Journal of
eISSN: 2473-0831


Research Article Volume 8 Issue 1
1Laboratory of Biophysics and Molecular Modeling Genesis, State Secretariat of Education of Parana, Brazil
2Green Land Landscaping and Gardening, Seedling Growth Laboratory, Brazil
3Faculty of Chemistry, California South University, USA
4Department of Marine Science, University of Calcutta, India
Correspondence: Ricardo Gobato, Laboratory of Biophysics and Molecular Modeling Genesis, State Secretariat of Education of Parana, Bela Vista do Paraíso, Parana, 86130-000, Brazil
Received: January 05, 2019 | Published: January 21, 2019
Citation: Gobato R, Gobato MRR, Heidari A, et al. New nano-molecule kurumi - C13H20BeLi2SeSi/C13H19BeLi2SeSi, and raman spectroscopy using ab initio, hartree-fock method in the base set CC-pVTZ and 6-311G** (3df, 3pd). J Anal Pharm Res. 2019;8(1):1?6. DOI: 10.15406/japlr.2019.08.00302
The work characterizes the Raman spectrum of the new nano-molecule C13H20BeLi2SeSi / C13H19BeLi2SeSi, nano-molecule Kurumi. Calculations obtained in the methods Restrict Hartree-Fock of the first principles (ab initio), on the set of basis used indicate that the simulated molecule C13H20BeLi2SeSi / C13H19BeLi2SeSi features the structure polar-apolar-polar predominant. The set of basis used that have are correlation-consistent polarized Triple-zeta (CC-pVTZ) and Pople’s basis sets six gaussian functions in the shell, three double zeta Gaussian functions, Slater type orbitals with polarization function (6-311G** (3df, 3pd)). In the CC-pVTZ base set, the charge density in relation to 6-311G** (3df, 3pd) is 50% lower. The length of the molecule C13H20BeLi2SeSi is of 15.799Å. The Raman spectrum was calculated indicating the characteristic of the nano-molecule and their frequency (cm-1) were obtained in the set of bases used. The highest for Raman scattering activities peaks are in the frequency 3,348cm-1 with 7.107609729Å4/amu and 2,163cm-1 with 8.902805583Å4/amu, for CC-pVTZ and 6-311G** (3df, 3pd), respectively. As the bio-inorganic molecule C13H20BeLi2SeSi is the basis for a new creation of a biomembrane, later calculations that challenge the current concepts of biomembrane should advance to such a purpose. The new nano-molecule Kurumi is well characterize computationally. As its scientific name 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane.
Keywords: biomembrane, CC-pVTZ, hartree-fock method, nano-molecule Kurumi, raman spectroscopy, 6-311G** (3df, 3pd)
The structure of the C13H20BeLi2SeSi is a bio-inorganic seed molecule for a biomembrane genesis that defies the current concepts of a protective mantle structure of a cell such as biomenbrane1–3 to date is promising, challenging. Leaving to the Biochemists their experimental synthesis. Structures of a liquid crystal such as a new membrane may occur, micelles.1
A large investigative study in the literature was carried out, but was not found of the structure studied here. There is an absence of a referential of the theme, finding only one work in (Gobato et al.).1 The present study is an extension of the work already begun in the creation of ab initio, computational methods of quantum chemistry, to prove the possible structure of the bio-inorganic nano-molecule genesis, C13H20BeLi2SeSi. Their characterization by well-known ab initio methods of extreme precision is essential for their future experimental achievement. Therefore, the ab initio study using RHF (Restrict Hartree-Fock method)4-13 and the set of basis used is CC-pVTZ10-14 and 6-311G ** (3df, 3pd).7,14-37
The calculation of the Raman spectrum of the nano-molecule Genesis has the purpose of deepening the knowledge and characterization of this. The work characterizes the Raman spectrum of the nano-molecule C13H20BeLi2SeSi. Calculations obtained in the ab initio RHF (Restrict Hartree-Fock method).4-13 The set of basis used that have are CC-pVTZ10-14 and 6-311G** (3df, 3pd).7,14-18
Hartree-fock methods
The Hartree-Fock self–consistent method4-13 is based on the one-electron approximation in which the motion of each electron in the effective field of all the other electrons is governed by a one-particle Schrodinger¨ equation. The Hartree-Fock approximation takes into account of the correlation arising due to the electrons of the same spin, however, the motion of the electrons of the opposite spin remains uncorrelated in this approximation. The methods beyond self-consistent field methods, which treat the phenomenon associated with the many-electron system properly, are known as the electron correlation methods. One of the approaches to electron correlation is the Møller-Plesset (MP)4-18 perturbation theory in which the Hartree-Fock energy is improved by obtaining a perturbation expansion for the correlation energy.4,5 However, MP calculations are not variational and can produce an energy value below the true energy.4,5
The exchange-correlation energy is expressed, at least formally, as a functional of the resulting electron density distribution, and the electronic states are solved for self-consistently as in the Hartree-Fock approximation.4-13
The full Hartree-Fock equations are given by
(1)
The minimal basis sets are not flexible enough for accurate representation of orbitals. Using multiple functions to represent each orbital, have the double-zeta basis set allows us to treat each orbital
separately when we conduct the Hartree-Fock calculation:
(2)
In Equation (2) 2s atomic orbital expressed as the sum of two STOs (Slater type orbitals).4,5 The two equations are the same except for the value of ξ which accounts for how large the orbital is. The constant d determines how much each STO will count towards the final orbital.4,5
The vast literature associated with these methods suggests that the following is a plausible hierarchy:
(3)
The extremes of ‘best’, FCI, and ‘worst’, HF, are irrefutable, but the intermediate methods are less clear and depend on the type of chemical problem being addressed. The use of HF4-13 in the case of FCI was due to the computational cost.
Hardware and software
For calculations a computer models was used: Intel® CoreTM i3-3220 CPU @ 3.3 GHz x 4 processors,38 Memory DDR3 4 GB, HD SATA WDC WD7500 AZEK-00RKKA0 750.1GB and DVD-RAM SATA GH24NS9 ATAPI, Graphics Intel® Ivy Bridge.1
The ab initio calculations have been performed to study the equilibrium configuration of C13H20BeLi2SeSi molecule using the GAMESS.14,15 The set of programs Mercury 3.8,39 Avogadro40,41 are the advanced semantic chemical editor, visualization, and analysis platform and GAMESS14,15 is a computational chemistry software program and stands for General Atomic and Molecular Electronic Structure System14,15 set of programs. For calculations of computational dynamics, the Ubuntu Linux version 16.10 system was used.42 The Graphic was edited in Origin software,43 for comparison of the spectra obtained in the set of bases used.
1Creative Commons, (CC BY 4.0), https://creativecommons.org/licenses/by/4.0/. “Ivy Bridge”, https://pt.wikipedia.org/wiki/Ivy_Bridge, Avaliable in: August 31, 2018.
Our results take place from the findings of the molecular dynamics of bio-inorganic nano-molecule C13H20BeLi2SeSi. The results were summarized and shown in Figures (1) and (2), Tables (1) and (2).
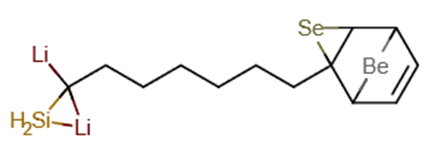
Figure 1 Representation of the molecular structure of C13H19BeLi2SeSi, Nano-molecule Kurumi, 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane, obtained through computer via ab initio calculation method RHF/CC-pVTZ.
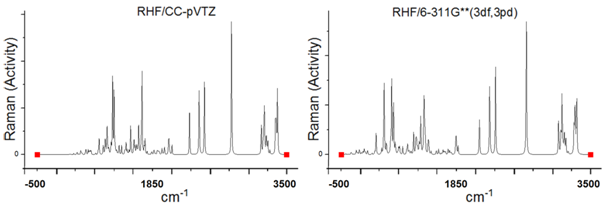
Figure 2 Representation of the Raman spectrum of C13H20BeLi2SeSi - Frequency (cm-1) for Raman scattering activities (SR, Å4/amu) -using computer programs GAMESS, in the ab initio for the RHF method, in sets of basis RHF/CC-pVTZ in left and 6-311G**(3df, 3pd) in right.
|
υ(cm-1) |
SR(Å4/amu) |
υ(cm-1) |
SR(Å4/amu) |
υ(cm-1) |
SR(Å4/amu) |
υ(cm-1) |
SR(Å4/amu) |
|
492 |
1.776731 |
812 |
1.028877 |
1212 |
1.041326 |
3124 |
1.903406 |
|
556 |
1.002196 |
852 |
1.041603 |
1228 |
1.750528 |
3132 |
3.589672 |
|
588 |
1.72755 |
924 |
1.360705 |
1604 |
1.699802 |
3140 |
5.264159 |
|
596 |
1.146249 |
996 |
3.12066 |
1612 |
1.730788 |
3148 |
1.707891 |
|
612 |
1.638659 |
1004 |
1.241567 |
1660 |
1.055684 |
3164 |
1.250803 |
|
620 |
3.087802 |
1044 |
1.055248 |
2172 |
2.528493 |
3172 |
2.086607 |
|
628 |
1.303999 |
1052 |
1.632863 |
2180 |
7.796951 |
3180 |
1.003229 |
|
684 |
1.370946 |
1068 |
1.058814 |
2188 |
1.353183 |
3196 |
1.523681 |
|
700 |
1.941684 |
1092 |
1.09638 |
2604 |
2.449866 |
3316 |
3.894353 |
|
708 |
8.448529 |
1124 |
3.183402 |
2612 |
4.231962 |
3324 |
3.953024 |
|
716 |
2.009311 |
1132 |
1.198932 |
2620 |
4.275745 |
3332 |
3.687871 |
|
724 |
1.737035 |
1164 |
1.702475 |
2628 |
1.112317 |
3340 |
2.315845 |
|
732 |
6.968083 |
1172 |
3.720249 |
3084 |
1.454578 |
3348 |
7.10761 |
|
740 |
1.80891 |
1180 |
8.947658 |
3092 |
3.202109 |
3356 |
2.854032 |
|
748 |
1.090269 |
1188 |
1.75441 |
3116 |
1.435717 |
- |
- |
Table 1 Table contains the frequencies (cm-1) for Raman scattering activities (SR, Å4/amu) of the C13H20BeLi2SeSi molecule via ab initio methods, set base RHF/CC-pVTZ for the infrared spectrum
|
υ(cm-1) |
SR(Å4/amu) |
υ(cm-1) |
SR(Å4/amu) |
υ(cm-1) |
SR(Å4/amu) |
υ(cm-1) |
SR(Å4/amu) |
|
490 |
2.182596 |
1050 |
1.851075 |
1932 |
1.885078 |
3094 |
6.21376 |
|
595 |
3.420969 |
1057 |
1.707548 |
1939 |
3.535241 |
3101 |
3.519756 |
|
602 |
7.279393 |
1092 |
1.388367 |
2072 |
1.463697 |
3108 |
1.427854 |
|
609 |
1.764814 |
1099 |
1.118466 |
2079 |
6.915053 |
3115 |
1.154649 |
|
630 |
1.192582 |
1106 |
1.109801 |
2086 |
4.240445 |
3122 |
1.346301 |
|
637 |
1.038282 |
1113 |
3.899045 |
2093 |
1.062849 |
3129 |
2.315457 |
|
693 |
1.755585 |
1120 |
1.974696 |
2156 |
2.115886 |
3136 |
1.096041 |
|
700 |
2.135401 |
1148 |
1.021344 |
2163 |
8.902806 |
3150 |
1.711815 |
|
707 |
7.719121 |
1155 |
3.520772 |
2170 |
2.324113 |
3262 |
3.23569 |
|
714 |
2.723003 |
1162 |
6.018009 |
2583 |
1.551622 |
3269 |
3.008913 |
|
721 |
1.36844 |
1169 |
3.541951 |
2590 |
5.920065 |
3276 |
5.031301 |
|
728 |
2.834444 |
1176 |
2.474677 |
2597 |
3.47486 |
3283 |
3.414571 |
|
735 |
5.297166 |
1183 |
1.335813 |
2604 |
2.774907 |
3290 |
2.106915 |
|
742 |
1.255979 |
1218 |
1.504666 |
3045 |
3.454342 |
3297 |
4.656712 |
|
756 |
1.287419 |
1225 |
1.55933 |
3052 |
1.894141 |
3304 |
5.694054 |
|
931 |
1.12492 |
1603 |
1.285718 |
3073 |
1.020319 |
3311 |
1.308788 |
|
1015 |
2.295633 |
1610 |
1.928284 |
3080 |
2.52559 |
- |
- |
|
1043 |
1.507847 |
1617 |
1.257005 |
3087 |
2.277247 |
- |
- |
Table 2 Table contains the frequencies (cm-1) for Raman scattering activities (SR, Å4/amu) of the C13H20BeLi2SeSi molecule via ab initio methods, set base RHF/6-311G**(3df,3pd) for the infrared spectrum
Nano-molecule Kurumi
The Figure 1 presentation of the molecular structure of C13H20BeLi2SeSi / C13H19BeLi2SeSi, Nano-molecule Kurumi, 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane.
Raman spectrum
In Figure (2) we have representation of the Raman spectrum of C13H20BeLi2SeSi-Nano-molecule Kurumi, 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane. Frequency (cm-1) for Raman scattering activities (SR, Å4/amu) -using computer programs GAMESS, in the ab initio for the RHF method, in sets of basis RHF/CC-pVTZ in left and 6-311G**(3df, 3pd) in right, obtained using computer software GAMESS. The Graphic edited in Origin software, for comparison of the spectra obtained in the set of bases used.
RHF/CC-pVTZ
The Table 1 contains the frequencies (cm-1) for Raman scattering activities (SR, Å4/amu) of the Nano-molecule Kurumi, 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane.via ab initio methods, set base RHF/CC-pVTZ for the infrared spectrum.
The highest for Raman scattering activities peaks are in the frequency 708cm-1 with 8.448529382Å4/amu, 732cm-1 with 6.968083358Å4/amu, 1,180cm-1 with 8.947657695 Å4/amu, 2,180cm-1 with 7.796950601Å4/amu and 3,348cm-1 with 7.107609729Å4/amu for RHF/CC-pVTZ.
RHF/6-311G**(3df,3pd)
The Table 2 contains the frequencies (cm-1) for Raman scattering activities (SR, Å4/amu) of the C13H20BeLi2SeSi molecule, of the Nano-molecule Kurumi, via ab initio methods, set base RHF/6-311G**(3df,3pd) for the infrared spectrum.
The highest for Raman scattering activities peaks are in the frequency 602cm-1 with 7.279393384Å4/amu, 707cm-1 with 7.719121469Å4/amu, 2,079cm-1 with 6.915053417Å4/amu, 2,163cm-1 with 8.902805583Å4/amu and 3,094cm-1 with 6.213759835Å4/amu for RHF/6-311G** (3df, 3pd),
The calculations made so far admit a seed molecule at this stage of the quantum calculations of the
arrangement of the elements we have chosen, obtaining a highly reactive molecule with the shape polar-apolar-polar. Its structure has polarity at its ends, having the characteristic polar-apolar-polar. Even using a simple base set the polar-apolar-polar characteristic is predominant. The set of bases used that have the best compatible, more precise results are CC-pVTZ and 6-311G(3df, 3pd). In the CC-pVTZ base set, the charge density in relation to 6-311G(3df, 3pd) is 50% lower. The structure of the bio-inorganic seed molecule for a biomembrane genesis that challenge the current concepts of a protective mantle structure of a cell such as biomembrane to date is promising, challenging. Leaving to the biochemists their experimental synthesis.
The study evolves to construct a biomembrane from the seed molecule. The calculations defaut already performed admit a hydrophobic and hydrophilic molecule, the basis of the formation of a micelle and or biomembrane, as the default template. But kurumi molecule is polar-apolar-polar characteristic is predominant, ie. a hydrophilic-hydrophobic-hydrophilic molecule, thus presenting another standard for the construction of a biomembrane, which is even more innovative because a bio-inorganic molecule. Going beyond imagination, the most innovative and challenging proposal of the work advances the construction of a structure compatible with the formation of a "new DNA", based now on the seed molecule.
Our discussion takes place from the findings of the molecular dynamics of bio-inorganic nano-molecule C13H20BeLi2SeSi. The results were summarized and shown in Figure 1 & Figure 2, Table 1 &Table 2.
The Figure (1) representation of the molecular structure of C13H19BeLi2SeSi, Nano-molecule Kurumi, 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane, obtained through computer via ab initio calculation method RHF/CC-pVTZ.
The Figure (2) represents of the Raman spectrum of C13H20BeLi2SeSi - Frequency (cm-1) for Raman scattering activities (SR, Å4/amu) - using computer programs GAMESS, in the ab initio for the Restrict Hartree-Fock method, in sets of basis CC-pVTZ and 6-311G**(3df, 3pd), obtained using computer software GAMESS. Graphic edited in origin software, for comparison of the spectra obtained in the set of bases used.
The Table 1 & Table 2 present the Raman spectrum frequencies for CC-pVTZ and 6-311G** (3df, 3pd), respectively, for Raman scattering activities (SR, Å4/amu), SR>1.
The highest for Raman scattering activities peaks are in the frequency 3,348cm-1 with 7.107609729Å4/amu and 2,163cm-1 with 8.902805583Å4/amu, for CC-pVTZ and 6-311G** (3df, 3pd), respectively. It presents “fingerprint” between the intermediate frequency intervals presented in Table 1 &Table 2.
Calculations obtained in the ab initio RHF method, on the set of basis used, indicate that the simulated molecule, C13H20BeLi2SeSi, is acceptable by quantum chemistry. Its structure has polarity at its ends, having the characteristic polar-apolar-polar.
The 6-311G** (3df, 3pd) set of basis exhibits the characteristic of the central chain, with a small density of negative charges, near the ends of the Carbons of this. In the CC-pVTZ base set, the charge density in relation to 6-311G (3df, 3pd) is 50% lower. It is characterized infrared spectrum of the molecule C13H20BeLi2SeSi, for absorbance and transmittance, in Hartree-Fock method in the set of bases CC-pVTZ and 6-311G (3df, 3pd). The dipole moments CC-pTZV are 3.69% bigger than 6-311G** (3df, 3pd).
The new nano-molecule Kurumi is well characterize computationally. As its scientific name 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane. Getting Kurumi name, which means boy in Tupi-Guarani language, which are indigenous inhabitants of southern Brazil.
Our study has so far been limited to computational simulation via quantum chemistry, an applied theory. Our results and calculations are compatible and with the theory of quantum chemistry, but their physical experimental verification depends on advanced techniques for their synthesis, obtaining laboratory for experimental biochemical.
The highest for Raman scattering activities peaks are in the frequency 3,348cm-1 with 7.107609729Å4/amu and 2,163cm-1 with 8.902805583Å4/amu, for CC-pVTZ and 6-311G** (3df, 3pd), respectively. The Raman spectrum was calculated, indicating the characteristic of the bioinioganic nano-molecule genesis. Now the challenge is to build the basic structure of the bio-inorganic membrane. From the unimaginable, going where our mind can take us and build a new DNA, that nanomolecule. Characterized its infrared spectrum and Raman. Quantically calculated, accepted by quantum chemistry parameters, with ab initio methods, in the bases CC-pVTZ and 6-311G ** (3df, 3pd). An experimental challenge to chemists. The new bio-inorganic nano-molecule C13H20BeLi2SeSi / C13H19BeLi2SeSi, is well characterize computationally. As its scientific name 3-lithio-3-(6-{3-selena-8-beryllatricyclo[3.2.1.0²,⁴]oct-6-en-2-yl}hexyl)-1-sila-2-lithacyclopropane, getting Kurumi name, which means boy in Tupi-Guarani language, which are indigenous inhabitants of southern Brazil. As the bio-inorganic nano-molecule Kurumi is the basis for a new creation of a biomembrane, later calculations that challenge the current concepts of biomembrane should advance to such a purpose.
Prof. Dr. Lauro Figueroa Valverde, University Autonomous of Campeche (Faculty of Chemical-Biological Sciences), Calle Avenida Agustín Melgar s/n, Buenavista, 24039 Campeche, Camp., Mexico, for their contribution.
The author declares that there is no conflicts of interest.

©2019 Gobato, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.