Open Access Journal of
eISSN: 2576-4578


Case Series Volume 2 Issue 4
Department of Oral Medicine and Radiology, Bapuji Dental College and Hospital, India
Correspondence: Kanad Chaudhuri, Department of Oral Medicine and Radiology, Bapuji Dental College and Hospital, Davangere, Karnataka, 577004, India, Tel +919880635794
Received: January 24, 2018 | Published: November 8, 2018
Citation: Krishnankutty K, Chaudhuri K, Ashok L. Erythema multiforme: a case series and review of literature. Open Access J Trans Med Res. 2018;2(4):124-130. DOI: 10.15406/oajtmr.2018.02.00052
Erythema Multiforme (EM) is an acute, immune-mediated condition, most commonly induced by Herpes Simplex Virus (HSV) infection, or by the use of certain medication and presents with cutaneous or mucosal lesions or both. Characteristic lesions are seen over the skin as distinctive target-like lesions with concentric color variation, sometimes accompanied by oral, genital, or ocular mucosal erosions or bullae. EM is usually self-limiting; however, frequent episodes over the years can lead to recurrent disease in a small group of patients. Patients can present with oral and lip lesions typical of EM without any evidence of skin lesions. It has also been reported that primary EM can be confined to the oral mucosa but the subsequent attacks can produce more severe forms of cutaneous EM. We present 3 case scenarios of EM minor managed in our department along with a review of the pathogenesis, clinical features, and management of EM.
Keywords: erythema multiforme, herpes simplex virus, stevens johnson syndrome
The term erythema multiforme was first coined by Ferdinand von Hebra in 1860. Erythema Multiforme (EM) is an acute mucocutaneous condition caused by a hypersensitivity reaction. Despite being listed with many etiological factors, the exact pathogenic mechanism of EM remains unclear, and as a consequence, there are no evidence-based, reliably effective therapies.1 EM can be triggered by a range of factors, but the best-reported stimulus is with a preceding viral infection with herpes simplex virus (HSV) and other cases are most often triggered by drugs.2 EM has been classified into a number of variants, mainly minor and major forms, as it may involve the mouth alone, or present as a skin eruption with or without oral or other lesions on the mucous membrane.2
Case 1: A 30-year-old male patient reported to our department with the complaint of a burning sensation and ulceration in the mouth for 10 days. History revealed that the patient had undergone oral prophylaxis at a private dental clinic 15 days back. Five days later he suffered from fever and sore throat, followed by a blister formation in the mouth and lips. The blisters subsequently burst and formed large ulcers in the mouth and lips. The burning sensation was severe in intensity and aggravated on taking hot and spicy food. The patient had been prescribed topical analgesic medication, cetirizine and multi-vitamin capsules by a private medical practitioner. As the patient didn’t get any relief, he was referred to our department for the same. The patient didn’t have a significant medical history. On examination, bilateral submandibular lymph nodes were palpable, enlarged and tender. Extra-orally, lips were swollen with bloody encrustations Figure 1. Multiple, diffuse erosive areas covered with grayish necrotic slough were seen on the upper and lower labial mucosae, left and right buccal mucosae and soft palate Figure 1. The areas bled on slight manipulation. A provisional diagnosis of EM minor was made. Routine hematological investigations were normal. The patient was started on systemic steroid therapy. Oral Prednisolone 30mg/day in divided doses was given which was tapered over 9 days. Benzydamine hydrochloride and chlorhexidine gluconate mouthwash were given for symptomatic relief. The patient was evaluated after a week and showed complete resolution of the intra-oral erosive areas Figure 2. The patient was advised to apply petroleum jelly to the lips to prevent dehydration and cracking of the encrustations.

Figure 1 Pretreatment photographs showing bloody encrustations on the lips, diffuse erosions covered with necrotic slough on the labial mucosae, buccal mucosae and soft palate.
Case 2: A 25-year-old male patients was referred to our department by a private medical practitioner for an opinion regarding non- healing intraoral ulcers for 3 weeks. History revealed that the patient had similar bouts of ulceration in the mouth 5 times in the past 1 year. The ulcers were associated with fever and burning sensation in the mouth, which aggravated on eating hot and spicy food. The ulcers healed in 10-15 days without any therapy. Presently, the ulcers persisted for 3 weeks due to which the patient visited a private medical practitioner. He was prescribed antibiotics, analgesics and multivitamins for the same. As he didn’t get any relief, he was referred to our department for further management. Presently, the patient gave a history of fever and sore throat 25 days back, followed by a burning sensation in the mouth and lips for 3 weeks. On examination, bilateral submandibular lymph nodes were palpable and tender. Extra-orally, the lips showed brownish encrustations which bled easily on stretching the mucosa. Intraorally, diffuse erythema was seen on the upper and lower labial mucosae, diffuse erosive areas on the ventral and lateral surfaces of the tongue Figure 3. A clinical diagnosis of recurrent erythema multiforme minor was made. Laboratory investigations revealed a raised serum IgG for Herpes Simplex Virus 1. Hematological investigations were within normal limits. A final diagnosis of HSV induced recurrent erythema multiforme was made. The patient was started oral Prednisolone therapy 30 mg/day in divided doses tapered over 9 days. Topical triamcinolone acetonide 0.1% was given for application on the lips. Benzydamine hydrochloride and chlorhexidine gluconate mouthwash were given for symptomatic relief. The patient was evaluated after a week and showed a complete resolution of the lesions Figure 4. The patient was educated regarding the recurrent nature of the disease and was advised to consult an oral physician at the earliest when the first prodromal symptoms appear.
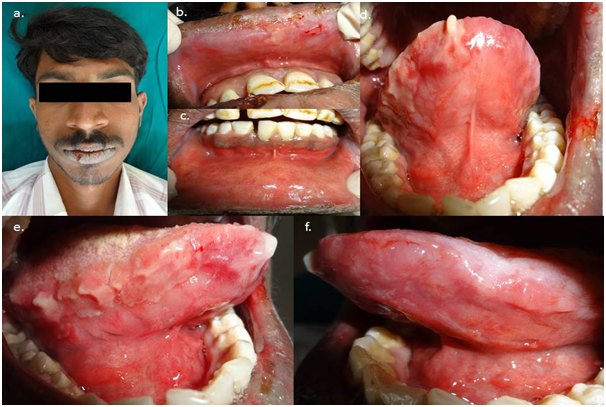
Figure 3 Pretreatment photographs showing bleeding encrustations on the lips, diffuse erosions on the labial mucosae and ventral and lateral borders of the tongue.
Case 3: A 40-year-old male patient reported to the department of oral medicine with the chief complaint of a burning sensation in the oral cavity since 2 days, which was severe in intensity, continuous and was associated with moderate pain on having food and opening the mouth. There was also associated bleeding from the lips. The patient gave a history of a headache 3 days back and self-medication with diclofenac sodium 50mg for the same. Next day he noticed swelling and blister formation on his lip, which later ulcerated and was bleeding. He consulted a physician regarding the same complaint and was given anti-allergic drugs. But later he developed more widespread ulcerations in his mouth with severe burning sensation and inability to eat. The patient did not have any positive medical history and was not on any other medication. On examination, there was a bulla present on his left palm Figure 5. Bilateral submandibular lymph nodes were palpable and tender. Intraoral examination revealed oedematous lips with bloody encrustations & irregular shaped superficial ulcerations & erosions with fresh bleeding spots. Multiple, diffuse, irregular superficial ulcerations were seen on the right side buccal mucosa, upper & lower labial mucosa and dorsal, lateral & ventral surfaces of the anterior 2/3rd of the tongue. The ulcers were covered with yellowish slough and surrounded by diffuse erythema Figure 5. The patient had hypersalivation and halitosis. Other dermal and mucosal surfaces showed no abnormality. Routine hematological investigations were within the normal limits. Clinically, a diagnosis of drug-induced erythema multiforme minor was made. Oral Prednisolone 30mg/ day was started along with topical triamcinolone acetonide 0.1% for local application, in addition, mouthwashes consisting of local anesthetics and antiseptics were added for symptomatic treatment. 1st recall on 5th day revealed almost complete healing of the oral lesions. Prednisolone was later withdrawn after tapering for the next 5 days & there was complete healing of the lesions Figure 6.
Erythema multiforme (EM) is an acute mucocutaneous hypersensitivity reaction characterized by a skin eruption, with or without oral or other mucous membrane lesions. Occasionally EM may involve the mouth alone. EM has been classified into different variants based on the degree of mucosal involvement, the nature of the disease and the distribution of the skin lesions.3,4 Historically, it is often classified based on the wide spectrum of severity ranging from a mild form or EM minor to a more severe form or EM major, traditionally known as Stevens-Johnson syndrome (SJS), to the most severe form toxic epidermal necrolysis (TEN; Lyell’s disease).3,5 However, SJS and TEN are now considered to be distinct etiological and pathological entities.2,4 Presently, EM spectra have been classified mainly as minor and major forms; EM minor typically affects only one mucosa, generally the oral mucosa and may be associated with symmetrical target skin lesions on the extremities. EM major typically involves two or more mucous membranes with more variable skin involvement.2 EM is considered relatively common, but its true incidence is unknown because cases requiring hospitalization have only been reported. The incidence of EM is postulated to be far less than 1%, but possibly greater than 0.01%. It occurs predominantly in young adults, with a slight female preponderance and without racial predilection.6,7 In EM, hypersensitivity reaction occurs primarily to antigens that are induced by exposure to microbes or drugs7,8; other reported factors include malignancy, autoimmune disease, radiation, immunization, pregnancy, menstruation and food additives or chemicals. 1,7
Of these factors, infection represents as a triggering agent in approximately 90% of cases, and the most common infectious agent responsible are viruses especially herpes simplex virus (HSV). HSV type 1 is the most commonly associated cause, but HSV type 2 can also induce EM.4,7,9 Other viruses like Epstein Barr virus, adenoviruses, enteroviruses (Coxsackievirus B5, echoviruses), hepatitis viruses (A, B and C), HIV, influenza, Para vaccinia, parvovirus B19, poliomyelitis, vaccinia, and variola have all been implicated1,4Other infectious agents less commonly associated with EM include bacteria, such as Mycoplasma pneumonia, Chlamydia, Corynebacterium diphtheria, hemolytic streptococci, Legionella, Borrelia, Neisseria meningitides, Mycobacterium avium complex, Pneumococcus, Proteus, Pseudomonas, Rickettsia, Salmonella, Staphylococcus, Vibrio parahaemolyticus, Yersinia, Chlamydia, Lymphogranuloma venereum, and psittacosis. Fungal infections include coccidioidomycosis, dermatophytes or histoplasmosis, sporotrichosis and parasites such as Trichomonas and Toxoplasma gondii.1,4 Recurrent erythema multiforme and the rare persistent erythema multiforme are two entities identified within the clinical spectrum of EM. Recurrent EM is a condition of substantial morbidity, it is the frequent occurrence of EM over a period of years; Patients show an average of six episodes per year and mean disease duration of 6–10 years.10,11 Recurrent EM is usually reported to be associated with HSV infections however many heterogeneous stimuli such as repeated M. Pneumonia infections, hepatitis C, vulvovaginal candidiasis, menstruation, complex aphthosis, and a high intake of food preservative have also been reported.7,10,11
Persistent EM is a rare variant defined as the continuous occurrence of typical and atypical lesions without interruption.12 Association of Persistent EM with viral infections, like HSV, Epstein–Barr virus, hepatitis C virus, influenza virus, and cytomegalovirus, inflammatory bowel disease and various neoplasms have been reported.13,14 Drug-associated EM is reported in less than 10% of cases and common medications that induce EM are nonsteroidal anti-inflammatory drugs such as diclofenac, ibuprofen, and salicylates, sulfa drugs -sulfonamides, co-trimoxazole, anti-consultants such as phenytoin and carbamazepine.7,15 Unlike EM, drugs precipitate 80% to 95% of the cases of TEN and more than 50% of cases of SJS.16 Drugs that are reported to be associated with EM are summarized in Table 1.15 Genetic susceptibility can be a predisposing factor in some patients with EM, especially in patients with HSV-associated EM. Increased disease susceptibility with HLA-B15 (B62), HLA-B35, HLA-A33, HLA-DR53, HLA DQ3 and HLADQB1*0301 alleles have been reported in recurrent EM.4,7,17 HLA DQ3 is also a useful marker in distinguishing herpes-associated EM from other diseases with EM-like lesions & HLA allele DQB1*0402 is a useful marker for predicting extensive mucosal involvement.4
Drugs associated with EM |
|
|
|
|
|
|
|
|
|
|
|
Analgesics (acetylsalicylic acid, codeine, diclofenac, phenylbutazone, piroxicam, tenoxicam) |
|||||||||||
Anticonvulsants (carbamazepine, hydantoin, phenytoin) |
|||||||||||
Antifungals (griseofulvin, fluconazole) |
|||||||||||
Antihypertensives (amlodipine, digitalis, diltiazem, nifedipine, verapamil) |
|||||||||||
Antimicrobials (clindamycin, chloramphenicol, ethambutol, penicillin derivatives, rifampicin, streptomycin, tetracyclines, vancomycin), cotrimoxazole |
|||||||||||
Diuretics (furosemide, hydrochlorothiazide, indapamide) |
|||||||||||
Hormones (minoxidil, mesterolone, progesterone) |
|||||||||||
Vaccines (Measles/mumps/rubella vaccines) |
|||||||||||
Others (Atropine, Allopurinol, busulfan, fluorouracil, Omeprazole, Retinol, Theophylline, Zidovudine) |
|
|
|
||||||||
Table 1 Drugs associated with EM
EM seems to be a T- cell-mediated immune reaction to precipitating stimuli, leading to a cytotoxic immunological attack on non-self-antigen expressing keratinocytes followed by sub-epithelial and intra-epithelial vesicle formation, eventually leading to widespread blistering and erosion.2 The mechanism of tissue damage in EM seems to vary in virus-induced EM and drug-induced EM and also differ from those in SJS and TEN.4 In HSV associated EM, HSV–DNA fragments in the skin or mucosa trigger the disease. Virus fragments get transported to the epithelium by CD34+ cells, and T cells accumulate in response to HSV antigens and damaged cells.4 CD4+ T helper 1 (Th1) lymphocytes, produce IFN-γ, characteristic of a delayed type hypersensitivity reaction. IFN-γ is a pro-inflammatory mediator which induces adhesion molecule expression on keratinocytes and endothelial cells. It also stimulates the production of chemokines and cytokines, which amplify further immune response by stimulating the production of more cytokines and chemokines, aiding in the recruitment of additional reactive T cells to the area. These cytotoxic T cells, NK cells or chemokines can all induce epithelial damage.4 unlike virus-induced EM, mechanism of damage in drug-induced EM does not appear to be a result of the delayed type of hypersensitivity. In drug-induced EM, reactive metabolites of the particular drug induce the disease and keratinocyte apoptosis is induced by tumor necrosis factor alpha (TNF-α) released from keratinocytes, macrophages, and monocytes leading to tissue damage.2,4
In TEN and SJS, the Fas/FasL apoptotic pathway is thought to be implicated in tissue damage. Fas is present on keratinocytes and FasL is found on activated T cells and NK cells and thus binding of keratinocytes to T cells or NK cells can induce apoptosis by the formation of caspases.2,4,18,19,20 However, there is also some evidence that shows the direct activation of cytotoxic T cells, leading to keratinocyte cell death in TEN, in the presence of the precipitating drug. The apoptotic mechanism seems to be mediated by perforin/granzyme.4,19,20 Auto antibodies against desmoplakins I & II and anti-epidermal auto antibodies have been reported in a subset of EM patients. However, despite a cellular immune response, humoral immune mechanisms may be implicated in the pathogenesis of the EM-like disease.21,22 EM can present clinically with varying range of severity, from mild, exanthematous, self-limiting condition to a severe, widespread and life-threatening illness.2 Currently, EM, SJS, and TEN are not considered to be variants of the same immune disorder, but SJS and TEN are probably the variants, of a single disease spectrum differing only in the area of involvement and the severity of systemic findings.16, 20
Dermal lesions are usually multiple, seen in a fixed position with symmetrical distribution. All lesions appear approximately within 3 days of onset. Hundreds of lesions may be seen, but involving less than 10% of the body surface area. Initially, the lesions are seen acrally and might appear at sites of trauma or physical irritation or at sites of sun exposure. Prodromal symptoms are rare and are nonspecific & mild (a cough, rhinitis, low-grade fever, malaise, diarrhea, myalgia, and arthritis). Most often the lesions are asymptomatic, occasionally itching or burning sensation may accompany.16
Skin lesions have been described as following types2,4:
Around 70% of EM patients have oral lesions and they may precede lesions on other stratified squamous epithelia and at times may be the only manifestation of the disease process.23 Intraoral lesions are characteristically seen on non-keratinized mucosae and affect the anterior part of the oral cavity. Lip involvement is almost universal and alveolar mucosae & palate, are other common sites. Lips become swollen and cracked, superficially ulcerated, and show diagnostically distinctive bloody encrustations. Target or iris lesions may be seen on the lips, but rarely on oral mucosa. Diffuse, multiple macules and blisters or bullae form on the oral mucosa and rupture to form superficial ulceration and pseudomembrane.2,4,8,16 Unlike the cutaneous lesions, oral lesions are often symptomatic and may compromise speech and mastication.16 Other mucosal sites such as ocular, nasal, pharyngeal, laryngeal, lower respiratory, and anogenital mucosa may be involved.1‒8 Ocular involvement results in excessive lacrimation and photophobia. Genital lesions are painful and can lead to retention of urine.2‒4 Scarring sequelae from ocular and pharyngeal involvement cause morbidity.1‒8 EM is a self-limiting disease, rarely recurrent or persistent, and lesions typically appear over 3–5 days and resolve in 1–2 weeks.7 Transient hypo or hyper pigmentation may be seen at skin lesion sites and oral lesions heal without scarring.11‒16
Diagnosis is almost always made based on the history and clinical presentation and there are no specific diagnostic tests for EM.7,9 If necessary, a biopsy of perilesional tissue, with histological and immunostaining examination, is performed for a specific diagnosis.1,2,4,7 Certain laboratory abnormalities like increased erythrocyte sedimentation rate, white blood cell count, and liver enzyme levels, can be seen in cases of severe EM.1,7,9 Intra-lesional HSV-DNA detection using polymerase chain reaction, and Immunohistochemistry for IFN-γ and TNF-α, may be useful tests to differentiate herpes-associated EM from drug-associated EM.24 No specific treatment exists for the disease itself, but as a primary goal of management, the suspected precipitating agent should be discontinued or treated as indicated. Mild forms are managed symptomatically but in severe cases, hospitalization and supportive care with intravenous fluids are often necessary.2 Mouthwashes containing a local anesthetic and mild antiseptic compound usually provide relief from painful oral symptoms.1 Herpes associated EM may be managed with antiviral agents, a 5 day course of acyclovir 200mg five times daily may be started when the lesions first appear, or 400mg of acyclovir four times daily for 6 months may be given, or continuous treatment using valacyclovir, 500mg twice a day, is useful for prophylaxis.25 In the case of EM-related to Mycoplasma pneumoniae, Tetracycline 250mg four times a day for at least 1 week may be indicated .1 Corticosteroids are the most widely used drugs in the management of EM. EM minor responds to topical steroids, however a systemic steroid administration may be necessary. EM major or SJS may require hospitalization and administration of systemic corticosteroids (Prednisolone 0.5–1.0mg/kg/ day tapered over 7–10 days).2,4 Immuno modulatory drugs such as azathioprine, cyclophosphamide, Dapsone, cyclosporine, levamisole, and thalidomide or interferon α are also tried alone or in combination or with steroids.11,26,27,28 Malignancy-associated EM is rare, but it has been described in patients with underlying hematologic cancers, such as leukemias and lymphomas. Persistent EM or EM unresponsive to therapy are rarely reported with solid organ cancers, such as gastric adenocarcinoma, renal cell carcinoma, and extra hepatic cholangiocarcinoma.7
None.
The authors declare that there is no conflict of interest.

©2018 Krishnankutty, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.