Journal of
eISSN: 2469 - 2786


Research Article Volume 6 Issue 5
Institute of Industrial Biotechnology, GC University, PAKISTAN
Correspondence: Shahriari Mahdi, Associate professor of Pediatric Hematology/Oncology, Shiraz University of Medical Sciences, Shiraz, Iran
Received: October 26, 2017 | Published: September 12, 2018
Citation: Mukhtar H, Bashir H, Nawaz A, et al. Optimization of growth conditions for Azotobacter species and their use as biofertilizer. J Bacteriol Mycol Open Access. 2018;6(5):274-278. DOI: 10.15406/jbmoa.2018.06.00217
The present study is concerned with the optimization of growth conditions for Azotobacter species. Five isolates were isolated from soil and Azotobacter IIB-3 found to be the best; along with strain of Azotobacter vinelandii NRRL-14641. The medium M2 gave maximum growth (0.65 and 0.75 mg/ml) of Azotobacter vinelandii and Azotobacter IIB-3. Optimum temperature, pH and incubation period for growth of Azotobacter were 30ºC, 8.0 and 48hrs, respectively. The growth of Azotobacter was also studied with the supplementation of growth medium with different carbon and nitrogen sources. 2.5% mannitol (as C-source) and 2% (NH4)2SO4 (as N-source) were found best for both Azotobacter vinelandii (1.16mg/ml) and Azotobacter IIB-3 (1.24mg/ml). 24 hrs old inoculum at a level of 1% was found best for the growth both Azotobacter vinelandii and Azotobacter IIB-3. The effect of Azotobacter biofertilizer was studied on maize plants in pot experiment and it was found that plants inoculated with Azotobacter gave better growth as compared to control plants.
Biofertilizer is a large population of a specific or a group of beneficial microorganisms for enhancing the productivity of soil either by fixing the atmospheric nitrogen , by solubilizing soil phosphorus or by stimulating plant growth through synthesis of growth promoting substances.1 Biofertilizer supply in addition to nitrogen considerable amount of organic matter enriching structure of soil. Inoculants of these microorganisms have proved their technical feasibility, economic viability and social acceptability. Biofertilizers are the most advanced biotechnology invention which is necessary to support developing organic agriculture, sustainable agriculture, green agriculture and non pollution agriculture. The biofertilizer can increase the output and improve the quality of agricultural products.2 Biofertilizers have definite advantage over chemical fertilizers. Chemical fertilizers supply only nitrogen whereas biofertilizers provide in addition to nitrogen certain growth promoting substances like hormones, vitamins, amino acids etc. to the plants.3 Crops have to be provided with chemical fertilizers repeatedly to replenish the loss of nitrogen utilized for crop growth. On the other hand biofertilizers supply nitrogen continuously throughout the entire period of crop growth in field under favorable conditions.4 Biofertilizers are formed by mixing of microorganisms with some carriers like lignite and peat moss in a certain amount. Some of the commonly used biofertilizers are Azotobacter biofertilizers, Phospho biofertilizers, Rhizo biofertilizers and Trichoderma biofertilizers. A large number of soil microorganisms are used as biofertilizer. Some of these are phosphate solubilizing microbes including Pseudomonas striata, Bacillus megaterium and Bacillus subtilis).5,6 Some fungi like Aspergillus and Penicillium are potential solubilizer of bound phosphates.7 Azotobacter is one of the most important microorganisms, which is widely used as biofertilizer (Pereira and Shetty, 1987). Azotobacter is Gram negative and polymorphic bacterium i.e. having different sizes and shapes. Their size ranges from 2-3 umin diameter and of various lengths. Azotobacter spp. are sensitive to acidic pHhigh salts and temperature above 35C.7
Azotobacter spp. shows well growth in a medium containing both acetate and glucose as as carbon source.8 Azotobacter spp. show rapid growth on nitrogen free medium.9 However under nitrogen fixing conditions Azotobacter spp. grow well.10 Hydrogen ion concentration (pH) is also an important parameter that directly affects the growth of living organisms. Azotobacter grows well in Burk s liquid medium at pH range of 7 to 9 but fail to grow below the pH of 6. Moreover other parameters such as temperature, incubation time, aeration rate and inoculum size also have effects on growth of Azotobacter species.
The main objective of the present study is to provide a cheaper source of nitrogen to the agriculture industry and to meet the needs of crops as there is spread of inflation day by day due to high prices of petroleum which influences the prices of chemical nitrogenous fertilizers. Also for a sustainable agriculture system, it is imperative to utilize renewable input that can maximize the ecological benefits and minimize the environmental hazards.
Microorganisms
Bacterial strains belonging to Azotobacter spp. were isolated from different soil samples of local habitat through serial dilution method. 1.0 gram of soil sample was suspended in 100ml of sterilized water in 250ml Erlenmeyer flask and was vigorously shaken. From this stock solution serial dilutions were prepared, and 0.5ml from each dilution was spread on the solidified surface of Burk agar medium plates {(g/L) K2HPO4, 0.64; KH2PO4, 0.16; NaCl, 0.20; MgSO4, 0.20; CaSO4, 0.05; FeSO4, 0.003; NaMoO4, 0.01; Glucose, 10; Agar, 15). One strain of Azotobacter vinelandii NRRL-14641 was also provided by the culture bank of Institute of Industrial Biotechnology, GC University Lahore. Different morphological and biochemical tests were performed for the identification of isolates, such as Gram staining; capsule staining, motility test, endospore test, protease test and catalase test.
Inoculum preparation
For the preparation of bacterial inoculum, a loop full of bacteria from slant was transferred to 250ml of Erlenmeyer flask containing 50ml of sterilized nitrogen free medium in laminar air flow chamber. Flask was then incubated in shaker at 30ºC and 200rpm for 24hrs.
Batch experiments
Batch experiment for the growth of Azotobacter was carried out in 250ml shake flasks containing culture medium that was prepared by dissolving 0.2g ammonium sulphate, 0.8g dipotassium hydrogen phosphate, 0.2g magnesium sulphate, 0.1g calcium sulphate, 1ml of Mo solution (0.1mg/ml),1ml of FeSO4 solution (1.0mg/ml) and 20g manitol in 1000ml of distilled water. The flasks were cotton plugged and autoclaved at 121C(15 lb/inch2 pressure) for 15min. then the medium was cooled down at room temperature and inoculated by adding 1ml of inoculum with the help of micropipette in laminar air flow chamber. The flasks were then incubated at 30ºC in a rotary shaker for 48hrs at 200rpm for growth of microorganisms. Fermentation broth was subjected to centrifugation at 6000 rpm for 10 min in pre weighed glass centrifuge tubes. Supernatant was removed and pelleted mass was dried out in oven at 90C over night. After drying, the centrifuge tubes were again weighed and the weight of biomass was obtained by subtracting the pre weight with after weight.
Selection of culture medium
Different culture media were screened for the maximum growth of Azotobacter spp. the composition of these media is given below.
M1: (g/L) glucose, 10.0; di potassium hydrogen phosphate, 0.64; potassium dihydrogen phosphate , 0.16; NaCl, 0.2; MgSO4.7H2O, 0.2; CaSO4.2H2O, 0.05; NaMoO4.2H2O, 0.01; FeSO4, 0.003. pH 7.1
M2: (g/L) Ammonium Sulphate,0.2; di potassium hydrogen phosphate , 0.8; MgSO4, 0.2; CaSO4, 0.1; , Mo solution, 1ml (0.1mg/ml); FeSO4, 1ml (1.0mg/ml); manitol, 20.0.pH 7.1
M3:(g/L) NaCl, 0.4; MgSO4.7H2O, 0.4; KH2PO4, 0.16; K2HPO4, 0.64; CaCl2, 0.084; NaMoO4.2H2O, 0.002; H3BO4, 0.003; FeSO4.7H2O, 0.006; CoSO4,0.012; CuSO4,5H2O, 0.0001; ZnSO4.7H2O,0.012; Sucrose, 40.pH 7.2
M4: (g/L) K2PO4, 0.2; MgSO4, 0.2; CaCl2, 0.2; FeCl3, 0.05 ml of 10%; NaMoO4, a trace; Manitol, 15.0. pH 7.2
M5: (g/L) Beef Extract, 3.0; NaCl, 8.0; Peptone,5.0. pH 7.1
M6: (g/L) glucose, 20.0 MgSO4.7H2O, 0.5; K2HPO4, 0.2; CaCl2, 0.05; FeCl3.6H2O, 0.10; NaMoO4.2H2O, 0.05. pH 7.4.
Application of biofertilizer
Effect of Bio fertilizer on growth of plants was determined by pot experiment in which seeds of maize inoculated with bacterial growth were sowed in sterilized soil in disinfected pots. Seeds were watered and placed under sunlight. A control was also taken in which seeds were not inoculated with bacterial strains. After the growth of plants, the size of leaves and roots were recorded in all experimental and control pots.
Data of Table 1 shows the screening of isolates for maximum growth on synthetic medium in shake flasks. Five different isolates of Azotobacter were screened and Azotobacter IIB-3 was found best strain showing highest dry cell biomass production i.e. 0.74 mg/ml. so Azotobacter IIB-3 was selected for further studies along with Azotobacter vinelandii NRRL- 14641. The source of Azotobacter IIB-3 was the soil sample taken from the Ligia garden of GC University Lahore. Other four isolates Azotobacter IIB-1, Azotobacter IIB-2, A.IIB-4 and IIB-5 showed lesser amount of dry cell biomass i.e. 0.23, 0.38, 0.29 and 0.46 mg/ml, respectively.
S No. |
Isolates |
Dry cell mass (mg/ml) |
1 |
Azotobacter IIB-1 |
0.23 |
2 |
Azotobacter IIB-2 |
0.38 |
3 |
Azotobacter IIB-3 |
0.74 |
4 |
Azotobacter IIB-4 |
0.29 |
5 |
Azotobacter IIB-5 |
0.46 |
6 |
Azotobacter NRRL-14641 |
0.63 |
Table 1 Screening of Azotobacter spp. for maximum biomass production
Identification of isolates
The isolates were subjected to morphological and biochemical tests Table 2). It was observed that colonies were white to off white in colour while the growth of colonies were brown to blakish due to production of melanin. The cells were ovoid in shape and their size was about 2-3 um in diameter. Isolates were observed gram negative, motile and form the cyst. The endospore formation and protease activity was not found but catalase activity was shown by the isolates. According to Bergey’s Manual (Kreig et al., 1984) the isolates belonged to the genus Azotobacter.
Criteria |
Characteristics |
Shape |
Rods |
Size |
2-3 um diameter |
Colony shape |
Raised circular |
Colony colour |
Off white |
Capsule formation |
+ |
Gram reaction |
Gram –ve |
Motility |
+ |
Endospore formation |
- |
Cyst formation |
+ |
Florescent pigment |
+ |
Capsular slime production |
+ |
Protease activity |
- |
Catalase activity |
+ |
Table 2 Morphological & Biochemical properties of the selected isolate
Selection of Culture medium
The growth of Azotobacter was investigated using different culture media through batch fermentation (Figure 1). Maximum growth (0.75mg/ml) of Azotobacter IIB-3 was observed in medium M2. While maximum growth (0.65 mg/ml) was observed in M2 medium by Azotobacter vinelandii. Growth of Azobacter was maximum when medium M2 was used because it contained all the essential nutrients in sufficient amounts required by the bacteria for growth e.g., it contained MgSO4, CaSO4, NaMoO4 and K2HPO4 etc. it has been reported that MgSO4 prolongs the viability of microorganisms suspended in potassium phosphate buffer (Gunter, 1954). Magnesium ions are essential not only for oxidative function of bacterial cells but also significant in the maintenance of permeability and transport mechanism (Goucner and Kocholaty, 1954).
Effect of pH
The effect of pH on Azotobacter spp. were studied by varying the pH of the medium from 5.0 to 9.0 (Figure 2). Azotobacter IIB-3 showed maximum growth i.e. 0.77mg/ml at pH 8.0. While A.vinelandii showed maximum growth of 0.7 mg/ml at pH 8.0. Azotobacter grows on wide range of pH (Kreig and Holt, 1987) but their growth is more on neutral pH (Waehner et al., 2007).
Effect of incubation period
The effect of incubation period on Azotobacter spp. were studied by varying the incubation period 24-120 hrs (Figure 3). Azotobacter IIB-3 showed maximum growth i.e. 0.91mg/ml after incubation period of 48hrs. While A.vinelandii showed maximum growth of 0.85mg/ml in 48hrs incubation period. Incubation period is directly proportional to the growth of bacteria up to a certain extent and after that growth of bacteria start decreasing that can be attributed to the decrease in the supply of nutrients to microorganisms or may be accumulation of some toxic compounds in the broth.
Effect of incubation temperature
The effect of incubation temperature on Azotobacter spp. were studied by varying the incubation temperature 25-40 C hrs (Figure 4). Azotobacter IIB-3 showed maximum growth i.e. 0.93mg/ml after incubation at 30C. While A.vinelandii showed maximum growth of 0.88 mg/ml at 30C incubation temperature. A little temperature rise increases the growth rate because the velocity of enzyme-catalyzed reaction also increases with it and as the rate of increases; microorganism grows faster (Prescott et al., 1999). Optimum temperature for the growth of Azotobacter has been reported as 30C but some species can grow at optimum temperature of 34C (Chen et al., 1984).
Screening of Carbon Sources
Fermentation medium was supplemented with 2.5% of different carbon sources (Figure 5). Azotobacter IIB-3 showed maximum growth (0.95mg/ml) in the presence of manitol as carbon source. While A.vinelandii showed maximum growth of 0.92mg/ml in manitol medium. Carbon is essential component of growth medium but an excess of carbon source can cause undesirable metabolic waste products to accumulate into the microorganism while deficiency of carbon source cause lesser growth as well (Prescott et al., 1999). Shawky et al. (2004) has also found manitol as a best carbon source for growth of A. vinelandii. Fermentation medium was supplemented with different amounts (0.5–3.0 %) of manitol (Figure 6). Azotobacter IIB-3 showed maximum growth (1.12mg/ml) in the medium containing 2.55 manitol while A.vinelandii showed maximum growth of 1.10 mg/ml in 2.5% manitol.
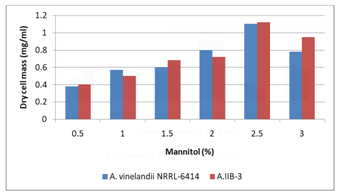
Figure 6 Effect of different concentrations of mannitol in the culture medium on growth of Azotobacter species.
Screening of nitrogen sources
Fermentation medium was supplemented with 2% of different nitrogen sources (Figure 7). Azotobacter IIB-3 showed maximum growth (1.12mg/ml) in the presence of ammonium sulphate as nitrogen source. While A.vinelandii showed maximum growth of 1.11mg/ml in medium containing ammonium sulphate as nitrogen source. Although Azotobacter grows in nitrogen free medium, but it has been reported that nitrogen source was supplied to Azotobacter to decrease the lag phase. Gadkari and Stolp (1974) have found ammonium sulphate as best nitrogen source and have reported in the presence of ammonium sulphate, Azotobacter growth was increased. Fermentation medium was supplemented with different concentration (1.0–3.0 %) of ammonium sulphate (Figure 8). Azotobacter IIB-3 showed maximum growth (1.24mg/ml) in the medium containing 2% ammonium sulphate. While A.vinelandii showed maximum growth of 1.16mg/ml in 2.5% ammonium sulphate.
Effect of inoculum
The effect of inoculum age on the growth of Azotobacter spp. was studied by using inoculum of different ages i.e. 20–32hrs (Figure 9). Azotobacter IIB-3 showed maximum growth i.e. 1.22mg/ml in 24hrs old inoculum. While A.vinelandii showed maximum growth of 1.15mg/ml in the medium containing inoculum of 24hrs old. Effect of inoculum age for the optimization of microorganism growth is also an important parameter. It has been reported 24 hrs old inoculum gave maximum growth of Azotobacter. The effect of inoculum size on the growth of Azotobacter spp was studied. For this purpose different sizes of inoculum i.e. 0.5–2.5% were used in the fermentation medium (Figure 10). Azotobacter IIB-3 showed maximum growth i.e. 1.24mg/ml in 1% of inoculum size. While A.vinelandii showed maximum growth of 1.15mg/ml in the medium containing inoculum size of 1%.
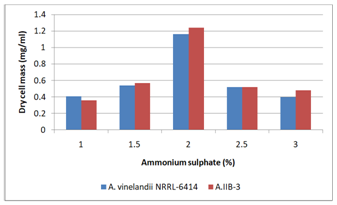
Figure 8Effect of different concentrations of ammonium sulphat in the culture medium on growth of Azotobacter species.
Application of Azotobacter biomass as biofertilizer
Effects of biofertilizers were studied on the growth performance of maize (Zea mayz) plants (Table 3). Maximum growth (195mm) of leaves and a less extensive (110mm) system of roots was observed in the experimental plants incubated with Azotobacter vinelandii. While the seeds inoculated with Azotobacter IIB-3 showed leaves size of 197 mm and roots of length 152mm. Results showed that plants inoculated with bacterial strains gave maximum leaf growth which confirms that these contained the maximum nutrients available for their growth and minimum proliferation of roots because sufficient nutrients were present in the soil for uptake of roots due to which their penetration into the soil was not so much high. It has been reported that inoculation of plants with Azotobacter increases their growth and yield as well.10
S. No. |
Treatments |
Leaves size (mm) |
Roots size |
(mm) |
|||
1 |
Control |
109 |
152 |
2 |
Azotobacter IIB-3 |
197 |
105 |
3 |
Azotobacter IIB-5 |
183 |
137 |
4 |
Azotobacter vinelandii NRRL-6414 |
195 |
110 |
Table 3 Effect of biofertilizer application on the growth performance of maize plant
It was concluded from the above studies that the optimization of cultural and nutrition parameters resulted in enhancement in the growth of Azotobacter species during fermentation. Furthermore, the biomass of Azobacter species was proved to be good source of biofertilizer.
None.
Author declares that there is no conflict of interest.

©2018 Mukhtar, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.