Journal of
eISSN: 2378-3184


Research Article Volume 4 Issue 6
1Department of Forensic Science, Central Police University, Taiwan
2Institute of Marine Affairs and Resource Management, National Taiwan Ocean University, Taiwan
3Department of Maritime Police, Central Police University, Taiwan
Correspondence: Yi-Che Shih, Department of Maritime Police, Central Police University, No.56, Taiwan, Tel 886-2-2377-3038
Received: November 11, 2016 | Published: December 21, 2016
Citation: DOI: 10.15406/jamb.2016.04.00101
Redox potential is a frequently used measurement in studies under and around marine cage farms to assess the environmental status of marine aquaculture. This study presents an application of a geographic information system (GIS) to document sediment geochemical measures in cobia aquaculture in order to assess the condition of the benthic environment using the Chu-Wan marine fish farm as a case study. The objectives of the study was to combine GIS presentation with chemical measures as an important and innovative methodology in marine aquaculture environmental management. The results show a real distribution patterns for both redox potential and sulfide content in relation to position of the farm and the current pattern.
In 2007, Taiwan produced over 1.49 million tonnes (MT) of fish, valued at NTD$ 94.8 billion (US$ 2.9 billion), according to the Fisheries Year Book 2007 (Fisheries Agency, Council of Agriculture, Taiwan).1 Finite resources and the land-based expansion of Taiwanese culture have caused environmental degradation. Therefore, the Taiwanese government has supported the development of offshore fish farming as a recent priority for national aquaculture development. Marine cage aquaculture began in the 1970s in the Penghu Archipelago. Among many species of cultured marine fish, cobia has become a popular species for cage aquaculture because of its fast growth rate and comparatively low production cost. Cobia is a finfish species with emerging global potential for mariculture, and it has been the subject of research on spawning and grow-out since the early 1990s.2
Marine aquaculture may also cause environmental degradation from feces, uneaten feed and use of chemicals if the local carrying capacity is exceeded.3 The most important benthic impact relates to the formation of anoxic sediments4-6 which can be easily monitored. Both sediment condition and waste accumulation are good indicators for detecting the environmental condition at aquaculture sites. Beveridge7 revealed that environmental deterioration because of high organic matter concentrations in the sediments may affect the health of farmed fish and hence profitability. Waste and waste-makes quality management for marine aquaculture in other countries has not only focused on the release limitations of organic matter, and nutrients8-11 but also on chemicals12,13 used on the farms regardless wherever into estuaries or near shore coastal waters.14,15 This investigation uses an environmental monitoring program to understand and assess the environmental impact of cobia farming.
The distribution patterns of redox and sulfide concentrations were mapped and presented at Penghu. Results show that the redox potential values at <-100 mV and high sulfide concentrations ranged from 306 to 1800 μM in sediments were clearly dominant in the feeding zone of aquaculture operations. Positive redox potential values and low sulfide concentrations < 300 μM were observed at the north direction nearby the crossing bridge that is away from the aquaculture site and upstream of the current flow inside the Inner Islets. Some sampling sites at the outward sites, that were 200 meters, 500 meters away from the aquaculture operations, had <-100mV. These suggest the wastes from the aquaculture sites were driven with the current and dispersed toward the outside inner islets.16 Summerfelt et al.17 reported that an ORP of 375-525 mV was required to reach the mean daily ozone concentration necessary to obtain full-flow disinfection in freshwater RAS. For southern rock lobster larvae, survival was higher and bacterial contamination was lower when the ORP was between 330 and 500 mV.17,18 With moderate ozonation corresponding to an ORP value of 250 mV in a low exchange freshwater RAS, rainbow trout showed improved performance compared to a system without ozonation.19,20
A Geographic Information System (GIS) contains powerful tools for processing spatial reference data and can be used to organize and present spatial data as part of effective environmental management planning.21 Furthermore, buffering is one of the proximity techniques in GIS.22 Buffer applications studies of the impact zones of amphibian species,23 riparian zones for non-point-source pollution,24 and water quality monitoring.25 The objective of this study was initiated by choosing the Penghu cobia cage site as a pilot study for the assessment of environmental capacity in support of developing risk assessment methodologies and guidelines for the sustainable use of marine aquaculture sites in the APEC region under the sponsorship of APEC Marine Resources Conservation Working Group.26
Study site characterization
The Penghu Archipelago, located in the middle of the Taiwan Strait, is a major fishing ground for most local community. Penghu Archipelagos have 276 km of rugged coastline, around 173.8 hectares of marine culture sites and the local community marine farmers 1,416. Chu-Wan, the largest marine cage culture site, there are around 400 cages per sites in the bay of Penghu, which is located near the town of Chu-Wan, Shiyu Islet, Penghu (23°38.398′ N 119°31.018′ E) (Figure 1a). The depth of the Chu-Wan culture area is around 8-15m on average, and the salinity is about 28.0-32.4%. The average temperature is 25-27°C from spring to autumn, declining to an average of 21-22°C in the winter, with the lowest short-term temperature decline to below 16°C during the winter season. The Chu-Wan cove farm site of Penghu Island was chosen for this investigation because Penghu’s farming production over 60% of the cobia net-cage culture products in Taiwan, and it was the first cobia farm in the world.
The current flow goes from north to south (Figure 1b). The Inner Islets have a tidal current velocity in the range of 0.5-339cm/sec. The highest current velocity is observed at the bridge connecting the Bei-Sha and Shi-Yu Islets. The lowest current velocity (0.5 cm/sec) is observed at Magong Cove. The tidal current can reach two-thirds of the total length of the Inner Bay within six hours. The turnover time of the water at the Inner Islets has been estimated to be one week.
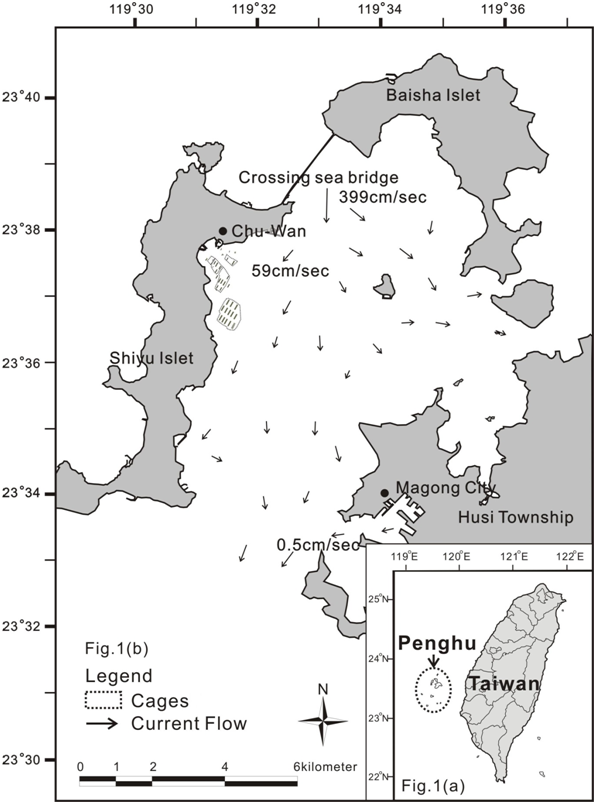
Figure 1 Marine cage culture sites and flow patterns of the residual current direction in Penghu, Taiwan. 1a) overall geographic location; 1b) current flow, cage sites and flotilla positioning within the licence boundaries (short strips) at Penghu Inner Islets.
Sampling collection
Sediment samples were collected by divers from May 2005 every quarter. Samples were collected, running parallel with the current, under each cage farm from 0m, 50m, 100m, 200m, 500m, 1000m, 2000m upstream to 2000m downstream. This was done to investigate environmental conditions as they gradually change from areas of intensive farming activity to areas farther away from the farm. Sediments were also sampled at reference sites with no aquaculture activities (Figure 2). In addition to parallel sampling, samples were also gathered from the cage boundary 45 degrees downstream at 1000m and 2000m.
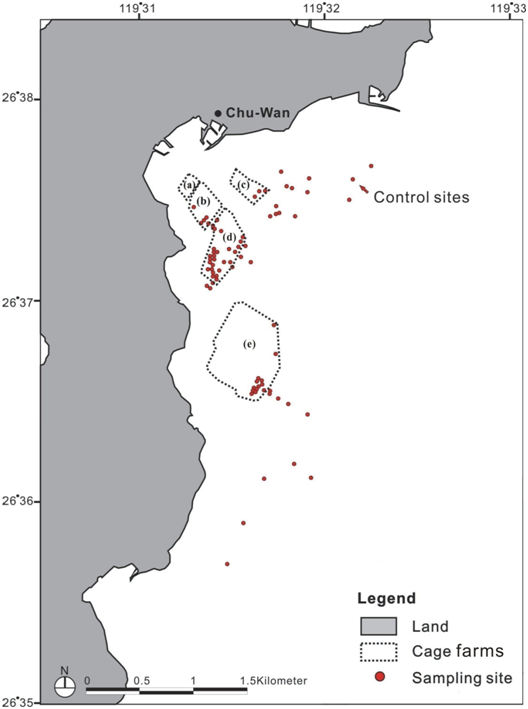
Figure 2 Chu-Wan Cove sampling site of Penghu bay. The cage farms licences are within the dotted line areas. There are 5 blacks of cage flotillas with the following production characteristics: (a)=10 cages; (b)=12 cages; (c)=26 cages; (d)= 240 cages; (e)= 360 cages with total annual biomass output of around 5,000 tonnes.
A time series of marine environmental impact assessment related to the whole cobia cage farming process at the Chu-Wan Cove grow-out site was conducted from May 2005 to December 2006. The study was performed for 18 months, until the adult fish were ready for harvest. This was to permit the assessment of long-term changes in cage conditions and discovery of the most appropriate method for monitoring changes in environmental conditions related to aquaculture development.
Environmental monitoring program
The environmental monitoring program (EMP) approach of Chou et al.27 was adopted to evaluate the marine environmental impact of cobia marine aquaculture environmental management. The EMP ratings were assessed by the diver on site following the guidelines defined by the Department of Environment and Local Government of New Brunswick, Canada.28 Redox potential in core samples was measured with a Fisher Scientific Co., USA. AP-63 (PH/MV/Temp/Ion), using methods described by Wildish et al.29 The sulfide probes were calibrated just before use on each sampling day by checking against freshly prepared Na2S.9H2O solutions at three concentrations (10μM, 100μM and 1000μM).29 Sediment samples were mixed immediately after collection with sulfide antioxidant buffer (SOAB from Fisher Science Co.) (1:1 v/v). Sulfide levels were recorded in millivolts (mV) when the reading stabilized. These methods were confirmed experimentally by Wildish et al. & Crawford et al.29-32 demonstrated that water quality parameters do not have significant effect on the culture environment and provided no help to reveal the impact condition of the culture site in Penghu and the redox potential was -420 mV.
Geographic Information System application
This study presents for use in cobia aquaculture a GIS application based on Arc Map,33 a GIS software package. The application includes sediment chemical measures (redox and sulfide) for assessing the impact of environment at the Chu-Wan marine fish farm. We use the buffer zone to show the impact for marine cage culture (Figure 3&4). On the other hand, the boundary use inverse distance weighted (IDW) interpolation determines cell values using a linearly weighted combination of a set of sample points. The weight is a function of inverse distance. The GIS derives the impact map by overlay and spatial analysis of a point layer of sampling sites representing control sites, outside and under the cages.
Figure 3&4 present the graphically position data plot distribution patterns of GIS, redox and sulfide concentration gradients respectively. Analytical results indicate that redox potential values of <−100mV and high sulfide concentrations of >301μM in sediments were clearly dominant under the cages and nearby of the aquaculture operations. Positive redox potential values and low sulfide concentrations of <50μM were observed in the north direction near the bridge, away from the aquaculture site and upstream of the main current flow inside the Inner Islets. Some outward sampling sites, specifically those 200m and 500m away from the aquaculture operations, had redox potential values of <−100mV. These values showed that wastes from the aquaculture sites were driven with the current and dispersed toward the outer section of the inner islets.
Figure 5 illustrates the plot of redox potential versus sulfide values. These results indicate an exponential relationship between sediment redox potential values and sulfide concentrations, with a correlation coefficient value of 0.734 (or 0.724 without outliers). This supports the view that the sediment geochemistry data from this study were valid and useful for resource management requirements.
Sites or region-specific environmental management programs can feasibly be developed from the two-year environmental data now available for comparison with the former survey data. The sampling data were compared with research data from Ku et al.32 that suggested the Chu-Wan mariculture area had not undergone huge environmental changes over the past two decades. However, the redox and sulfide survey data from cage sediment in Chu-Wan aquaculture areas for 2005 and 2006 indicate that the environment has not been maintained in a suitable condition for sustainable development, especially the inshore area.
Many countries have set mandatory minimum distances between farms, e.g., 5 km in Norway and 8 km in Scotland Beveridge & Crawford et al.7,15 studied marine farming development plans (MFDP) in Tasmania that were based on studies from Europe where farm wastes were generally found to be concentrated within 35m of the edge of the cage. However, the distance between culture areas in Chu-Wan is only around 100m, showing that the marine culture capacity in this area is too dense. The benthic environmental impact of marine culture is typically at its greatest at 20-50m from the site.31 The sediment impact is dependent on farm size, water depth, hydrodynamic regime, and bottom topography as well as sediment characteristics.34,35 For example in Canadian marine cage cultures it can reach the 100m zone.27 While in more emerg system Chile this may be a distance more then twice of farm.34 Our analytical results for the redox potential and sulfide value in the downstream area of Chu-Wan indicate that the benthic impact can reach 500m. The impact map can clearly help farmers and fishery authorities plan the environmental capacity, rotation timing and culture guidelines according to the impact map.36
The environmental impacts of aquaculture need to be measured and quantified, so that the fishery authorities can estimate the potential impacts of new lease proposals and use this information to restrict the expansion of sites to a safe level, thus minimizing the risk of environmental degradation. GIS provides an ideal visualisation platform for professionals to analyze the many data on a variety of environmental parameters, so that results for such factor can be over layed and interactions be interposed, apply models and make the most suitable decisions for the fishery authorities and farmers. The GIS combined method with geochemical measures is particularly important in monitoring the future development of coastal zone, and can provide early indicators signals for inshore, and offshore culture environment management. This type of investigation could contribute to a fully integrated, sustainable decision support baseline for aquaculture site selection and future development.
The Gamma distribution is parameterized in this paper in terms of the mean and the standard deviation. The probability density function of a Gamma distributed variable x is
None.
None.

©2016 , et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.