Three key extraction-constants,
,
and were determined at 298K with an approximate method. Here
,
and were defined as
,
and
, respectively: L shows 18-crown-6 ether, A− does picrate ion, and the subscript “org” denotes an organic phase. Diluents employed as the org phases were o-dichlorobenzene, bromobenzene, dibutylether, and nitrobenzene. In order to determine briefly these key constants, the following approximate equations were used:
, and
, where
refers to an experimental distribution ratio of Cd(II) between water and the org phases. The former two constants were compared with the corresponding values previously-determined. By comparing other many values, validity of the method was also examined and it was consequently clarified.
Keywords: extraction constants, distribution ratio, ion-pair formation constants, primary diagnosis, distribution constant of picrate ion,cadmium picrate, 18-crown-6 ether
Simplification of the convoluted procedures3,4,10,12-17 for the divalent-metal, M(II), extraction systems can stimulate an estimate of L functions by many workers. The overall M(II) extraction system with a univalent pairing anion (A−) can be expressed as the following three extraction equilibria:
, (1)
,(2)
And
. (3)
The expressions of these equilibria as equilibrium constants are
, (1a)
, (2a)
And
. 3,15 (3a)
When one defines conditional distribution ratios,
,
, and
as
,
, and
respectively, Eqs. (1a), (2a), and (3a) become
, (1b)
, (2b)
And
. (3b)
Moreover, expressing as
, Eqs. (2b) and (3b) become
(2C),
(3c) respectively. Here, although it was a rough handling, we assumed that the D0,
,
values equal the experimental distribution ratio (D).
So, these assumptions were examined experimentally by comparison in
,
,and
between the procedures. The
values can be directly compared with each other. Thevalues can be also compared with those calculated from the relation
.15,17Furthermore, the
values calculated from
can be compared with those done from
.15-17 Here, the symbols K with the asterisks denote the equilibrium constants determined with the other procedure reported in the previous papers15,17 anddenotes a distribution constant,
of singleion into the org phase. In this report, the author calls this procedure15,17 reported before “the formal procedure”. In all calculations, the data17 reported before were re-used.
In the previous paper,17 the plot of
for
and
has given the straight line with the slope (a) of 0.81 and an intercept (b) of 3.90, the value corresponding to log
(circle in Figure 1). Here
,
and [Cd2+]have been calculated in terms of a successive approximation.10,15-17 The same is true of the extraction systems with the other diluents. The slope less than unity shows the dissociation of
in the
phase.17

Figure 1 The plots (1B) (circle), (2C) (square), and (3C) (triangle) for the BBz system with L = 18C6.
The plot of log
yielded a straight line with a=0.93 and b=1.17 , the b value to
(Figure 1). This a value shows the extraction of
into the
phase. The plot of log
gave a straight line with
and
, the value to
(Figure 1). This a value shows the extraction ofinto thephase. From these results, one can easily see the co-extraction of
,
and
into the
phase. Here, we call the
(x-axis) plots of
,
and logD the plots (1B), (2C), and (3C), respectively. Similar data calculated for other systems are summarized in Table 1. The plots (1B) clearly indicate the dissociation of
in the oDCBz and NB phases.
|
Diluent
|
Plot (1B)1)
|
Plot (2C)
|
Plot (3C)
|
|
A2)
|
b2,3)
|
a2)
|
b2,4)
|
a2)
|
b2,5)
|
|
oDCBz
|
0.76
|
3.39
|
1.02
|
2.01
|
0.94
|
−0.51
|
|
DBE
|
1.02
|
4.61
|
0.96
|
0.78
|
0.99
|
−1.53
|
|
NB
|
0.55
|
4.80
|
0.99
|
4.92
|
1.05
|
3.23
|
Table 1 Data of the plots for composition determination of extracted species in the CdPic2 extraction with 18C6 at 298 K
In discussing the data obtained from the above plots, it is important to examine the overlap of the [L]org or ionic strength (I) values used for the data analyses; especially, the (Iorg) values of the org phases are important for the latter case. The larger overlaps among the data make comparisons among them possible. Figure 2 shows the overlap of IBBz among the plots for the extraction system with
. On the other hand, Figure 3 shows an example of a minimum
overlap in the report. The BBz system indicates the better overlap, while the NB one does the less one. The latter suggests the larger deviation of the data determined with the present approximate procedure from the data with “the formal one”. A degree of the Iorg overlap increased with the order,
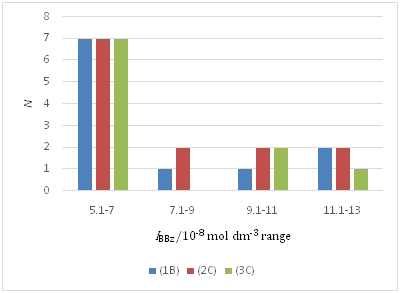
Figure 2 A distribution in IBBz among the plots, (1B), (2C), and (3C) for the BBz extraction system. The symbol N refers to the number of data.
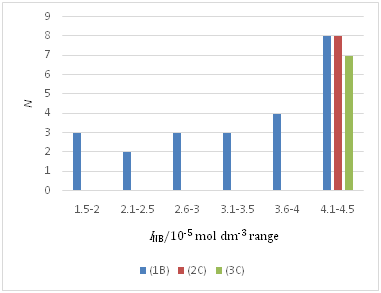
Figure 3 A distribution in INB among the plots, (1B), (2C), and (3C) for the NB extraction system; see Figure 2 for N. This is the case of the minimum overlap in the report.
Table 2 lists the data of mainly logKex and logKex+ for comparison. The both values agreed with each other within experimental errors, except for the NB systems. Also, the values for the oDCBz system were close to those17 reported before. Considering the order, the both values even for the NB system agreed with each other. These facts indicate that the procedure proposed here, the approximate one, is effective for the determination of such extraction constants. The approximation procedure can be used for the primary diagnosis of the system at least.
|
Diluent
|
log Kex
|
log Kex+
|
log KCd/CdL
|
|
This Report
|
Ref. [17]1)
|
This Report
|
Calculated1,2)
|
This Report
|
|
oDCBz
|
4.44 ± 0.46
|
4.31, 4.21
|
1.98 ± 0.22
|
2.46 ± 0.28
|
−0.25 ± 0.20
|
|
BBz
|
4.26 ± 0.45
|
4.38
|
1.70 ± 0.57
|
1.97 ± 0.42
|
−1.11 ± 0.39
|
|
DBE
|
3.81 ± 0.53
|
4.2
|
1.13 ± 0.34
|
1.81 ± 0.53
|
−1.33 ± 0.32
|
|
NB
|
6.63 ± 0.40
|
6.14
|
4.96 ± 0.04
|
4.61 ± 0.14
|
2.95 ± 0.03
|
Table 2 Fundamental data for comparisons between the both procedures
1)Values determined with the formal procedure. 2) Values calculated from the data in ref. 17 by using log Kex+ = log {*Kex+/(*KD,Pic)}
The orders in Kex , Kex+ , and KCd/CdL were DBE≤BBz≤oDCBz<NB , except for the Kex order in the reference.17 Such orders were also observed in the plots, (2C) and (3C), with the a values of about unity (Table 1). A plot of log (approximate Kex , Kex+ , or K2,org ) versus log (formal *Kex , *Kex or *K2,org ) yielded a straight line of a=1.16 and b=−0.74 at R=0.995 and N=13
(Figure 4). Similarly, these facts indicate that the results of the approximate procedure well reflect those of the formal one.
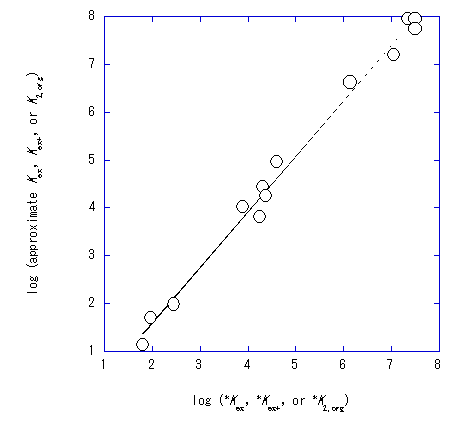
Figure 4 A plot of log (approximate Kex, Kex+, or K2,org) vs. log (formal *Kex, *Kex+, or *K2,org)17 for the CdPic2 extraction with 18C6.
From the values in Table 2, the following constants were obtainable:
,
15-17,
, and
.14 These constants K1,org , K2,org , and
, are defined as
,
, and
, respectively. Their logarithmic values are summarized in Table 3. The K1,org values were larger than or equal to the K2,org ones. These facts suggest that a coordination structure around Cd(II) does not almost change in the two-step reactions with Pic−.18 Also, the K2,org values thus-calculated were the same as those done from the formal procedures within experimental errors. As similar to the results in Table 2, these facts show the validity of the present procedure.
|
Diluent (log *KD,Pic)2)
|
log K1,org
|
log K2,org )2)
|
log KD,CdL
|
log Kex2±
|
|
oDCBz (−5.5, −4.2)
|
8.20 ± 0.37
|
7.95 ± 0.53 (7.3, 7.5)
|
−1.33
|
−11.23 ± 0.28
|
|
BBz (−4.6)
|
7.72 ± 0.61
|
7.20 ± 0.75 (7.0)
|
−2.18
|
−10.39 ± 0.49
|
|
DBE (−5.1)
|
8.21 ± 0.75
|
7.75 ± 0.76 (7.5)
|
−4.05
|
−11.48 ± 0.68
|
|
NB (−2.35)
|
4.02 ± 0.15
|
4.02 ± 0.40 (3.9)
|
2.00
|
−1.76 ± 0.07
|
Table 3 Some equilibrium constants estimated from Kex, Kex+, and KCd/CdL for the CdPic2 extraction with 18C6 at 298K1)
1)See the text. 2) Values reported in ref.17
Figure 2& 3 show the degrees of the overlap of the IBBz and INB data used for calculation. These degrees, together with those of the other two systems, rationally make comparisons between or among the data, such as Kex , Kex+ , and K1,org , possible, although the finding of the NB system may create dissatisfaction.
Simplification of the convoluted procedures3,4,10,12-17 for the M(II) extraction systems, compared with the procedures1-3,5,6,8,9,11 for the monovalent-metal extraction ones, must stimulate an estimate of L functions by many workers. Unfortunately, there are still complicated treatments for the evaluation of equilibrium concentrations, such as
,
and
, by the successive approximation,17 before the use of the present procedure. However, the procedure proposed here can be useful for the workers who expect a primary diagnostic determination of some extraction constants, because the procedure uses directly the experimental D values. Besides, such constants derive valuable component equilibrium-constants from several thermodynamic cycles, as shown in Case Presentation. Additionally, Figure 2 & 3 suggest an importance of the ionic strength for the phases in the determination of the equilibrium constants by the extraction experiments.1,6,8-11,13






