Journal of
eISSN: 2373-4426


Review Article Volume 13 Issue 1
1Multiprofessional Health Residency, Hospital de Clínicas de Porto Alegre, Porto Alegre, Brazil
2Graduate Program in Child and Adolescent Health, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil
3Graduate Program in Food, Nutrition and Health, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil
Correspondence: Paula Ruffoni Moreira, Graduate Program in Child and Adolescent Health, Graduate Program in Food, Nutrition and Health, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brazil, Tel (51) 98100-4923
Received: February 25, 2023 | Published: March 20, 2023
Citation: Plucenio LP, Belin CHS, Moreira PR, et al Feeding difficulties in premature children: a systematic review. J Pediatr Neonatal Care. 2023;13(1):33-42. DOI: 10.15406/jpnc.2023.13.00488
Objective: The objective of this systematic review is to discuss the feeding difficulties (FD) in premature infant (PI), the factors involved, and, when applicable, to compare these FD with full-term infants.
Method: Articles were considered eligible in the databases Pubmed, Lilacs, and Scopus, published until March 2020, in Portuguese or English.
Results: 11 studies were included. FD mentioned was food selectivity, food refusal, and vomiting. In studies comparing IP with full-term children, PI showed less impulse to eat, a tendency to have less food variety, less appetite, and less pleasure with food.
Discussion: It is still inconclusive whether there is a pattern of FD related to prematurity and which may be associated with their involved factors in PI. Therefore, we highlighted the importance of clarifying how to promote healthy feeding, observing families that have PI, as well as the need for further studies that can better elucidate the occurrence of FD in PI.
Keywords: premature infant, infant nutritional physiological phenomena, feeding and eating disorders of childhood; feeding behavior, feeding difficulties, systematic review
Premature infant (PI) is those whose birth occurred before completing 37 weeks of gestation.1 Approximately 15 million infants are born preterm annually worldwide, indicating a global preterm birth rate of about 11%.2 Complication resulting from prematurity is the main cause of infant mortality in children under five years old, totaling one million deaths worldwide,3 which shows how much prematurity impacts public health. Premature birth is linked to several factors,4 such as poverty, low education, low maternal age, single mothers, inadequate prenatal care, infection, inflammation, genetic factors,5 low maternal weight gain,6 and smoking.7,8 Prematurity can affect the patient, the family, and the health system,4 resulting in both immediate damages, such as respiratory,9 and ophthalmological problems,10 and in the long term, motor, and developmental outcomes,11,12 learning difficulties,13 and greater risk of developing metabolic diseases.14,15
One of the areas of a PI life that can be affected is nutrition, leading to the emergence of feeding difficulties (FD). According to Schädler et al., from 1995 to 2004,16 of all patients that were sent to a referral center for treatment of eating disorders, 32% were premature. Complementary, meta-analysis study, from 2000 to 2020, found that the prevalence of problematic feeding was 42% in young children born prematurely.17 Rommel et al. found the rate of 35% for PI among children with severe eating problems.17 Some signs and symptoms of FD in children may be lengthening meals and breastfeeding, food refusal for at least one-month, stressful meals, need for distraction to increase intake, and difficulty progressing textures,18 which favor inadequate growth and development.19
Interventions that aim to optimize children’s eating habits can help reduce morbidities and are estimated to be capable of reducing infant mortality in children aged up to five years.20 There is evidence that shows a critical gap for the period of complementary feeding (CF) if it occurs after 10 months of life, the chances of developing FD are increased. Studies suggest that breastfed children have greater acceptance of varied foods than children who received infant milk formula.21–23 This advantage of breastfeeding (BF) over infant milk formula use is that the mother’s diet influences the flavor of her breast milk (BM), unlike the IF which always has the same composition and taste.21,23
Because prematurity is a risk factor for the development of several sequelae, the period of food introduction and CF, which is already important for full-term infants, becomes even more crucial in PI, since the constitution and establishment of healthy eating habits contribute to the promotion of the individual's quality of life. It is essential that more studies explore the subject to clarify the FD found in the population of PI and, thus, to be able to promote more-driven interventions to each of the difficulties and, therefore, provide these patients and their families with a higher quality of life. In this review, we will appraise (1) to identify the feeding difficulties that occur in PI in food introduction and/or CF (2) to identify the factors related to the appearance of FD in premature infants, and, when applicable, (3) to compare them with the factors in full-term infants.
This is a systematic review study that investigated the FD present in food introduction and CF periods of PI. The analysis applied was descriptive and the project was submitted to the International Prospective Register of Ongoing Systematic Reviews (PROSPERO) under registration number CRD42020163471. This study was also marked by the checklist Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), which provides the requirements for the preparation of systematic reviews.
In this review, it was determined that the studies included were observational (cohort, cross-sectional, and case-control design), addressing FD as the outcome, found in the period of introduction food and/or CF of PI and published in Portuguese or English, published until March 2020. Exclusion criteria were not applied.
The definition of FD considered was that of Kerzner et al. (2015) classifying dietary difficulties, in principle, between organic and behavioral difficulties, both of which require investigation and management. In addition, the act of feeding a child involves a caregiver, who will provide the child with food; thus, when there is a chance that a child will have difficulty feeding, the behavior of the caregiver during meals should be considered. Some symptoms presented by the child that may signal the presence of eating difficulties are meals eaten for a very long period, stressful meals, difficulty in progressing the food textures, breastfeeding or bottle feeding too long, among others.24
The search terms were specifically built for each of the databases used: Pubmed, Lilacs, and Scopus considering their specificities and in order not to neglect any article that could meet the inclusion criteria for this work (Table 1). The descriptors used were based on the research question structured according to the population, exposure, comparison, outcome, and study design (PECOS) method (Table 2).
Database |
Search terms |
Lilacs |
Infant Nutritional Physiological Phenomena OR food introduction AND Infant, Premature OR Premature Infant OR Preterm Infant OR Neonatal Prematurity |
PubMed |
Infant Nutritional Physiological Phenomena OR food introduction AND Infant, Premature OR Premature Infant OR Preterm Infant OR Neonatal Prematurity |
Scopus |
Food introduction OR Complementary Feeding OR Supplementary Feedings AND premature AND infant OR preterm AND infant OR Neonatal Prematurity |
Table 1 Search terms used in the databases
Population |
Children aged 6 months to 2 years born prematurely |
Exposure |
Prematurity |
Comparison |
Infants born at term |
Outcome |
Feeding difficulties |
Study design |
Cross-sectional, cohort and case-control studies |
Table 2 Acronym for the population, exposure, comparison, outcome method, and study design (PECOS).
In March 2020, after the research that made use of search strategies, the list of studies found was inserted into an Excel table to select the studies from the determined inclusion criteria. The selection of the studies was carried out by two independent nutrition researchers (L.P.P. and C.H.S.B.), initially by reading the titles, and abstracts and, later, reading the articles in full. The researchers came to a consensus when there was disagreement between their selections. The reference list of the included articles was investigated to identify other studies that could be relevant to this research.
Data from all included studies were extracted into an Excel spreadsheet and were grouped by subject matter. Data extraction fields included authors, title, study design, database, research question or objective, sample method, setting or country, participant characteristics, child’s age and presence of FD, outcome, findings, or results. All findings, outcomes, and measures relating to FD on PI were extracted.
The evidence was synthesized qualitatively by comparing each study to identify overarching themes or key concepts from the findings, similarities, and differences between studies, and factors that may have affected the results.
The quality of the studies was assessed by two different tools (Tables 4 & 5), aiming at adapting the tool to the design of the studies. For cross-sectional studies, quality was assessed using the Agency for Healthcare Research and Quality (AHRQ), which consists of a checklist with 11 questions about the definition of the information source, listing the inclusion and exclusion criteria, masking the evaluators, and confusion control, among others. The cohort studies were evaluated using the Newcastle-Ottawa Scale (NOS), which consists of eight questions regarding the representativeness of the exposed cohort, and the selected source of the unexposed cohort, demonstrating that the outcome of interest was not present at the beginning of the study and if there was enough time for the outcome to occur.
This review found 4,783 studies, of which six were selected for this review. After checking the references for each study initially selected, another five studies were included, totaling 11 articles. A flow diagram is shown in Figure 1, detailing the selection steps.
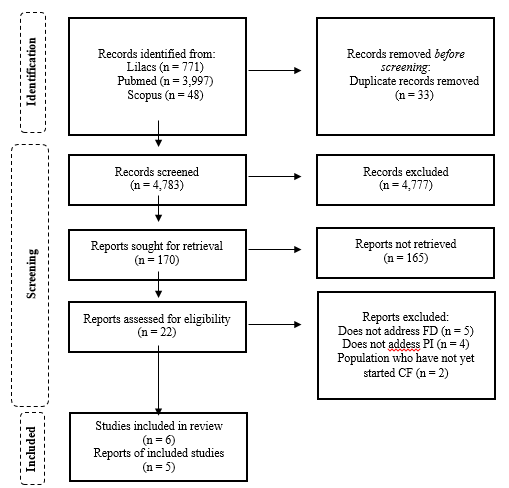
Figure 1 PRISMA Flow diagram of literature search and selection process.
Abbreviations: FD, feeding difficulties; CF, complementary feeding; PI, premature infant
All the studies analyzed approached the FD from the perspective of the children’s parents/caregivers. The main characteristics of the studies in the present review are described in Table 3.
Author(s), year of publication, and country |
Study design |
Objective |
Sample characteristic |
Finding |
Adams-Chapman et al., 2013, United States |
Cohort |
To evaluate the relationship between abnormal feeding patterns and language performance on the Bayley Scales of Infant Development-Third Edition at 18-22 months adjusted age among a cohort of extremely premature infants. |
1,477 PI: average GA (26+2); 50% female; corrected age (CA) (18 months). |
13% had FD. FD was independently associated with days of MV, hearing impairment, and evidence of neuromotor impairment. Children with FD had significantly lower percentiles for length and head circumference when compared to children without FD. |
Brusco, Thaísa R.; Delgado, Susana E.; 2014, Brazil |
Cross-sectional |
To characterize the development of food for a group of premature infants, aged between three and 12 months, verifying the type of breastfeeding, time of introduction of complementary feeding, deleterious oral habits, guidelines received, feeding difficulties, and sociodemographic profile. |
32 PI: average GA (31.3 weeks); 65.6% male, average CA (1 month and 27 days). |
A significant association was found between “crying because the child does not want to eat” and extreme prematurity and extremely low birth weight (BW). The FD found nausea or vomiting during or after feeding (40.6%), choking or coughing during or after feeding (25%), crying because the child does not want to eat (12.5%), and taking too long to eat (6.3 %). |
Cerro et al., 2002, Australia |
Cohort |
To investigate the parents' perception of the feeding behavior of premature children. |
95 PI or those weighing less than 1.5 kg at birth (53% female); average GA (29.2 weeks); average CA (31 months). Parallel study with CBAT: GA of at least 37 weeks (50.3% male); average age (97.4 weeks). |
FI occurred earlier in PI than in term. Among PI, 19% refuse new foods and 21% refuse food at least once a day. |
Chung et al., 2014, United States |
Cross-sectional |
To examine the application of the adjusted GA in the growth curves and the assessment of developmental readiness during the recommendation of solid foods in PT children with a chronological age of 3.5 to 24 months (2 to 23 months of CA). |
76 PI: average GA (30.3 weeks); 54% female; average CA (11.3 months). |
Children considered “not ready” demonstrated significantly more avoidant eating behaviors than those “ready”: pushing food, gagging, keeping food in their mouths, and crying. |
DeMauro et al., 2011, United States |
Cohort |
To compare the incidence of postdischarge feeding dysfunction and hospital/subspecialty visits for feeding problems during the first year of life in late and early-preterm infants. |
319 PI: GA (between 25 to 33+6 weeks); 53% male. |
Parents of early PI reported higher rates of oromotor dysfunction and greater discomfort in their children when feeding. Mothers of late PI reported greater anxiety regarding the feeding of their children. The study concluded that FD rates between the two groups were similar. |
Johnson et al., 2016, England |
Cohort |
To assess the prevalence of eating difficulties in infants born late and moderately preterm infant (LMPT) at 2 y corrected age and to explore the impact of neonatal and neurodevelopmental factors. |
628 PI: GA (between 32 to 36+6 weeks); 53.8% male. (corrected, in the case of PI) of the 2-year survey. |
14.9% of LMPT and 9.5% of full-term had FD. LMPT had a significantly increased risk only for refusal/picky eating and oral motor problems. Probe use for more than two weeks, behavioral problems, and delayed social competence were identified as independent risk factors for FD at 2 years of CA. It was considered that difficulties were mediated by neurodevelopmental sequelae. |
Jonsson; Van Doorn; Van Den Berg, 2013, Sweden |
Cohort |
To describe parents’ perceptions of developmental eating patterns and occurrence of eating difficulties in a group of preterm infants, compared with a control group. |
27 PI: GA (from 28 to 33 weeks); 55.6% male; the average age at the time of the research (36.6 months); time of the survey (was 36.4 months). |
All parents of PI who used a probe for more than 8 weeks reported eating problems at NU discharge. FI occurred significantly earlier among PI than in the control group. Seven percent of PI remained with FD at 36 months (none of the control group had FD at that time). |
Kmita et al., 2011, Poland |
Cohort |
The study was aimed at exploring feeding behavior problems reported by parents of premature infants during the first 12 months of corrected age. |
22 PI: GA (up to 29 weeks); 50% female; |
Early PI had more FD than late preterm infants and FD is more associated with gastrointestinal or neurological issues. Qualitative analysis identified six categories of FD, of which the most associated with the FI period were difficulties related to the introduction of new foods, textures, flavors, and refusal to try to eat independently. |
Kirk et al., 2017, Rwanda |
Cross-sectional |
To assess the health, nutrition, and development of children born preterm/low birth weight who were discharged from a district hospital neonatal care unit in rural Rwanda. |
86 children: GA (less than 37 weeks or weighing less than 2 kg); average GA (33 weeks); the average age at the time of the survey (22.5 months); 51.2% female. |
46.5% had FD; low scores on the development questionnaire were associated with low weight at birth or SGA, symptoms of anemia, presence of FD, and chronic malnutrition. |
Menezes; Steinberg; Nóbrega, 2018, Brazil |
Cross-sectional |
To investigate if there is difficulty in introducing complementary feeding in premature infants. |
38 PI: 60.5% female; CA aged 6 to 24 months at the time of the survey. |
73.7% of parents reported FD. The IF use demonstrated an increase of 41% of the chance of the children having FD compared to those who received EBF or MF. FD found: refusal to open the mouth, food selectivity, food refusal, arching back, difficulty chewing and swallowing, increased time between meals, nausea, refusal of solid foods, and crying. |
Migraine et al., 2013, France |
Cohort |
We assessed the influence of neonatal and maternal characteristics on eating behaviors at 2 y of age. |
234 children: GA (up to 33 weeks); 52.1% male; 245 CBAT; 53.5% male; age (corrected, in the case of PI) at the time of the 2-year survey. |
PI showed less impulse to eat, a tendency to have less food repertoire, less appetite, and less pleasure with food than those born at term. The impulse to eat increases significantly as the Z score of BW decreases in PI. Female gender and BW with a Z score less than -1 were associated with FD. |
Table 3 Characteristics of the studies included in the systematic review
Abbreviations: BW, birth weight; CA, corrected age; CBAT, children born at term; EBF, exclusively breastfeeding; GA, gestational age; LMPT: late and moderately preterm infant; LW, low weight; MF, mixed feeding; MV, Mechanical Ventilation; NU: neonatal unit; PI, premature infant; SGA, small for gestational age; FD, feeding difficulties
The studies were divided into categories to favor a better data presentation, and each of the studies comprises one of the following categories: (1) Comparative studies between preterm and full-term infants, (2) Studies with PI, and (3) Studies with PI classified into two groups according to their gestational age (GA). Three other categories were used to present other data considered relevant to the study: (4) Breastfeeding in PI, (5) Perception, satisfaction, and management of parents regarding their children’s feeding and (6) Demographic data and socioeconomic factors in prematurity.
Four studies addressed FD in PI compared to full-term infants. Among them, one was carried out with a cross-sectional design and three,25 through cohort studies.26–28
Two of these studies, by Cerro et al.,25 and Jonsson et al.,27 present data about the onset of food introduction and found that PI started it significantly earlier than full-term infants. Jonsson et al. found in the PI group a significantly higher proportion of children with symptoms of vomiting during the transition to the solid stage, compared to the control group. At 36 months, 7% of PI remained with eating problems, while none of the control group had a problem during this period.
Migraine et al.28 observed that PI showed less impulse to eat, a tendency to have less food repertoire, less appetite, and less pleasure with food than full-term infants. As for the frequency of food consumption, Migraine et al.28 observed that PI ate dairy products and potatoes more frequently and consumed fewer fruits, cereals, and prepared dishes than those born at term. Jonsson et al.,27 pointed to lumpy food as the most problematic food consistency among PI, while full-term infants had no clear pattern considered as problematic. As for the supply of new foods, concerning the PI, Cerro et al.,25 pointed out that new foods are generally tasted by 78% of them, while 19% refuse new foods, and 21% refuse food at least once a day.
Johnson et al.,26 identified preterm boys were at increased risk for total FD, refusal, and picky eating compared to boys born at term. Preterm girls were already at increased risk for oral-motor problems compared to those born at term.
Johnson et al.,26 Jonsson et al.,27 and Migraine et al.,28 present results regarding neonatal factors that were associated with FD. The first authors identified the use of probes for more than two weeks, behavioral problems, and delayed social competence as independent risk factors for FD at two years of corrected age (CA). However, after adjusting the analysis, the authors concluded that these difficulties were mediated by neurodevelopmental sequelae and that they would be unlikely to represent a specific functional deficit due to moderate or late premature birth. The second study found that all parents of PI who used a probe for more than eight weeks reported eating problems at neonatal unit discharge, and no patient in the control group made use of probes. Migraine et al.,28 on the other hand, identified that intrauterine growth with a Z score less than -1 was associated with FD in both preterm infants and full-term infants.
Finally, Cerro et al.,25 and Johnson et al.,26 present interesting data regarding the prescription of medications and supplements. The first study identified a higher prescription of medications and nutritional supplements in the group of PI than in the group of full-term infants, while the second identified a higher prescription of ant reflux drugs in PI.
Five studies addressed FD in sample studies including only premature children: four of them with a cross-sectional design,29–32 and one cohort.33
Adams-Chapman et al. present results regarding the prevalence of FD in their sample: 13% of PI’s parents reported dysfunctional eating behavior at 18 months of CA.33 When compared to children who did not behave similarly, the results showed significantly shorter GA and lower birth weight (BW), the prevalence of skin color (black), the occurrence of intraventricular hemorrhage/periventricular leukomalacia, late sepsis, necrotizing enterocolitis, bronchopulmonary dysplasia, administration of postnatal steroids, prolonged mechanical ventilation (MV) and significantly smaller percentiles for length and head circumference. They were also more likely to receive mostly fluids, thickened fluids, and soft foods, compared to the group without FD. The FD in this study was independently associated with MV, hearing impairment, and evidence of neuromotor impairment. Statistical analysis showed that a cut-off point of 34.5 days on MV resulted in a sensitivity of 0.64 and specificity of 0.74 to identify children with dysfunctional eating patterns.
Menezes et al.,32 found that food refusal was associated with the use of infant milk formula, showing 44.4% of children with such behavior. Children using infant milk formula showed a 41% increased chance of having difficulty in CF, compared to those who received exclusive breastfeeding (EBF) or mixed breastfeeding (MBF). Brusco and Delgado identified a significant association between “crying because the child does not want to eat,” extreme prematurity, and extremely low weight.29 Kirk et al. observed that worse performance in child development was associated with the presence of FD, very low birth weight or small for gestational age (SGA), symptoms of anemia, and chronic malnutrition.31
Data on the use of a feeding probe in PI are shown in two studies: in the first study, all the children received a feeding probe, whereas, in the second, only 3.1% of them did not use this feeding device.29,32
The last two studies in this review are cohorts that compared PI by classifying them into two different groups, according to their GA: early and late PI34,35 DeMauro et al. concluded that FD was similar in both groups of PI: both rates of oral motor dysfunction and avoidant eating behavior (spitting, gagging, and crying) decreased over time in both groups.34
In contrast, Kmita et al. identified that extremely PI had more FD than late premature ones, although they did not present statistical calculations for this difference. They also identified that FD is more associated with gastrointestinal or neurological existing issues, such as periventricular leukomalacia, cerebral palsy, gastrointestinal complications of prematurity, gastroesophageal reflux, restriction of intrauterine growth, and bronchopulmonary dysplasia.35 Still, according to Kmita et al., in the qualitative analysis, six categories of FD were identified, showing the following aspects as the most clearly related to the CF period: difficulty introducing new foods, textures, flavors (refusal, vomiting, difficulty biting, chewing, or swallowing food) and refusal to try to eat independently.35
Three studies are unanimous in concluding that PI is less likely to receive BF than full-term infant.25,27,28 Cerro et al. found that there was a significantly higher prevalence of full-term infant who are breastfed up to the sixth month of life (55%) than PI (23%).25 The authors also concluded that among PI, those who were predominantly breastfed up to the sixth month of life had two advantages over PI fed predominantly with CF: they started earlier autonomous feeding, and their parents more often referred to as having “good” or “very good food” in the first year of life.
Regarding the use of pacifiers and nursing bottles, Brusco and Delgado29 found no significant association between using a pacifier and decreased EBF time, whereas Menezes et al.,32 found that, before six months of CA, no PI who received EBF had parafunctional habits, such as the use of pacifiers, nursing bottles, or both.
It is interesting to highlight the parents' perception of the FD in their children's development. When they were asked broadly if their children had FD, in two studies, several parents responded positively (18.8% in the first and 50% in the second study).29,32 However, when they were asked specifically about the presence of each FD separately, the number increased: 53.1% and 73.7%, respectively, for the first and second studies.
Two studies showed interesting a result regarding parents’ satisfaction with their children’s eating patterns. In the first one, although 29% of PI’s parents reported their children were underweight or very underweight, most of them reported being satisfied, happy, or very happy concerning their children’s growth (92%) and health (98%). In the second study, PI’s parents were significantly more satisfied with their children’s eating habits than full-term infant’s parents: 67% of PI’s parents were “very satisfied” (67%) while 31% of full-term infant’s parents reported the same.25,27
Chung et al. concluded that parents were more satisfied with their children’s growth when they were “informed” by the health professional about the age correction in the child's growth curves if compared to those classified as “uninformed”.30 At the time of CF, parents of children considered “ready” for CF (assessed through a questionnaire applied to parents) were more satisfied and comfortable with their children’s eating habits when feeding them; less stressed at mealtimes; and reported a better appetite of their children when compared to the parents of children considered “not ready.” Thus, Chung et al. indicate that the health professional’s failure did not inform parents about CA can contribute to early CF despite the lack of developmental readiness.30
Cerro et al. also indicate that quality and quantity of food consumed are considered the most important issues (78% and 15%, respectively), while only 8% identify the development of food skills or food interaction as an important issue.25 In general, PI’s parents “agreed” or “strongly agreed” that their children should “be free to eat whenever they are hungry” and “decide when they are already satisfied.” On the other hand, 69% “disagreed” or “strongly disagreed” with the idea their children can choose the type of food they are going to consume the most common management for the children’s eating refusal was to set the food aside and offer an alternative (46%); offer the food again later (42%) and force the children to eat (5%). Regarding eating, the worst performance was reported by PI’s parents as less favorable in terms of growth, health, and weight of their children if compared to full-term infant’s parents. PI’s parents were more likely to offer a food reward to encourage their children to eat and agreed less with the statement “children can be free to decide when they are already satisfied.”
Regarding family income, one study carried out in Brazil evidenced that 50% of families had an income lower than two minimum wages,29 whereas another study carried out in Rwanda identified that 77.1% of families were classified as poor and 22.8% as very poor.31
Regarding educational level, Migraine et al. identified that CBAT’s mothers had a higher educational level than PI’s mothers and that the level above high school was associated with a lower risk of low food repertoire at the age of two.28 Brusco and Delgado identified PI’s mothers who had completed high school (43.8%) and those who had an incomplete elementary school (34.4%).29 Kirk et al. also found similar results: caregivers who had no formal education (22.1%) and those who had completed primary school (69.6%).31 Still, in that same study, they found an association between high scores in the child development questionnaire concerning the parents’ highest educational level and the least children in the house, pointing out three as the average number of children in the house of these families.
Cerro et al. also identified that parents who were more favorable about their child’s health were more likely to report that they were supported by a significantly larger number of people when exercising their parenting.25 The application of the tools for evaluating the quality of cross-sectional and cohort studies is shown in Tables 4 and 5, respectively. No included study met all items. As for the Newcastle-Ottawa Scale application on cohort studies, all of them scored on the items on exposed cohort representativeness, exposure verification, demonstration of the absence of result of interest at the beginning of the study, and outcome evaluation.
|
Brusco, Thaísa R.; Delgado, Susana E.; 2014 |
Chung et al., 2014 |
Kirk et al., 2017 |
Menezes; Steinberg; Nóbrega, 2018 |
Y |
Y |
Y |
Y |
|
|
Y |
Y |
Y |
Y |
|
Y |
Y |
Y |
Y |
|
NA |
NA |
NA |
NA |
|
N |
N |
N |
N |
|
NA |
NA |
NA |
NA |
|
NA |
Y |
Y |
Y |
|
NA |
N |
N |
A |
|
NA |
NA |
NA |
NA |
|
N |
N |
Y |
N |
|
N |
N |
N |
N |
Table 4 Agency for Healthcare Research and Quality (AHRQ) checklist to assess the quality of the included studies
Abbreviations: N, no; NA, not applicable, Y, yes
|
|
Adams-Chapman et al., 2013 |
Cerro et al., 2002 |
DeMauro et al., 2011 |
Johnson et al., 2016 |
Jonsson; Van Doorn; Van Den Berg, 2013 |
Kmita et al., 2011 |
Migraine et al., 2013 |
Selection |
|
* |
* |
* |
* |
* |
* |
* |
|
* |
- |
* |
* |
* |
* |
- |
|
|
* |
* |
* |
* |
* |
* |
* |
|
|
* |
* |
* |
* |
* |
* |
* |
|
Comparability |
|
* |
- |
- |
** |
- |
- |
* |
Outcome |
|
* |
* |
* |
* |
* |
* |
* |
|
- |
* |
- |
* |
* |
- |
* |
|
|
* |
* |
- |
* |
- |
- |
- |
Table 5 Quality assessment based on the Newcastle-Ottawa Scale of included studies
*: The study has met the criteria
When applying the AHRQ, it is noteworthy that all studies scored positively on the information source, clarification on the inclusion and exclusion criteria of the individuals in the research, and period indication used to identify the patients. Three items were considered not applicable to the design of the studies due to the inadequacy of the question.
Efforts toward PI’s feeding are extremely important since studies show that a considerable number of children with FD are premature. As DeMauro et al. did not identify a difference in the presence of FD between late and early PI,34 Kmita et al. in turn,35 point out that earlier PI had more FD than late PI, and those children presenting gastrointestinal or neurological issues showed more FD. This is corroborated by Park et al., whose study evaluated the presence of FD in three groups (extremely premature, moderate to late, and full-term infant) and identified that extremely premature infants showed more symptoms of eating problems, followed by moderate to late and, finally, full-term infant.36 Park et al.,36 also reinforce the studies by Kmita et al. when they identify several medical risk factors associated with increased symptoms of dietary problems, such as the need for oxygen after 40 weeks of GA, congenital heart disease, structural anomaly, genetic disease, infantile paralysis, developmental or speech delay, sensory processing disorder, visual impairment, and gastroesophageal reflux.35 Recently, a meta-analysis found that the prevalence of problematic feeding was significantly different across categories of gestational age.37
The benefits of breastfeeding are already well established and among them is the reduced risk of infection in the neonatal period, which is even more important in the case of PI, who have an increased risk of developing infections and necrotizing enterocolitis, for example.1,38 Reinforcing even more the benefits of breastfeeding in PI, Menezes et al., found the exclusive use of IF increased the risk of PI developing FD in the CF period, compared to those receiving EBF or MBF.32
Besides disadvantaging breastfeeding, prematurity causes the rupture of the mother-baby binomial due to hospitalization in a neonatal unit. Thus, it is even more important to highlight the Baby-Friendly Hospital Initiative and the Kangaroo Care,39 which promote and encourage the maintenance of BF.40 In addition to the psychological issues involved, this rupture because of the neonate’s hospitalization can harm breast milk production.41 So, as far as possible, it is necessary that these mothers can be supported by health professionals, to maintain the stimulus of lactation through milking (at the Human Milk Banks or home), while their children cannot be directly breastfed, especially in the case of extremely PI.
The results obtained by Brusco and Delgado29 and by Menezes et al.,32 may be curious when they indicate that there were different response rates when parents were asked broadly about the presence of FD and when were asked in a specific way. Depending on the type of question, the change in response may indicate these parents need some guidance from health professionals regarding the expected behaviors in their children’s development, or what can be considered an issue that requires greater attention or even treatment.
Regarding the perception and acceptance of parents about their children’s feeding, it is interesting to highlight the fact that PI’s parents were more satisfied with their children’s eating habits than full-term infant parents.27 Cerro et al. point out premature’s parents reported almost one-third of children with low or very low weight, and more than 90% were happy or very happy with their children’s growth and health.25 This result may ultimately indicate that PI’s parents have a particular view on their children’s development, that is, from the moment of their children’s birth (experience of complicated situations due to prematurity), they may be more satisfied with their children’s current development because they have re-signified the way how they deal with their children’s challenges and achievements.
Cerro et al. bring important results when they find that parents of PI agreed less on the idea that children can be free to decide when they are already satisfied and, a low rate of them identifies the development of eating skills/food interaction as important.25 This fact is aligned with what Haines et al.,42 recommended when they advise that, to promote healthy eating in children, their parents’ performance must be positive, allowing children to actively participate in meals, gently encouraging them to eat and avoiding coercive strategies, such as food reward found by Cerro et al.25
The results presented here about the socio-demographic and economic data are consistent regarding poverty and low educational level as risk factors for prematurity,1 according to the World Health Organization’s document published in 2012, which brings the implementation of reducing-poverty strategies and the guarantee of universal access to education and the food security promotion among the actions for the reduction of premature births. These strategies are also important in reducing FD in PI, according to Migraine et al.,28 who state that mothers’ educational level was inversely associated with the risk of low food repertoire at the age of two. This can be due to the lack of access to a variety of foods or also to unreachable information about nutrition.
It is important to note that the term “feeding difficulties” chosen to make part of the title of this work is, to some extent, widely used. However, the studies included in this review have not always used the same expression to refer to dysfunctional eating behaviors. Therefore, it is necessary for a more appropriate and precise way to deal with the topic, as Goday et al.,43 recently proposed, presenting the term Pediatric Feeding Disorder, which can be briefly defined as “impaired oral intake that is not appropriate for the age and is associated with medical and nutritional dysfunction, eating ability and/or psychosocial dysfunction”.43 The definition of a precise term will be able to offer greater cohesion of the knowledge acquired so far and provide patients and families suffering from FD with a more efficient and better-based approach as already proposed by Milano, Chatoor, and Kerzner in a study published also in 2019.44 Also, a recent meta-analysis found that the main limitation was that few of the studies used formalized assessments of problematic feeding with evidence of adequate psychometric properties.37
It is worth highlighting as strengths the care in elaborating terms in order not to neglect results, and not to limit the data of the studies. Thus, there is still no specific term to refer to dietary difficulties, and, therefore, it was important to use wide-ranging terms; thus, the initial search result proved to be broad, preventing the omission of results. Also, this aspect enabled the selection process to be more dependent on the researchers’ rigor when reading the studies.
As a limitation, we can highlight this is a systematic review of observational studies, followed by the selection in Portuguese and English and the search in three databases. Moreover, all studies were based on the report of parents/guardians regarding the presence or absence of FD in their children’s development. This means the perception was given by lay individuals, which can generate confusion between the concept of FD and normal/expected behaviors for age.
As for the quality of the studies included in the review, it was considered that they presented an average score both on the Newcastle-Ottawa scale and on the AHRQ checklist since none of the studies filled out all the items questioned.
Considering the studies available to date, the question is still inconclusive as to whether there is a pattern of FD, which is more consistent with prematurity, and in what ways FD occurs in this population to the detriment of the occurrence of FD in full-term infants. The heterogeneity of the studies included in this review, the small sample of some, and their observational nature prevent a strong conclusion regarding their findings. However, these same characteristics make us realize how incipient this field of investigation is and needs further studies, which would be optimized with the proposal of establishing a precise term for the subject. Simultaneously, the complexity of identifying and defining FD currently suggests that the treatment performed by only a medical team approach is not enough. It is evident the importance of considering the various perspectives related in a multi-professional follow-up to ensure that this premature population can have adequate development in all areas of health, including child growth and development, development of oral motor skills, eating behavior, and healthy eating habits.
Therefore, it is important to direct efforts both to prevent premature birth and to treat its consequences, which affect the individual’s and family’s health as well as public health. It is also interesting to highlight the need to provide specific material showing guidelines on the introduction of food in PI, including questions about caregivers’ suitable approach when feeding, and assistance to parents and health professionals in this process, since PI is at greater risk of developing these difficulties.
None.
Luíza Piletti Plucenio: Conceptualization; Data curation; Formal analysis; Methodology; Resources; Writing – original draft.
Christy Hannah Sanini Belin: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing – original draft; Writing – review & editing.
Paula Ruffoni Moreira: Formal analysis; Methodology; Writing – review & editing.
Roberta Aguiar Sarmento: Conceptualization; Supervision; Writing – review & editing.
The study was funded by the Research Incentive Fund (FIPE/HCPA number 2020-0112).
The authors declare that there is no conflict of interest.

©2023 Plucenio, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.