4 |
0.2969 |
0.7308 |
0.3919 |
0.4826 |
1.8272* |
0.5847 |
|
5 |
0.3401 |
0.3947 |
0.5588 |
0.4742 |
0.3378 |
1.1869 |
|
6 |
0.3929 |
1.4000 |
2.4079* |
1.0000 |
0.2400 |
0.7638 |
|
7 |
8.0741* |
0.6667 |
0.2404 |
0.5652 |
1.2500 |
0.6701 |
|
8 |
0.6005 |
1.1429 |
1.9545* |
0.8077 |
2.0667* |
0.8154 |
|
9 |
0.8250 |
0.5625 |
0.2326 |
0.7143 |
0.3548 |
0.8113 |
|
10 |
0.1288 |
0.8889 |
1.2000 |
1.3333 |
1.1818 |
0.8837 |
Table 2 Collection efficiency based on comparisons of consecutive sampling. Interquartile range (IQR) = 0.46801, Outliers* defined as IQR x 1.5 and shown in red on table
Mean = 1.00, SD = 1.0657. N = 6.
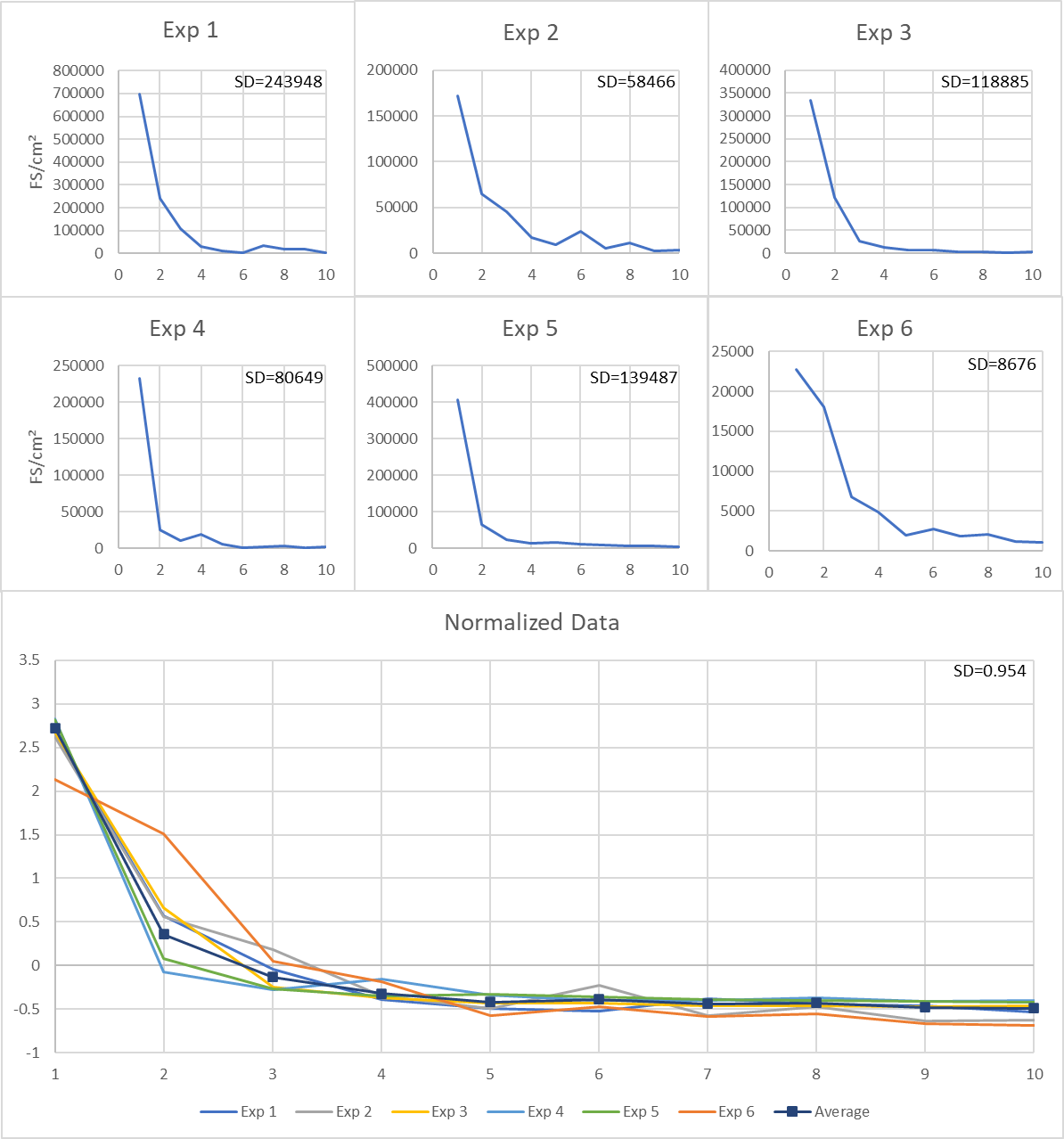
Figure 1 Total fungal structures from consecutive sampling for each experiment. (Bottom) Normalized data from consecutive Bio-Tape collection experiments (Exp1 - Exp6). Separate experiments are listed on the normalised graph. N = 6.
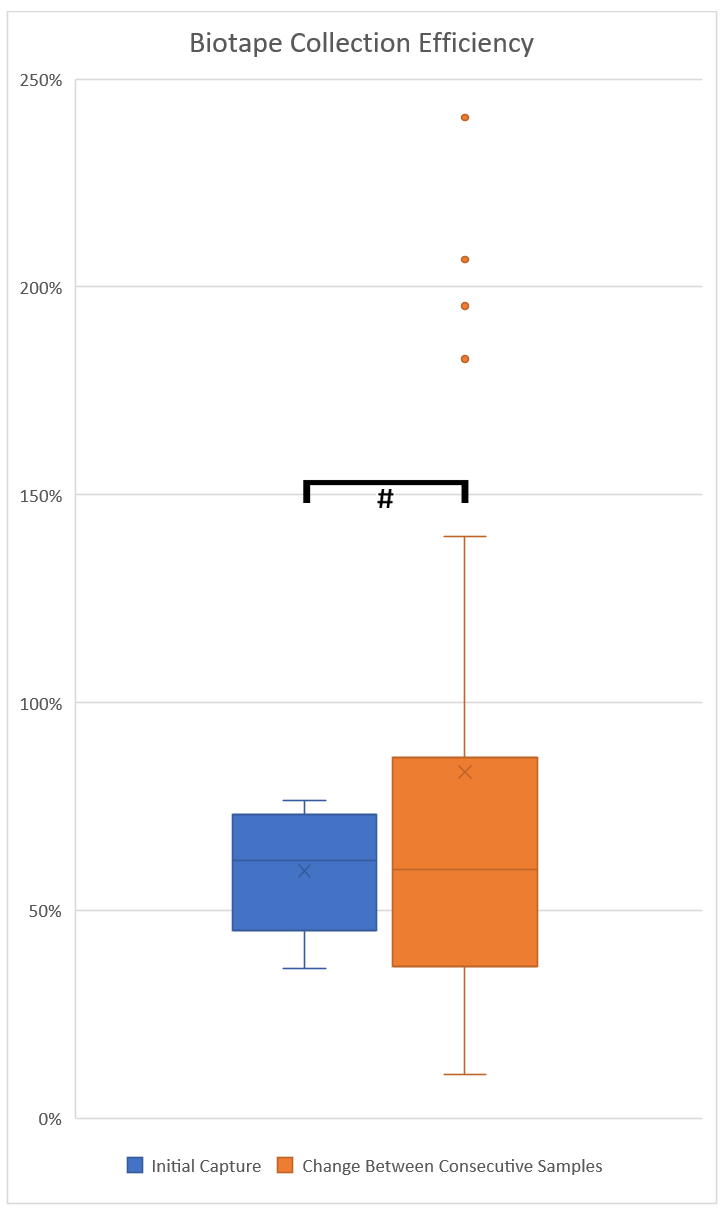
Figure 2 Capture efficiency of Bio-Tape surface samplers based on initial capture efficiency compared to total mould from sampling (blue). Capture efficiency calculated from differences observed between collection of each sample as compared to the previous sample (orange). An outlier at 807% for ‘Change Between Consecutive Samples’ is not shown on the graph in order to retain a useful scale. # Not statistically significant P = 0.5889. N = 6.
Swab sample efficiency
In order to assess dry-swab samples the same methodology as per Bio-Tape samples was adapted. A swab sample was collected using the methodology indicated, followed by subsequent tape lift samples to aid in gauging the total mould present. The results are shown in Figure 3 & Table 3. Provided consistent, repeatable sampling technique of a precisely set area is conducted, capture efficiency appears similar to that of tape lift samples. Our data indicates the efficiency of swab sampling in this context is approximately 59.2%.

Figure 3 Data for collection efficiency of surface mould sample capture using a swab sampler (blue) followed by re-sampling with a Bio-Tape sampler (orange). * Statistically significant P = 0.0022. N = 5.
|
Collection efficiency |
Total spore count in tested area |
||||
|
Swab |
Bio-tape taken after swab |
Swab |
Bio-tape |
Sum of sampling |
|
|
Exp 1 |
0.6827 |
0.1061 |
2620000 |
407277 |
3837903 |
|
Exp 2 |
0.4719 |
0.2976 |
1245000 |
785082 |
2638079 |
|
Exp 3 |
0.5777 |
0.2499 |
690000 |
298423 |
1194368 |
|
Exp 4 |
0.3956 |
0.2576 |
410000 |
266941 |
1036361 |
|
Exp 5 |
0.8322 |
0.0728 |
300000 |
26235 |
360487 |
|
Mean |
0.5920 |
0.1968 |
1053000 |
356792 |
1813439 |
|
SD |
0.1726 |
0.1003 |
949287 |
276916 |
1403269 |
Table 3 Collection efficiency of swab samples and a subsequent Bio-Tape sample calculated as a ratio of the total mould collected from all sampling in a location
N = 5.
‘Surface air’ efficiencies
In order to assess the capture efficiency of ‘surface air’ samples on paper-faced gypsum board samples were collected using one of the two methodologies outlined, followed by subsequent sampling with Bio-Tape samplers. Results are shown in Table 4 & Figure 4. For samples collected by sampling technique 1 (hovering above surface) efficiency of collection was approximately 2.4%. This efficiency is far below that of either Bio-Tape samplers or swab samples. Low capture efficiency of this sampling method may introduce error in the results obtained, and the reliability of the data is likely low.
|
Collection efficiency |
Fungal Structures per cm² |
|
|
|||||
|
|
Technique 1 (Hover) |
Technique 2 (Touch) |
Bio-tape taken after 'Surface air' |
Technique 1 |
Technique 2 |
Bio-tape 1 taken after ‘Surface air’ |
Bio-tape 2 taken after ‘Surface air’ |
Sum of Sampling |
|
Exp 1 |
0.0024 |
0.3619 |
0.2095 |
1203 |
180857 |
104714 |
213000 |
499774 |
|
Exp 2 |
0.0742 |
0.1334 |
0.2595 |
86886 |
156143 |
303857 |
624000 |
1170886 |
|
Exp 3 |
0.0075 |
0.1873 |
0.1510 |
11218 |
279667 |
225429 |
977000 |
1493313 |
|
Exp 4 |
0.0152 |
0.0842 |
0.7621 |
4543 |
25143 |
227500 |
41333 |
298519 |
|
Exp 5 |
0.0202 |
0.0642 |
0.6950 |
12476 |
39571 |
428500 |
136000 |
616548 |
|
Mean |
0.0239 |
0.1662 |
0.4154 |
23265 |
136276 |
258000 |
398267 |
815808 |
|
SD |
0.0289 |
0.1193 |
0.2894 |
35869 |
105647 |
119018 |
392555 |
498062 |
Table 4 Collection efficiency of ‘Surface air’ sampling techniques and subsequent Bio-Tape sampling calculated as a ratio of the total mould collected in each experiment
N = 5.

Figure 4 Collection efficiency of ‘Surface air’ samples collected using Technique 1 (blue) and Technique 2 (orange) as compared to the initial Bio-Tape sample collected after sampling (grey) with each of the ‘Surface air’ methods. * Statistically significant P = 0.0321. ** Statistically significant P = 0.0168. # Not statistically significant P = 0.1129. N = 5.
For samples collected by sampling technique 2 (touching the surface in overlapping sections) efficiency of collection was assessed by conducting the air sample, followed by multiple Bio-Tape samples in the same sampled location. Efficiency of sampling using this technique was approximately 16.6% (Figure 4). Sampling efficiency was again far lower than that of Bio-Tape or swab samples, however 6-7-fold higher than technique 1. These findings highlight how important sampling technique is to obtain consistent results.
Multiple surface sampling of paper-faced gypsum board demonstrated that the initial sampling from a Bio-Tape in this context will only capture approximately 59.5% of mould on the surface (Figure 2). Determining the average reduction in mould levels between consecutive sampling from each dataset also demonstrated the approximate efficiency of mould capture with a mean of 61.2% when ignoring outliers in the data and a median of 56.4%, in agreement with the estimation from the initial capture (Table 2). These findings are significant as it demonstrates that sampling is underestimating the presence of mould on paper faced gypsum board, and mould investigators and hygienists should consider adjusting their recommendations accordingly.
Interestingly an anomaly was detected in sampling of surfaces multiple times. Typically, mould levels detected were reduced with each subsequent sampling. However, once several samples had been collected an increase in mould levels was observed (typically at 5-7 samplings on the same location), followed by the levels dropping again (Figure 1 & Table 1). A possible explanation for this phenomenon is that the adhesive has disrupted the surface enough to remove the top layer of the paper and reveal further mould contamination in subsequent layers. This finding highlights the importance of physical removal of mould contaminated material in preference to cleaning techniques as mould ingrained in the material and not on the surface will not be removed by a surface treatment, in agreement with guidance from the IICRC on remediation.2
The sampling efficiency of a swab sample appeared to be similar to that of a tape lift sample at approximately 59% (Figure 3). However, it is worth noting that errors and inconsistencies from this type of sampling, and sampling technique is far more likely that with a tape lift sample. It should also be noted that swab samples require additional processing in laboratories and Bio-Tapes are often preferred by laboratories due to this reason. Additionally, to sample consistently with swabs is considerably more difficult. The sampling size must be set – and variations from the sampling size will directly affect results. Furthermore, there is always the possibility that areas of the surface within the defined area will be missed by the sampler and there could be significant variation in the efficiency of the swab and sampler in collecting material.27 Finally, non-viable mould swab samples introduce another step in the processing of samples before analysis as mould on the swab must be extracted and transferred for analysis which may potentially introduce error into the counts.3,27
The efficacy of surface sampling with the ‘surface air’ type sample showed very poor capture of mould with only 2.4% for technique 1 and 16.6% for technique 2 (Figure 4). It should be noted that samples collected using technique 2 had previously been sampled using technique 1 and as such results are likely slightly lower than the values indicated. Subsequent sampling of this material with a Bio-Tape sampler resulted in far greater capture efficiency, even with the reduction in total spores present on the material due to prior sampling taken into account. Poor sampling efficiency may result in bias from missing mould spores, or capture of spores with a strong preference to spores which readily become airborne. These ‘surface air’ samples do not have a well-defined sampling technique and a large amount of variation in sampling techniques can render data obtained from these techniques useless. For instance, the distance from the material the tube is held from the material (or if it is touched to the material) makes a vast difference in the sampling results, as we have demonstrated. Additionally, if the sampling tube is scraped across the surface the results will be much greater than samples in which the tube is gently pressed to the surface due to disruption of the mould spores on the surface. It should also be noted that technique 2 involved touching the surface of the sampled material with the end of the sampling tube and as such a dry swab sample would have similar potential of damaging the material, but with much greater collection efficiency. Use of this type of sampling is therefore discouraged – with a possible exception being testing of a high value item in which touching the item is to be avoided. Regardless, if this technique is used our data provides a better estimate of the mould levels on an item based on calculations on the efficiency listed here.
Combining Microscopy with qPCR
Development of a sampling technique in which the sample can be microscopically examined followed by molecular analysis using qPCR may be a viable option in balancing high-quality data with costs of analysis. Microscopy techniques using phase-contrast for instance do not require input of chemicals4,40 which may affect the samples use in PCR analysis. A microscopic count using phase contrast – or even a stain to increase contrast once tested for interference with PCR analysis could provide initial information on mould composition, likelihood of water issues, hidden mould, etc. qPCR could then be used to obtain quantitative data of common mould species present using specific primers and mitigate uncertainty such as the distinction between Aspergillus and Penicillium species inherent in microscopic analysis.33 Following the initial microscopic analysis, areas of concern could then employ further testing using molecular techniques to determine the exact composition of samples to the species level. This data would be extremely useful in cases where health concerns have been noted and the data is valuable for healthcare practitioners in regards to illnesses and toxicities. Speciation of the fungi present in these cases may also aid in determining which compounds and mycotoxins are likely being produced and contributing to adverse health reactions of occupants.
We have demonstrated limitations in current sampling methodologies for non-viable mould analysis such as sampling inefficiencies of tape lift and swab samples. We have also examined the efficiency of ‘surface air’ samples and have concluded that ‘surface air’ samples should only be used in exceptional circumstances in which tape lift or swab samples have the potential of damaging the item. Finally, we propose a sampling strategy which utilises existing sampling techniques such as spore trap cassettes and tape lift samples analysed by microscopy followed by molecular identification of mould species on an as required basis.
Investigations of other materials such as pine boards, plywood panels, chipboard, or concrete in collection efficiency of sampling will provide mould inspectors and remediators more reliable data on the mould present on surfaces. Preliminary data indicates that capture efficiency of Bio-Tapes on pine boards also lies in the 50-70% range (data unpublished). Further studies on the efficacy of sampling techniques should be conducted to confirm these findings and help achieve true reference values for collection efficiency of sampling media.
Development of a protocol for pairing traditional microscopic analysis with molecular analysis will aid in maintaining low-cost sampling for mould while providing more relevant data to healthcare practitioners. Such a technique has the advantage of being fast and cost effective where only microscopic analysis is required, however adds the option of closer examination of the material for potential health hazards by identification to the species level. This protocol also has the advantage over viable sampling as results from both microscopic examination and molecular analysis may be achievable in less than 24 hours.
The authors would like to thank IECL and NLR Restorations for funding this research.
The authors declare no conflicts of interest.

©2022 Wilkie, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.