eISSN: 2373-6372


Research Article Volume 8 Issue 3
1Department of Hepatology, Japanese Red Cross Society Himeji Hospital, Japan
2Department of Oral and Maxillofacial Surgery, Japanese Red Cross Society Himeji Hospital, Japan
Correspondence: Kazuhiko Morii, Department of Hepatology, Japanese Red Cross Society Himeji Hospital, 1-12-1 Shimoteno, Himeji, Hyogo 670-8540, Japan, Tel +81 79-294-2251, Fax +81 79-296-4050;
Received: November 08, 2017 | Published: December 28, 2017
Citation: Morii K, Yamamoto T, Nakamura S, Fujiwara S, kushin H (2017) Multilocular Pyogenic Liver Abscess with Poorly Liquefied Content: A Potential Therapeutic Concern. Gastroenterol Hepatol Open Access 8(3): 00280. DOI: 10.15406/ghoa.2017.08.00280
Background: Percutaneous catheter drainage (PCD) is currently the preferred therapeutic procedure of pyogenic liver abscesses (PLAs). However, multilocular poorly liquefied PLAs (MPLPs) are unsuitable for PCD. As reports on MPLPs are scarce, we aimed to explore possible therapeutic strategies for this serious type of PLA.
Methods: The medical records of patients with PLA who were hospitalized at our institution between January 2011 to December 2015 were retrospectively reviewed. Demographic factors, biochemical, bacteriological, and imaging results, and treatment outcomes were evaluated.
Results: Fourteen patients with MPLPs and 37 with drainable PLAs were identified and included in the study. Clinical characteristics were generally similar between the two groups. Klebsiella pneumoniae accounted for the majority of the culprit pathogens in MPLPs. Patients with MPLPs were managed medically without PCD. Despite this, treatment outcomes were not inferior compared to patients with drainable PLAs. Contrast-enhanced images revealed that the intricate septa of the MPLP maintained its blood perfusion. Most MPLPs did not liquefy over the treatment period.
Conclusion: We should remember that MPLP is not rare. The septa may have delivered antibiotics adequately into the MPLPs, thus producing good treatment outcomes. Despite our findings, a high level of suspicion of treatment failure is crucial in these patients to ensure favorable treatment outcomes.
Keywords: pyogenic liver abscess, multilocular, liquefaction, drainage, klebsiella pneumonia
ESBL, extended-spectrum β-lactamase; PCD, percutaneous catheter drainage; PLA, pyogenic liver abscess; MPLP, multilocular poorly liquefied pla
Pyogenic liver abscess (PLA) is the most frequent, potentially life-threatening type of visceral abscess, with an increasing incidence worldwide.1,2 The etiologies and microbiological spectrum show distinct regional differences around the world. Klebsiella pneumoniae is the most common causative pathogen of PLA in eastern Asian countries,3–5 whereas Streptococcus spp. predominates in Europe, Canada, and Australia.6–10 However, recently, Klebsiella pneumoniae has become a more frequent causative pathogen of PLA in Western countries.11,12
Although there is no consensus on the guidelines regarding optimal management strategies of PLA, percutaneous catheter drainage (PCD) is currently the initial treatment option of choice.13,14 In the majority of patients, PCD is reported to be a safe and sufficient treatment, even for huge PLAs measuring ≥10cm.15 In this modern era of minimally invasive PLA management, multilocular poorly liquefied PLA (MPLP) remains a challenge. MPLP is unsuitable for PCD due to its intricate septations and thick viscid pus, and therefore it is treated rather reluctantly with antibiotics alone. This conservative treatment strategy sometimes raises concerns about the risk of treatment failure and certain complications. Indeed, we have experienced a case of huge MPLP with an intraperitoneal rupture who required surgical treatment.16 This MPLP was caused by Klebsiella pneumoniae infection, which is reported to be often associated with multilocular PLA.17,18
Diagnosis of multiloculation of PLA may vary depending on the capacity of the imaging modalities and the reader’s subjective interpretation. According to the previous studies, multilocular abscesses account for about 50% of PLAs.15,19,20 Half of these multilocular PLAs were difficult to be managed with PCD in our recent experience, and accordingly, MPLPs may account for approximately a quarter of PLAs (Figure 1). Despite the relatively high prevalence, there are few reports on MPLPs that describe their clinical characteristics and treatment outcomes.
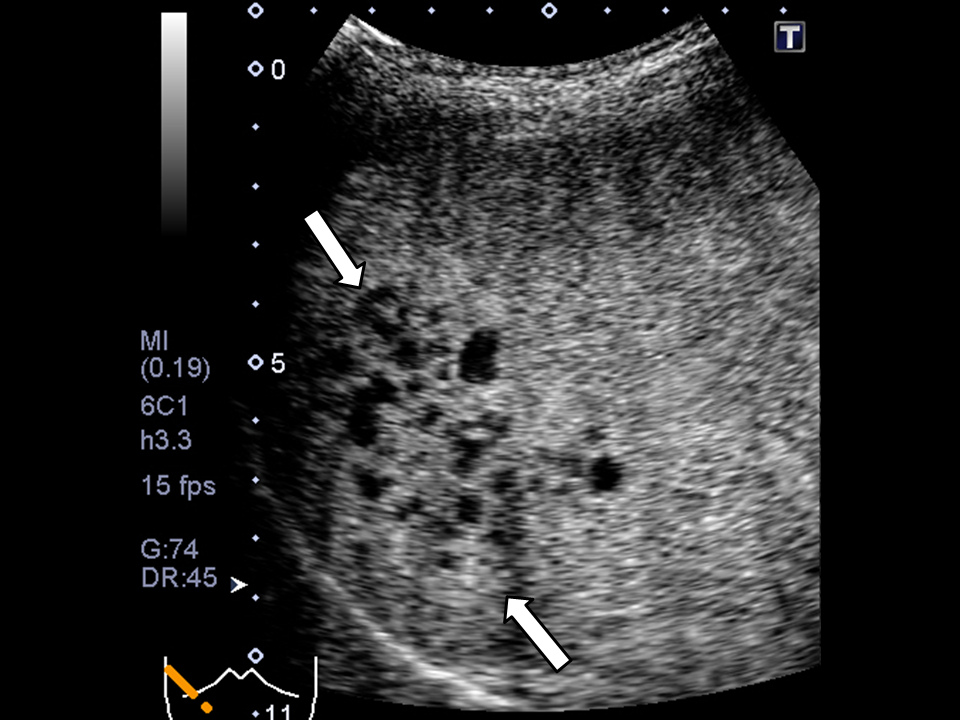
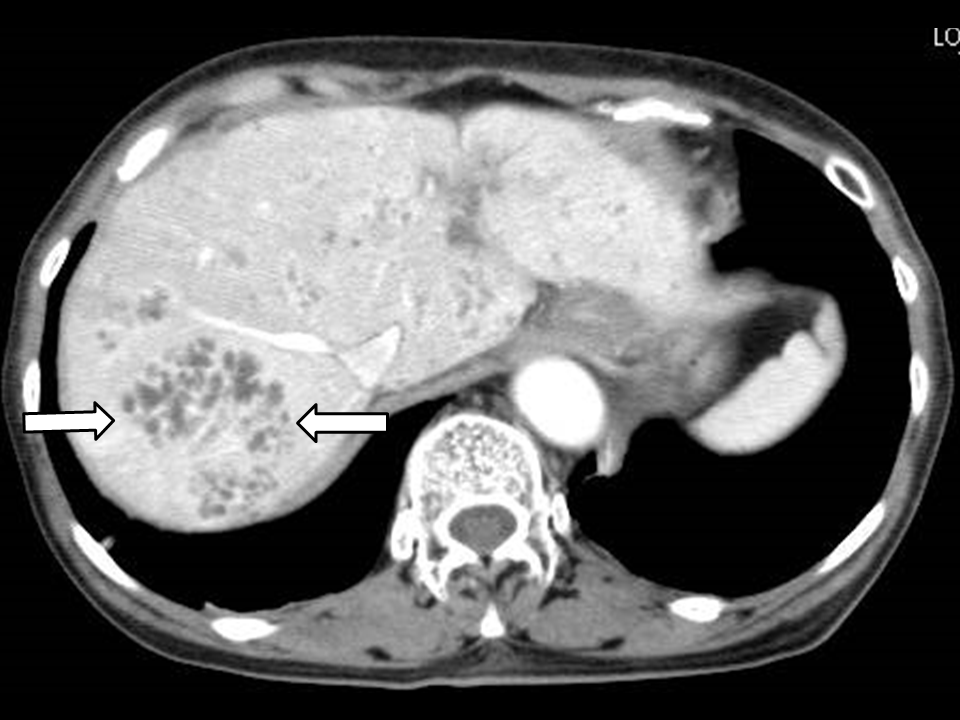
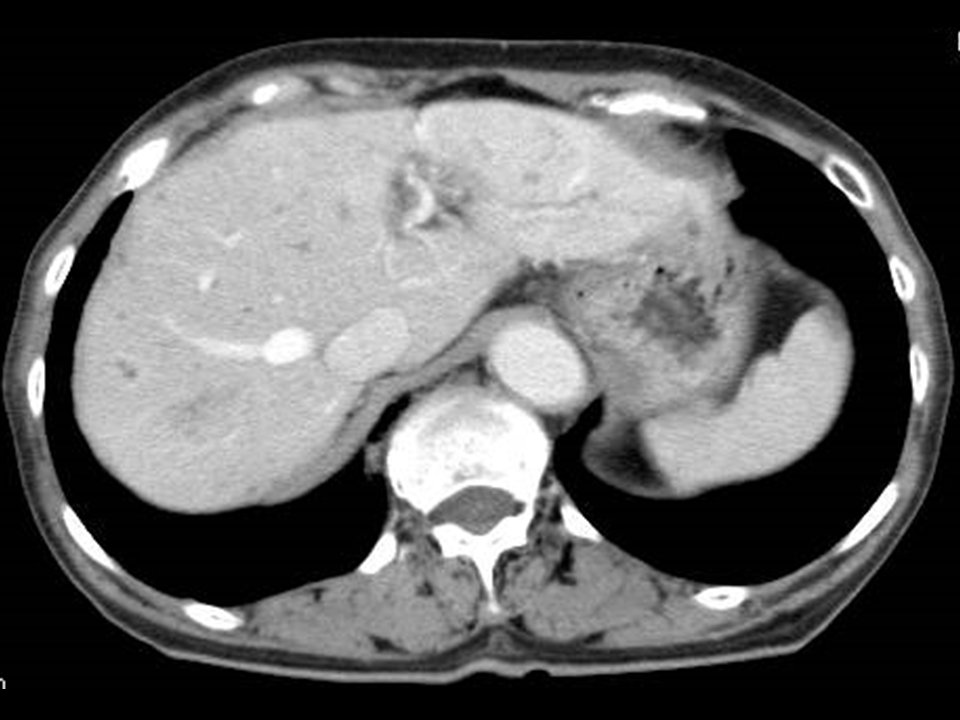
Figure 1 Images of a 76-year-old woman showing a number of MPLPs associated with Klebsiella pneumonia.
A) The post-vascular phase of perfluorobutane-enhanced ultrasonography. B) Contrast-enhanced CT scan. Both (A) and (B) show images obtained before treatment. These images demonstrate the enhancing intricate septation of the MPLP, which exhibits a honeycomb-pattern (arrows).C) Contrast-enhanced CT scan obtained 4 weeks after the antibiotic introduction shows marked improvement of the MPLPs.
MPLP denotes multilocular poorly liquefied pyogenic liver abscess.
Therefore, this study was aimed to investigate MPLPs, comparing their clinical and therapeutic characteristics with those of liquefied drainable PLAs. Further, in this study, we aimed to elucidate the unique abscess structures, clinical and bacteriological features, and treatment outcomes for MPLPs.
Study design
The present study is a retrospective cohort analysis of patients with PLA who were hospitalized at our institute between January 2011 and December 2015. Our institute is a community-based tertiary referral hospital with a specialized hepatology department. The patient inclusion criteria were as follows: (a) patients who were hospitalized at our institute because of PLA between January 2011 and December 2015, and (b) the primary reason for hospitalization was a PLA. The exclusion criterion was the coexistence of presumable primary infectious foci other than PLAs. Electronic medical records were reviewed to evaluate clinical presentation, demographic features, biochemical and serological markers, imaging findings, microbiology of pathogenic organisms, antimicrobial resistance testing, and the therapeutic course of the illness. The ethics committees of our institutional review board approved this study in accordance with the Declaration of Helsinki (number CTM16-011).
Diagnosis of PLA
The initial workup involved abdominal ultrasonography, and if PLA was suspected, computed tomography (CT) was performed to define its anatomical features in detail and to determine its route of infection. In case of biliary-origin PLA, magnetic resonance cholangiopancreatography was used to investigate biliary tract pathologies. Diagnosis of PLA was based on the following criteria: (a) the clinical signs of bacterial infection such as fevers with leukocytosis, (b) the characteristic imaging features with ultrasonography (US) and computed tomography (CT), (c) samples consistent with PLAs were obtained by image-guided aspiration, (d) positive bacterial cultures of blood or pus, (e) resolution of the lesions with antibiotic therapy with/without drainages, (f) a negative fluorescent antibody test against Entamoeba histolytica, and (g) surgical findings, if available, were consistent with PLAs.2
The characteristic imaging features of PLAs were regarded as follows: (a) solitary or multiple lesions tending to coalesce focally, (b) usually well-defined hypoattenuating lesions with faint rim enhancement and perilesional edema by contrast-enhanced CT, (c) unilocular with smooth margins or multilocular with septations and an irregular contour, (d) varying degrees of internal contents depending on the stage of suppuration, liquefaction, and fibrosis, and (e) occasionally containing gas in the lesions.21
Multilocular abscess was defined as an abscess with 2 or more septa. MPLP was defined as a multilocular abscess in which proper source control could not be achieved by PCD due to complex septations and poorly liquefied contents (Figure 1). If a multilocular PLA had liquefied contents and could be effectively managed with PCD, it was categorized as a drainable PLA (a multilocular drainable PLA, Figure 2).
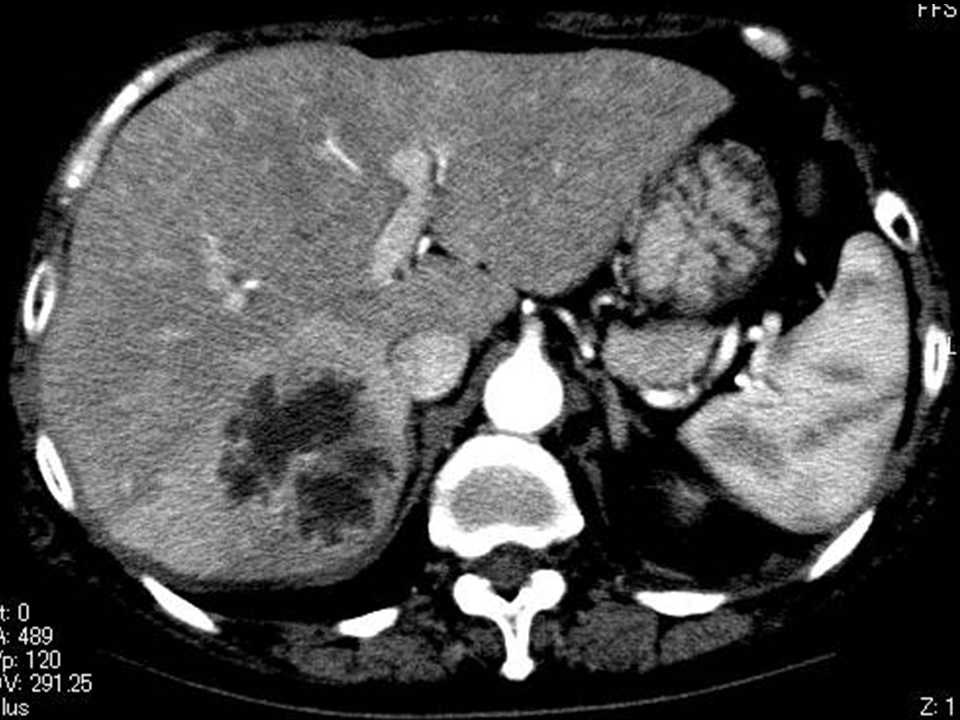
Figure 2 Images of multilocular drainable PLAs.
Contrast-enhanced CT scan of a 70-year-old woman shows a multilocular PLA associated with Klebsiella pneumoniae. This abscess had been effectively managed with percutaneous drainage by breaking through the septa with the catheter. This is not categorized as MPLPs but drainable PLAs.
PLA denotes pyogenic liver abscess.
Data collection
The following variables were explored: sex, age, presenting symptoms, interval from symptomatic onset to diagnosis, route or etiology of infection, preexisting comorbidities, length of hospital stay, and treatment outcomes at discharge (recovery or deceased). Routes of infection were divided into biliary-, portal pyemia-, urinary tract infection-, hematogenous-, and cryptogenic-origin. Biliary-origin routes included cholecystitis, cholangitis, and pancreatitis. Portal pyemia-origin routes implied ascending portal venous sepsis and included clinical presentations such as colonic diverticulitis, enteritis, appendicitis, inflammatory bowel disease, and supra-infected gastrointestinal neoplasms (i.e. ulcerated tumors that allowed bacterial invasion). Hematogenous-origin routes were defined as secondary to hematogenous seeding with a documented bacteremia before the onset of PLA and included clinical pictures such as skin and soft tissue infection and catheter-related blood stream infection. Cryptogenic-origin routes were defined as having no identifiable source of infection despite extensive explorations. Preexisting comorbidities included diabetes mellitus, alcoholic abuse, prior gastroduodenal ulcer, prior abdominal surgery, cardiovascular diseases (hypertension, ischemic heart disease, stroke), and chronic renal failure, all of which have been reported to be associated with PLA fatality.1,2,22,23
Imaging findings were recorded including the number of distinct abscess cavities, their lobar distribution in the liver, their diameters, whether the contents of the PLAs were uni- or multilocular and liquefied or poorly liquefied, and gas-formation. If there were multiple lesions, the largest diameter was recorded. We investigated whether liquefaction of the MPLPs was observed over the treatment courses. All laboratory tests, blood culture samplings, and imaging studies were performed at the initial assessment. All pus specimens were sent for culture during PCD or surgery. Blood and pus samples were obtained for aerobic and anaerobic cultures. Extended-spectrum β-lactamase (ESBL) production was examined when strains of Klebsiella spp. and Escherichia coli were isolated.
Principal treatment strategies for PLA
Treatment procedures consisted of medical treatment, image-guided PCD, and surgical intervention. After the referral of patients with presumable PLA, blood cultures were performed, and empirical treatments with broad-spectrum antibiotics were commenced immediately. A commonly used empirical antibiotic is the third-generation cephalosporin (e.g. ceftriaxone). The antibiotic treatment was optimized afterward according to the results of the bacterial cultures and antimicrobial susceptibility analysis of the isolated pathogens. Patients with abscesses larger than 3cm in diameter predominantly underwent source control by PCD with 7 Fr catheter drainage within 24hours, except in the following situations: (a) when the lesions were MPLPs, and (b) when there was a safety concern about PCD such as bleeding tendencies and a moderate-high amount of ascites.13,14,24 The duration of drainage catheter placement ranged from 4 to 10days. The methodological contrivances of PCD for multilocular PLAs were as follows: (a) breaking through the septa with catheter manipulation to make separated cavities contiguous (Figure 2), and (b) placing a drainage catheter in the main lumen if a PLA had a large main lumen with several ancillary lumens.
Endoscopic biliopancreatic drainage was performed when necessary. Surgical intervention was the last treatment option when medical treatment and PCD failed23 and was only required for one patient with a ruptured abscess.16 Finally, the resolution of the PLA was confirmed by follow-up imaging studies.
Statistical analysis
The primary statistical objective was to identify meaningful differences in clinical characteristics or laboratory data between patients with MPLPs and drainable PLAs. Standard statistical methods were used to analyze the results. Continuous variables were compared using the Mann-Whitney rank-sum or Student’s T test and categorical variables by chi-square tests. Continuous variables are presented as mean ± standard deviation or median and categorical variables as absolute values or percentages. P values below 0.05 (two-tailed) were considered statistically significant. All analyses were performed using the EZR v1.26 software program.25
After application of the inclusion and exclusion criteria, a total of 51 patients were included in this study (Table 1). Twenty-six (51%) patients had multilocular PLAs and the other 25 (49%) unilocular PLAs. Twelve of the 26 patients with multilocular PLA could be effectively managed with PCD, and accordingly the remaining 14 (27%) patients accounted for those with MPLPs. Patients with drainable PLAs consisted of 12 patients with multilocular PLAs and all of 25 with unilocular PLAs. The baseline demographic and clinical characteristics of the patients with MPLP and drainable PLA were very similar (Table 1 & 2). The chief complaint, abdominal pain, tended to be less common in patients with MPLP. The location, number, and diameters of the abscesses were not different between the two groups. MPLPs were significantly associated with a cryptogenic source of infection.
MPLP |
Drainable |
P Value |
|
Patients, n (male: female) |
14 (11:3) |
37 (24:13) |
0.3 |
Age, year |
68.8±13.1 |
72.2±12.2 |
0.5 |
Symptomatic onset to diagnosis, days |
6.4±7.6 |
6.6±6.4 |
0.5 |
Clinical presentation, n (%) |
|||
Fever and chills |
10 (71.4) |
21 (56.7) |
0.3 |
Abdominal pain |
1 (7.1) |
10 (27.0) |
0.1 |
General malaise |
3 (21.4) |
4 (10.8) |
0.3 |
Anorexia |
0 |
2 (5.4) |
0.4 |
Number of abscess cavities, n |
2.7±1.9 |
2.0±2.0 |
0.6 |
range, n (%) 1 |
10 (71.4) |
26 (70.3) |
|
2-4 |
3 (21.4) |
5 (13.5) |
|
≥5 |
1 (7.1) |
6 (16.2) |
|
Location of abscess, n (%) |
|||
Right lobe |
10 (71.4) |
25 (67.6) |
0.8 |
Left lobe |
2 (14.3) |
9 (24.3) |
0.4 |
Bilateral lobes |
2 (14.3) |
3 (8.1) |
0.5 |
Diameter of the abscess, cm |
5.9±2.5 |
6.0±3.2 |
0.6 |
range, n (%) ≤3cm |
3 (21.4) |
10 (27.0) |
|
3.1-5.0cm |
4 (28.6) |
5 (13.5) |
|
5.1-7.4cm |
4 (28.6) |
11 (29.7) |
|
7.5-9.9cm |
0 |
7 (18.9) |
|
≥10cm |
3 (21.4) |
4 (10.8) |
|
Route of infection, n (%) |
|||
Cryptogenic |
10 (71.4) |
10 (27.0) |
0.003 |
Biliary |
3 (21.4) |
16 (43.2) |
0.1 |
Portal pyemia |
1 (7.1) |
7 (18.9) |
0.3 |
Hematogenous |
0 |
2 (5.4) |
0.4 |
Urinary tract infection |
0 |
2 (5.4) |
0.4 |
Table 1 Clinical and biochemical variables of individuals with overweight-obesity
P values were calculated to identify statistically significant differences between patients with MPLP and drainable PLAs
MPLP |
Drainable |
P value |
|
Underlying diseases, n (%) |
14 |
37 |
|
Diabetes mellitus |
6 (42.9) |
14 (37.8) |
0.7 |
Biliary disease |
2 (14.3) |
12 (32.4) |
0.2 |
Pancreatic disease |
1 (7.1) |
2 (5.4) |
0.8 |
Liver cirrhosis |
1 (7.1) |
4 (10.8) |
0.7 |
Gastroduodenal ulcer |
0 |
4 (10.8) |
0.2 |
Inflammatory bowel disease |
0 |
2 (5.4) |
0.4 |
Colonic diverticulitis |
0 |
3 (8.1) |
0.3 |
Hypertension |
3 (21.4) |
11 (29.7) |
0.6 |
Ischemic heart disease |
2 (14.3) |
2 (5.4) |
0.3 |
Stroke |
0 |
1 (2.7) |
0.5 |
Chronic renal failure |
3 (21.4) |
2 (5.4) |
0.08 |
Risk factors, n (%) |
|||
Smoking |
3 (21.4) |
8 (21.6) |
0.9 |
Prior abdominal surgery |
4 (28.6) |
4 (10.8) |
0.1 |
Alcoholic abuse |
3 (21.4) |
5 (13.5) |
0.5 |
Prior malignancy |
1 (7.1) |
4 (10.8) |
0.7 |
Immunosuppressive medication |
1 (7.1) |
2 (5.4) |
0.8 |
Percutaneous catheter drainage |
0 |
24 |
- |
Surgical treatment |
1 |
0 |
0.5 |
Length of hospital stay, days |
24.4±16.2 |
20.3±14.6 |
0.3 |
Fatality, n (%) |
1 (7.1) |
1 (2.7) |
0.5 |
Table 2 Underlying conditions and treatment outcomes in patients with MPLPs and drainable PLAs
Laboratory data were also not different between the two groups (Table 3). The overall positive rate of bacterial culture was 51%. The isolated bacterial strains had good sensitivity to commonly used antibiotics, and no ESBL-producing strains were found. Seven of the 14 patients with MPLPs showed positive bacterial culture results: 6 (86%) of them were Klebsiella pneumoniae, and only 1 (14%) was Streptococcus bovis. Conversely, Klebsiella pneumoniae was isolated in 14 patients, 6 (43%) of whom had MPLP. Thus, patients with MPLPs were significantly more likely to exhibit Klebsiella pneumoniae infection than those with drainable PLAs. In addition, 7 of 12 patients with multilocular drainable PLAs exhibited positive bacterial culture results: 4 (57%) of them were Klebsiella pneumoniae.
Reference Range |
MPLP |
Drainable |
P Value |
|
Peripheral white-cell count, /mm3 |
3,300-8,600 |
11,200±4,000 |
10,100±4,700 |
0.5 |
Total bilirubin, mg/dL |
0.4-1.5 |
1.6±1.5 |
1.5±1.9 |
0.3 |
Albumin, g/dL |
4.1-5.1 |
2.9±0.8 |
3.1±0.5 |
0.8 |
Prothrombin-time INR |
1.2±0.2 |
1.2±0.3 |
0.7 |
|
Alanine aminotransferase, U/L |
7-23 |
92±147 |
164±531 |
1 |
Alkaline phosphatase, U/L |
106-322 |
505±257 |
609±373 |
0.5 |
Creatinine, mg/dL |
0.46-0.79 |
1.0±0.4 |
0.9±0.4 |
0.6 |
C-reactive protein, mg/L |
0-1.4 |
150±81 |
150±97 |
0.9 |
Procalcitonin, ng/mL |
≤0.05 |
8.3±11.7 |
6.1±13 |
0.9 |
Overall culture positive rate, n (%) |
7 (50) |
19 (51.3) |
0.9 |
|
Positive blood culture, n (%) |
7 (50) |
12 (32.4) |
0.2 |
|
Positive pus culture, n (%) |
- |
17 (45.9) |
- |
|
Isolated bacterial pathogens, % |
||||
Klebsiella pneumoniae |
42.9 |
21.6 |
0.004 |
|
Streptococcus anginosus group |
0 |
10.8 |
<0.001 |
|
Enterococcus spp. |
0 |
8.1 |
0.003 |
|
Escherichia coli |
0 |
5.4 |
0.02 |
|
others |
7.1 |
5.4 |
0.5 |
|
negative culture |
50.0 |
48.7 |
0.9 |
Table 3 Laboratory and bacteriological test results in patients with MPLPs and drainable PLAs
Twenty-seven (53%) patients were treated with antibiotic treatment alone, and the other 24 (47%) patients with a combination of antibiotics and PCD (Table 2). Although all patients with MPLPs were treated without PCD, this conservative approach was successful in 92% (13 of 14) of patients. Treatment outcomes, e.g. length of hospital stay, were similar in the MPLP and drainable PLA groups (Table 2). Only 1 (2%) patient with a huge MPLP required conversion to surgical treatment because of intra-abdominal abscess rupture.16 Forty-nine (96%) patients were discharged alive with resolution of the abscesses. There were 2 (4%) fatalities, and both were attributed to septic multiorgan failure. Fatality rates were not different for the MPLP and drainable PLA groups. Liquefaction of the abscess contents was not observed in all but 1 MPLPs over the ensuing weeks (Figure 3). These abscesses shrunk in response to antibiotic administration, and their cavities were gradually replaced by fibrous granulation.
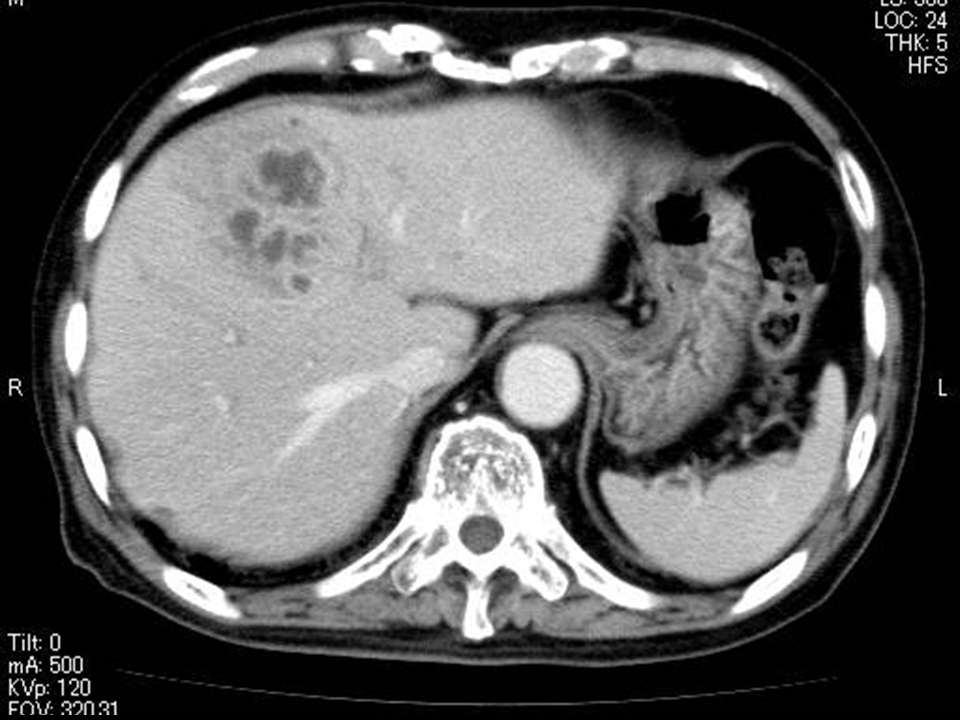
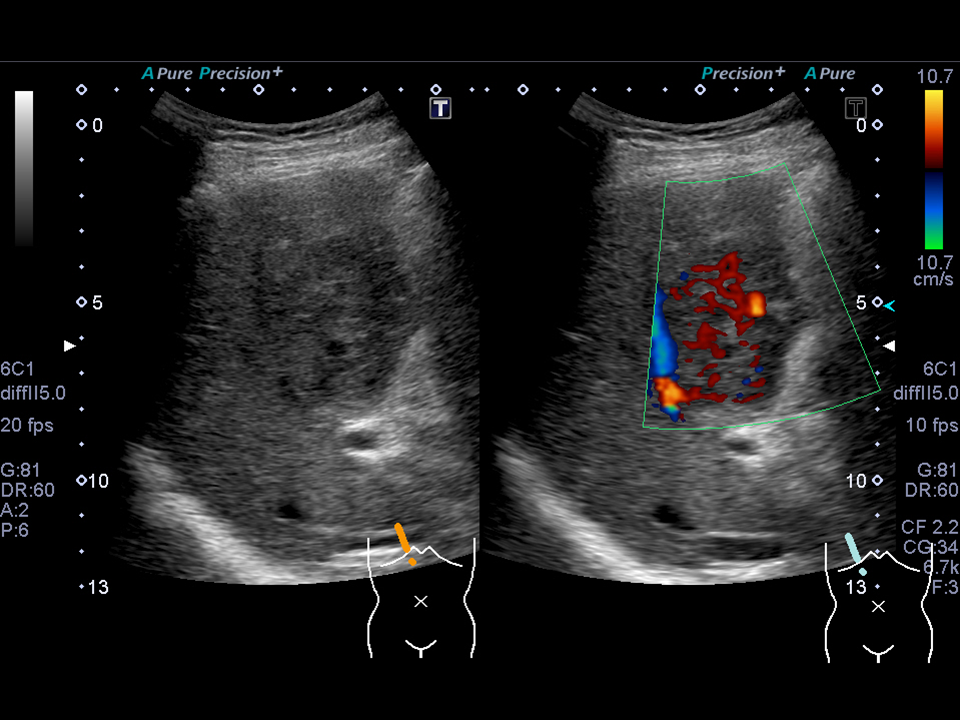
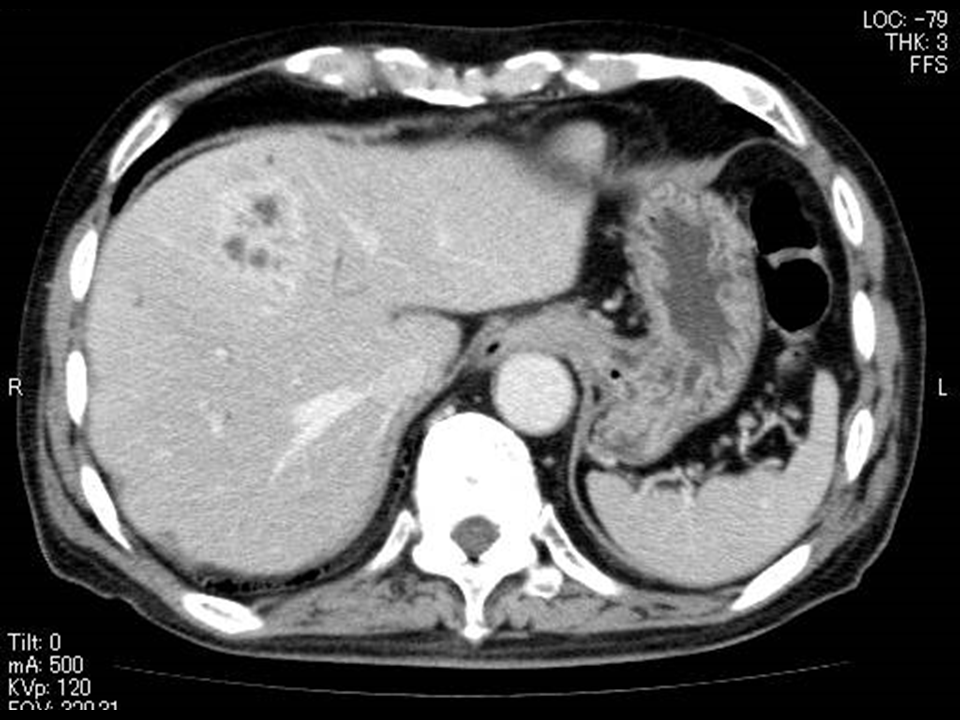
Figure 3 Images of a 75-year-old man showing MPLP changes associated with Klebsiella pneumonia.
A) The image shows contrast-enhanced CT scan, obtained before treatment. The CT scan demonstrates a multilocular abscess with enhancing rim and septa. This finding indicates that the internal septa are maintaining their blood perfusions.
B) Ultrasonography obtained before treatment shows a poorly-liquefied abscess with color Doppler signals in the internal septa, indicating that the septa are maintaining their blood perfusions.
C) A week after the treatment, contrast-enhanced CT scan shows the abscess has decreased in size without lequefaction of the contents.
†MPLP denotes multilocular poorly liquefied pyogenic liver abscess, ‡PLA pyogenic liver abscess.
†INR denotes international normalized ratio.
This study aimed to evaluate MPLPs in terms of clinical and therapeutic characteristics, in comparison with drainable PLAs. Fourteen (27%) patients of our cohort had MPLPs. Patients with MPLPs had significantly higher incidences of Klebsiella pneumoniae infection and cryptogenic source of infection. These findings are in line with prior reports that Klebsiella pneumoniae-associated PLA is often cryptogenic and commonly presents as a single large abscess with predominantly multilocular content.17,18 One of the reasons that Klebsiella pneumoniae infection is often associated with MPLP is speculated as follows. A loss or reduction of capsule synthesis by the pathogenic Klebsiella pneumoniae strain renders an antiphagocytic effect against macrophages and neutrophils,26 and this predisposes the pathogen to invade and destroy the parenchyma rapidly, without allowing enough time for the tissues to break down and liquefy into homogeneous pus.27 These MPLAs were unsuitable for PCD and often managed with antibiotic administration alone. We initially assumed that a MPLP reflected its immature stage and might subsequently shift to a liquefaction process that would make PCD available. However, most abscesses did not liquefy, but simply shrank over the following treatment weeks (Figure 3). The reason why conservative treatment without PCD was generally successful in our cohort with MPLPs can be explained as follows: the internal septa of the MPLPs extended intricately into the abscess cavities, maintaining their blood perfusion, and therefore allowing the antibiotics to penetrate adequately into the abscess (Figures 1 & 3).
Multilocular PLAs larger than 3cm have been previously recommended to be managed with surgical intervention.13 In addition, greater age (≥55years), presence of multiple abscesses, malignancy as etiology, and endoscopic intervention were reported to be the risk factors for the development of PLA treatment failure with antibiotics alone.24 In our cohort, 50 % of MPLPs were larger than 5cm in diameter and 29 % were multiple. Despite these unfavorable factors, the overall fatality rate in our cohort was 4%, which was equal to or better than rates reported in previous studies about PLAs (Table 2).1,2,22,23,28 Fatality rates did not differ between MPLP and drainable PLA groups in our cohort. The new knowledges from this study are: (i) about a quarter of PLAs may be MPLPs, i.e. unsuitable for PCD, (ii) MPLP often results from Klebsiella pneumoniae infection, (iii) the intricate septa of MPLP may maintain their blood flow, (iv) most such MPLPs shrink to disappear without liquefaction following antibiotic treatment, and (v) importantly, the conservative treatment outcomes of MPLPs may not be associated with inferior outcomes.
How to apply these knowledges for routine clinical practice? We recommend: (a) in case of MPLP unsuitable for PCD, it should be carefully examined whether the septa maintain their blood perfusions with imaging studies, (b) the adequate antibiotic penetration into the abscess is essential for a successful conservative treatment, and (c) MPLP that has septa with well-preserved blood flow can often be managed medically without PCD. Of course, prompt decision to proceed with more aggressive management is mandatory in emergency situations. Alkofer et al.,23 determined two situations that lead to surgical PLA management.23 The first situation involves factors that increase the risk of PCD failure: multilocular abscesses, gas-forming abscesses, and septic shock at presentation. The second situation involves factors associated with high mortality: poor physiological state, severe sepsis, pleural effusion, and a malignant PLA origin. Unfortunately, there are few salvage options except for surgical treatment, when medical treatment of MPLP fails.29 Ke et al.,30 suggested preliminary radiofrequency ablation as an alternative to surgical intervention in patients unsuitable for PCD. 30 Kusumoto et al.,31 reported a continuous perfusion drainage procedure in which two tubes were placed to accelerate irrigation and excretion of the abscess contents, respectively.31
Two recent literatures about PLA from China reported that PLA patients treated without PCD were 19.6 % and 10.6 %. Thirty three percent and all of them were surgically drained, respectively.32,33 They also alerted that about 10 % of the patients with Klebsiella pneumoniae-associated PLA had invasive liver abscess syndrome with distant metastasis infection, as compared to only one (0.2 %) of our cohort.16 On the other hand, a report from Spain revealed that 31.6% of patients with PLA were treated with antibiotics alone.34 Unfortunately, these literatures do not mention the loculation of PLAs. There may be geographical difference in the prevailing culprit bacterial strains and their virulence.
This study had several limitations. First, the sample size was small, and this may detract from the statistical strength of our findings. Second, this was a retrospective study based on medical records from routine clinical practice. This limited the ability to provide exhaustive information. Third, our patient group may exhibit regional variations that are not reflected in the general population. Thus, our findings should be validated in a larger population.
In our retrospective cohort analysis of 51 consecutive patients with PLA, 14 (27%) patients had MPLPs, and thus, we need to remember that MPLP is not rare. Klebsiella pneumoniae accounted for the majority of identified pathogens in these patients. MPLPs were managed with antibiotics alone. Despite this conservative treatment strategy, the length of hospital stays and fatality rates of the patients with MPLPs was not inferior to that of the patients with drainable PLAs. The intricate septa of the MPLP maintains its blood perfusion and may have delivered antibiotics adequately into the MPLPs, thus producing good treatment outcomes. Nevertheless, high suspicion of treatment failure is crucial in patients with MPLPs to ensure that management strategies are implemented in a timely manner.
The authors declare that they have no conflict of interest (COI).
We would like to acknowledge the staffs in our Hepatology Department for their assistance: Mitsuki Sumino, Hidemi Nakaida, Rina Nishikawa, Kana Umei, Ayumi Irie, Misaki Higashihata, Ryota Ohshima, and Kayo Sekiguchi.
None.

©2017 Morii, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.