MOJ
eISSN: 2471-139X


Research Article Volume 7 Issue 3
1Department of Medicinal Chemistry, University of Tripoli, Libya
2Food and Drug Control Centre (LFDA), Libya
3Department of Zoology, Tripoli University, Libya
4Department of Biosciences, University of Salzburg, Austria
Correspondence: Abdul M Gbaj, Associate Professor of Genetics and Biochemistry, Department of Medicinal Chemistry, Faculty of Pharmacy, University of Tripoli, Libya, Tel 00218913556785
Received: May 14, 2020 | Published: May 28, 2020
Citation: Meiqal NH, Sadawe IA, Bensaber SM, et al. Novel ortho- phenylenediamine derivatives as spike glycoprotein coronavirus 2019-nCoV inhibitors: molecular docking study. MOJ Anat Physiol. 2020;7(3):70-74. DOI: 10.15406/mojap.2020.07.00293
A series of new ortho-phenylenediamine derivatives has been designed. The crystal structure of the post fusion core of 2019-nCoV S2 subunit and perfusion 2019-nCoV spike glycoprotein with a single receptor-binding domain was used as target protein for molecular docking of ortho- phenylenediamine derivatives. in addition a protein-ligand interaction analysis was performed using Auto Dock 4.2 software. Based on the docking score and after three-dimensional similarity analysis, NHM7[(10,10'-((1E,1'E)-(1,2-Phenylenebis(azanylylidene)) bis(methanylylidene)) bis(anthracen-9(8aH)-one)] had the highest binding energy. The calculated binding energy of ortho- phenylenediamine indicates effective binding of proposed inhibitors to the fusion core of 2019-nCoV S2 subunit and pre-fusion 2019-nCoV spike glycoprotein with a single receptor-binding domain.
Keywords: severe acute respiratory syndrome, coronavirus, COVID-19 spike glycoprotein inhibitors, chloroquine, hydroxychloroquine, molecular docking
Eruption of the coronavirus disease 2019 (COVID-19) has uncovered serious threat to worldwide public health and economies. Human coronaviruses (CoV) are enveloped positive-stranded RNA viruses which belong to the category of Nidovirales, and are frequently responsible for digestive tract and upper respiratory infections.1,2 During acute respiratory condition coronavirus (SARS-CoV) can cause severe and occasionally deadly respiratory tract infections in humans.3–5 The present pandemic emphasizes the tremendous threat of these viruses.5–7 The entry of coronavirus into host cells is mediated by a transmembrane spike (S) glycoprotein that forms homotrimers the exterior envelop (corona) of the virus.8–10 S consists of two functional subunits responsible for binding to the host cell receptor (S1 subunit) and fusion of the viral and cellular membranes (S2 subunit). For many CoVs, S is cleaved at the boundary between the S1 and S2 subunits, which stay non-covalently bound in pre-fusion conformation.11–14 The distal S1 subunit comprises the receptor-binding domain(s) and contributes to stabilization of the pre-fusion state of the membrane-anchored S2 subunit that contains the fusion machinery.15–17 There are various potential targets against COVID-19 and among these targets replication-related enzymes, such as spike glycoprotein are extremely conserved. It has been reported that drugs that inhibit spike glycoprotein are able preventing proliferation and replication of the virus.18,19
Chloroquine is an extensively used anti-malarial drug with immunomodulatory properties. Chloroquine and hydroxychloroquine were reported to have anti-SARS-CoV activity in vitro.20–22 However, molecular mechanism of action of chloroquine and hydroxychloroquine has not been completely explored. Findings from earlier studies have suggested that chloroquine and hydroxychloroquine may inhibit the coronavirus through a series of steps.20–22 Hydroxychloroquine was found to be more potent than chloroquine to inhibit SARS-CoV-2 in vitro.23,24 Following the spike glycoprotein inhibition approach two supposed standard spike glycoprotein inhibitors were used as lead compound including chloroquine and hydroxychloroquine inhibitors and their chemical structures as shown in Figure 1.
The post-fusion core of 2019-nCoV S2 subunit and pre-fusion 2019-nCoV spike glycoprotein with a single receptor-binding domain may be utilized as a homologous target for screening of ortho- phenylenediamine derivatives that could inhibit the proliferation and replication of COVID-19.25–28 The ortho- phenylenediamine derivatives are Schiff bases recognized for their therapeutic value as they were reported to have anti-inflammatory, analgesic, antiviral, antitumor, antifungal and antibacterial properties.29–34 Molecular modeling is a recognized computational tool to aid early drug discovery and development. It is used to generate ideas of a compounds or macromolecules 3D conformation, protein–ligand interactions, and allows forecasts about biological activities. Furthermore, the integration of molecular modeling in drug or vaccine design can help in early drug or vaccine discoveries.35–37 The main aim of this study was to further identify the spike glycoprotein as a target, and by computational drug repurposing procedures to allocate appropriate inhibitory agents.
Molecular docking
The starting geometry of the ortho-phenylenediamine derivatives was constructed using chem3D Ultra software (version 8.0, Cambridge soft Com., USA). The optimized geometry of ortho- phenylenediamine derivatives with the lowest energy was used for molecular dockings. The crystal structure of pre-fusion 2019-nCoV spike glycoprotein with a single receptor-binding domain (6VSB) was downloaded from the Protein Data Bank https://www.rcsb.org/structure/6vsb and post fusion core of 2019-nCoV S2 subunit (6LXT) was downloaded from the Protein Data Bank https://www.rcsb.org/structure/6lxt. Molecular dockings of ortho-phenylenediamine derivatives with 6VSB and 6LXT was accomplished by Auto Dock 4.2 software from Scripps Research Institute (TSRI) (http://autodock.scripps.edu/). Firstly, polar hydrogen atoms were added into protein molecules. Then, partial atomic charges of the protease enzymes and ortho- phenylenediamine derivatives molecules were calculated using Kollman methods.38 In the process of molecular docking, the grid maps of dimensions: (60Å X 60Å X 60Å) and (36.8Å X 64.6Å X 60Å) for 6VSB and 6LXT, respectively, with a grid-point spacing of 0.376Å and the grid boxes centered were used. The number of genetic algorithm runs and the number of evaluations were set to 100. All other parameters were default settings. Cluster analysis was performed on the basis of docking results by using a root mean square (RMS) tolerance of 2.0Å, dependent on the binding free energy. Lastly, the dominating configuration of the binding complex of ortho- phenylenediamine derivatives and spike glycoprotein fragments with minimum binging energy of binding were determined which relied strongly on the information of 3D-structures of the spike glycoprotein binding site and ultimately generated a series of spike glycoprotein-binding complexes.
Molecular docking analysis
Table 1 shows the binding energies of chloroquine and hydroxychloroquine, (Figure 1, as standards), ortho-phenylenediamine derivatives, and spike glycoprotein (6VSB and 6LXT) obtained by the molecular docking strategy. Molecular dockings of the ortho-phenylenediamine derivatives with spike glycoprotein (6VSB and 6LXT) were performed using Auto Dock 4.2 to obtain information about interaction forces between ortho-phenylenediamine derivatives and spike glycoprotein (6VSB and 6LXT). Ortho-phenylenediamine derivatives and spike glycoprotein (6VSB and 6LXT) were kept as flexible molecules and were docked into seven forms of rigid spike glycoprotein (6VSB and 6LXT) to obtain the preferential binding site of ortho-phenylenediamine derivatives to spike glycoprotein (6VSB and 6LXT). The molecular docking results are shown in Table 1. The modeling studies indicate van der Waals, hydrogen bonding (Table 1) and electrostatic interactions between ortho-phenylenediamine derivatives with spike glycoprotein (6VSB and 6LXT). The contribution of van der Waals and hydrogen bonding interaction is much greater than that of the electrostatic interaction because the sum of van der Waals energy, hydrogen bonding energy and desolvation free energy is larger than the electrostatic energy.39,40 The ortho-phenylenediamine derivatives, and spike glycoprotein (6LXT and 6VSB) interactions are shown in Figure 2 & 3, respectively. Ortho-phenylenediamine derivatives provide higher binding energy (-7.8 to -11.6 kcal/mol) compared to chloroquine and hydroxychloroquine standard spike glycoprotein (6VSB and 6LXT) (-4.7 to -6.0 kcal/mol) Table 1. Figure 2 indicates four hydrogen bonds between NHM7 and 6LXT. In addition, NHM7 showed good docking interaction of -11.6kcal/mol with the 6LXT binding site (Figure 2). Compound NHM7 has the highest binding energy of the series. This compound has an extra phenyl moiety attached to the naphthyl analogue of the phenylenediamine Schiff`s base derivative with a log P value of 7.49 indicating the importance of the lipophilicity for the interaction with the active site. The interaction of similar Schiff's base ortho-phenylenediamine derivatives with the spike glycoprotein binding site is essential for effective inhibition as previously reported.41,42 Therefore, NHM7 (ortho-phenylenediamine derivatives) may be considered the most effective spike glycoprotein inhibitors.
|
Substituent (R) |
Compounds (M Wt) g/mol |
Log P* Calculated |
Hydrogen bonds** |
Binding energy (ΔG) kcal/mol.
|
||
|
donors |
acceptors |
6LXT |
6VSB |
|||
|
Chloroquine
|
319.89g/mol |
4.63 |
1 |
2 |
-5.2 |
-4.7 |
|
Hydroxychloroquine |
335.87g/mol |
3.58 |
2 |
3 |
-6.0 |
-5.6 |
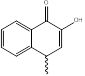 |
NHM1
|
|
2 |
6 |
-9.3 |
-8.2 |
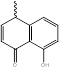 |
NHM2
|
|
2 |
6 |
-10.1 |
8.2 |
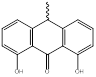 |
NHM3 552.53g/mol
|
|
2 |
8 |
-10.4 |
-8.7 |
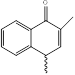 |
NHM4 416.47g/mol
|
|
0 |
4 |
-9.4 |
-7.8 |
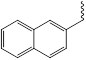 |
NHM5 384.47g/mol
|
7.02 |
0 |
2 |
-8.1 |
-9.3 |
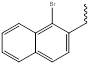 |
542.26g/moL
|
|
0 |
2 |
-8.5 |
-8.7 |
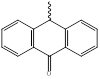 |
488.53g/mol
|
|
0 |
4 |
-11.6 |
-9.0 |
Table 1 Binding energies of ortho-phenylenediamine derivatives, chloroquine and hydroxychloroquine with COVID-19 spike glycoprotein (6VSB and 6LXT) obtained from molecular dockings. The unit of all energies (ΔG) is kcal/mol
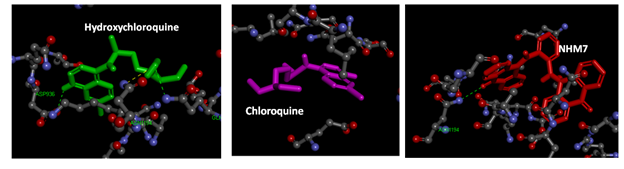
Figure 2 Interaction model between hydroxychloroquine (green), chloroquine (pink) and NHM7 (red) with 2019-nCoV Spike glycoprotein (6LXT) active site. . Hydrogen bonds green broken line.
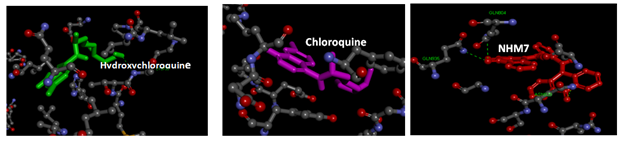
Figure 3 Interaction model between hydroxychloroquine (green), chloroquine (pink) and NHM7 (red) with 2019-nCoV Spike glycoprotein (6VSB) active site. Hydrogen bonds green broken line.
The obtained results using computational drug repurposing is an efficient way to find novel applications for already known drugs.43 Molecular docking and binding free energy calculations for ortho-phenylenediamine derivatives can be used to forecast drug-target interactions and binding affinity. The appearance of resistance to existing antiviral drugs or vaccines is a major challenge in antiviral drug development. The drug repurposing technique allows finding novel antiviral agents within a short period in order to overcome the challenges in antiviral therapy. Computational drug repurposing has previously been used to recognize drug candidates for viral infectious diseases like ZIKA, Ebola, influenza and dengue infections. These methods were also utilized to recognize possible drugs against MERS-CoV and SARS-CoV44,45 and following the COVID-19 outbreak, computational repurposing has been and are used for COVID-19.
Chloroquine and hydroxychloroquine are weak bases recognized to raise the pH of acidic intracellular organelles, such as endosomes/lysosomes, which are necessary for membrane fusion.46 Chloroquine was also found to inhibit SARS-CoV access by altering the glycosylation of angiotensin converting enzyme 2 receptor and spike protein.42,47 It has been also reported that hydroxychloroquine efficiently inhibited the entry step, as well as the post-entry stages of SARS-CoV-2 previously established by chloroquine treatment. Mingo et al.48 suggested that both chloroquine and hydroxychloroquine blocked the transport of SARS-CoV-2 from early endosomes (EEs) or endolysosomes (ELs) that are needed to release the viral genome of SARS-CoV-2.
Owing to the lack of experimental and clinical data, as well as the infectivity of the deadly coronaviruses, we used computational analyses to study the 2019-nCoV virus in terms of protein structures, phylogeny, functions, and its interactions with small chemical molecules. Rapid development and use of a broad-spectrum spike glycoprotein inhibitor might fill the therapeutic gap spanning vaccination, quarantine and hospital setting.
Further elaborative work is necessary for better understanding the mechanisms of spike glycoprotein inhibition. According to our modeling studies ortho-phenylenediamine derivatives may have the potential to inhibit COVID-19 spike glycoprotein making them reasonable candidates for consideration of clinical trials and further examination. Ortho-phenylenediamine derivatives could act at cleavage site in the viral cycle, down regulate pathogenicity and may help in the development of antiviral drugs.
The authors gratefully acknowledge the technical support and valuable suggestions obtained from MS Amira Abdul Gbaj (Novelien Zone, Tripoli, Libya).
The authors declare there are no conflicts of interest.
None.

©2020 Meiqal, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.