eISSN: 2379-6367


Research Article Volume 10 Issue 6
1School Science, Technology & Environment, Ana G. Méndez University, USA
2Morningside Graduate School of Biomedical Sciences, UMASS Chan Medical School, USA
3Department of Biological Sciences, Toxicology Graduate Program, North Carolina State University, USA
Correspondence: Beatriz Zayas, Ana G. Méndez University, School Science, Technology & Environment, San Juan, Puerto Rico, USA
Received: December 01, 2022 | Published: December 15, 2022
Citation: Acevedo A, Rivera A, Ríos K, et al. Mechanism involved in cell death induced by DEHP (Di-ethylhexyl phthalate) on primary brain cells from Zebrafish (Danio rerio). Pharm Pharmacol Int J. 2022;10(6):218-223. DOI: 10.15406/ppij.2022.10.00390
Human and ecological exposure to chemical contaminants continue to increase given anthropogenic activities and sources. Phthalates are among the most persistent toxic chemicals found in aquatic systems. Phthalates belong to a family of chemical compounds that are heavily used in global manufacturing, distinguished for being able to provide durability and elasticity to plastic products, with poor water solubility and low evaporation. In terms of health impact, these compounds have been shown to breach the placental barrier having effect on human development and neurodegeneration. The main purpose of our study has been to assess the neurotoxicity of phthalates implementing the zebrafish (Danio rerio) model to determine the cell death induction of DEHP on brain cells. For many years, the zebrafish embryonic model has been used to study vertebrate development. Our study, in contrast, used adult zebrafish to resemble mature species that can be continuously exposed to low concentrations of DEHP. To assess the neurotoxicity, brain tissue from zebrafish was extracted via dissection, and primary neural cells dissociated to obtain primary cell subcultures. The neural cells where subsequently exposed to DEHP (Di-ethylhexyl phthalate) at concentrations from 10µM to 100µM for 48 hours to observe cell growth inhibition (GI50). The results indicate neural cell death at a GI50 of 62.9 µM. Apoptosis induction, caspase activation, mitochondrial membrane damage as well as autophagy cell death were analyzed to identify DEHP cell death mechanism at the GI50 concentration. In conclusion, this study clearly demonstrated that DEHP can induce apoptosis cell death on zebrafish neural brain cells in culture through the activation of an intrinsic apoptotic pathways with caspases 3 and 7 activation as well as autophagic pathway and mitochondrial damage. The presence of phthalates as well as other chemical contaminants on aquatic environments needs to be earnestly controlled in order to reduce neurotoxicity to aquatic species as well as human.
Keywords: neurotoxicity, ecotoxicology, phthalates, cell death mechanism, zebrafish, apoptosis
Modern manufacturing practices have relied on plastics to improve the physical properties of consumer goods. In turn, plastic manufacturing technologies have evolved to modify the characteristics of their products. Phthalates are widely used as plasticizers to increase the flexibility and durability of plastic products. Among the most commonly used phthalates in consumer products, are di-(2-ethylhexyl) phthalate (DEHP), diisononyl phthalate (DINP), dibutyl phthalate (DBP), diisodecyl phthalate (DIDP), di-n-octyl phthalate (DnOP), and benzyl butyl phthalate (BBP or BzBP). DEHP being the most used phthalate, which has been widely employed in a variety of items, including construction materials, flooring, pipes, food packaging, and medical devices.1-3 In addition, using microwaves to prepare food or drinks can increase the leaching of phthalates from containers.4 Furthermore, they are frequently released by industrial waste, manufacturing runoff, solid waste, sewage drainage, and the degradation of phthalate-containing items.5
These chemicals are commonly found to have poor water solubility and difficulty evaporating, as well as low to moderate mobility in soils and aquatic environments, allowing them to stay in surrounding environments for prolonged periods of time.6 Individual molecules can spread in a variety of ways after entering the environment; for example, they can bind to dust particles and travel large distances through the air. A considerable portion of particles, however, falls into bodies of water, posing the biggest threat to aquatic life.5
In terms of health impact, these compounds have been shown to breach the placental barrier and have an effect on human development, particularly the reproductive and endocrine systems. Due to its ubiquity in the environment, DEHP has raised concerns pertaining to continuous exposure on the human population and the formation of toxic metabolites such as 2-ethyl-1-hexanol (2EH) and mono-(2-ethylhexyl) phthalate (MEHP) during the degradation of DEHP.7 Some microorganisms that can degrade di-ester plasticizers can also be sources of 2-EH.4
The brain has been determined to be at risk of DEHP exposure. This phthalate can affect neurodevelopment and lead to teratogenic anomalies by disrupting normal fetal brain development, as DEHP can cross the placenta and enter the fetal circulation.8-10 In postnatal rats, post-DEHP exposure was found to be harmful to the development of the hippocampus (large part of the brain that is involved in memory and spatial navigation) in males but not females.11 In addition to rats, DEHP neurotoxicity has been observed in Caenorhabditis elegans, a nematode. DEHP exposure can result in an intracellular accumulation of ROS, which causes neurotoxicity, it can also inhibit the expression of many genes required for AFD sensory neuron differentiation and function (necessary for the thermosensory response).11 Given the high sensitivity of zebrafish for the analysis of novel compounds, as well as a screening model for detecting toxicity and drug efficacy, the zebrafish model is being commonly used as a neurotoxicological model.12-14
Our study uses adult zebrafish brain tissue as an animal model to investigate the cellular events induced by phthalate exposure at levels that could be found in the environment. As a model for determining toxicity, zebrafish has a strong resemblance to humans (~70% of human genes & ~84% associated with human diseases),12,14 making it an ideal model for developmental toxicity, as it demonstrates similarities with the nervous system.13,15 In comparison to other vertebrates, zebrafish have a basic nervous system organization; many neural circuits and cell types relevant to human studies are found in zebrafish. For many years, the zebrafish embryonic model has been used to study vertebrate development.16 Our study, on the other hand, will use the adult zebrafish as a model for human or adult aquatic species that have been continuously exposed to low concentrations of DEHP.13
Based on research that assesses contaminants on aquatic ecosystems in Puerto Rico,16,17 significant levels of contaminants were found in some rivers adjacent to urbanized communities. The study screened certain metals, as well as eight phthalates and revealed the presence of DBP, a phthalate utilized in our previous published study.13 Other studies revealed higher concentration of MEHP metabolites in pregnant women in Puerto Rico, in comparison to the United States population.18 Among other studies, the presence of phthalates that is known to be present in rivers, is also showcased in the air, especially DEHP. According to our previous findings using the zebrafish model13 and given the limited research on the effects of DEHP on the brain, it is clearly significant to evaluate the DEHP neurotoxicity. Expanding our previous research, here we screen different cell death endpoints such as growth inhibition, apoptosis, caspase activation, mitochondrial damage, and autophagy. Results show that even at low DEHP concentrations,19 cell death-related events like apoptosis and autophagy can be observed after 48 hours of continued exposure.
Ethics statement and institutional IBC/ACUC compliance
This study was evaluated by the Animal Care and Use Committee (ACUC) federal regulations and approved by the Universidad Ana G. Mendez System ACUC and Biosafety Committee (IBC) with the approved protocols numbers: IBC protocol number B01-069-22 and ACUC protocol number A01-020-22. The model used for this research was zebrafish (Danio rerio). All the processes and guidelines as recommended by the American Veterinary Medical Association Guidelines for the Euthanasia of Animals: 2013 Edition. Similar guidelines are incorporated in the ARRIVE guidelines.
Zebrafish population care and maintenance
The zebrafish were obtained from the Caribe Fisheries Inc. a tropical aquaculture farm located in Lajas, Puerto Rico. After obtaining the zebrafish, they were transported according to the guidelines established by the ACUC Committee. After arriving at the ChEMTox Lab (UAGM-Cupey), the container filled with zebrafish is placed into the water tank, in order to acclimate the zebrafish population, following the specific requirements. After 15 minutes in the tank, the container filled with zebrafish is opened to release them into the new environment. After 24 hours from arriving at our facility, zebrafish can be used as needed.
Reagents and materials
Stock Solutions: Di-ethylhexyl phthalate (DEHP) (experimental compound), Cisplatin (positive control), and Dimethyl Sulfoxide (DMSO) used as vehicle, were obtained from Sigma Millipore Corp. (St. Louis, MO). A stock concentration of 2 mM DEHP diluted with DMSO was prepared and used to establish the GI50 determination Assay. The positive control, Cisplatin, was prepared at a 3 mM stock solution following the protocol provided by the manufacturer. The concentration applied as positive control was 24 µM. DMSO, as the vehicle was used as negative control. All the solutions were prepared under sterile conditions, labeled, and stored until used.
Cell death assay reagents
The Annexin V kit (APOPAF-20TST) used for apoptosis detection was purchased from Sigma Millipore (St. Louis, MO). The Caspase 3/7 activity assay kit (Caspase-Glo (R) 3/7 Assay 2.5mL) and Mitochondrial ToxGlo™ Assay 10mL were obtained from Promega Corp. (Madison, WI). Both kits were prepared the day of the experiment and stored following the manufacturer’s protocol. For the Autophagy cell death detection assay, Dansylcadaverine was obtained from Sigma Millipore Corp. (St. Louis, MO) and prepared according to the protocol provided by the manufacturer.
Zebrafish brain dissection solutions
The dissection (DMEM F:12) and neurobasal media (GIBCO Neurobasal plus medium) purchased from ATCC and Life Technologies. The media was prepared in a sterile environment using the biological biosafety cabin. The dissection media was prepared using a 50 mL conical tube filled with DMEM F:12 (ATCC) and the addition of 20µL of penicillin-streptomycin (Sigma Millipore). The neural media was prepared using a 15mL conical tube filled with 5mL of neurobasal media (Gibco, Life Technologies) and the addition of 10 µL of primocin (Invivogen). Both media solutions were labeled with the corresponding information and stored until used.
Dissection of adult zebrafish
Prior to dissection, the workbench area is thoroughly cleaned using LPH to sanitize the surface before the dissection takes place and prevent any contamination that can affect the experiment. Zebrafish are maintained in the water tank until all solutions and material are ready for dissection. Once the working area is ready, Zebrafish are gently placed via immersion in a rapid cooling bath to be euthanized by hypothermia. This process takes less than 20 seconds until the loss of orientation and loss of operculum movements. Within 10-20 seconds in cold water, the fish have lost consciousness. Euthanized fish are held with a tweezer, put on a dissection bed (a large petri dish of ice, covered with autoclave paper and parafilm) and immediately decapitated, just below the gills. The fish body is placed in a 75% ethanol bath for disinfection. The head is handled with tweezers and placed in the dissection medium in a petri dish where brain tissue extraction will take place. With scissors, the head is cut open, the tissue surrounding the brain is removed and cleaned until access to the brain and optic nerves are clear. Optic nerves connected to brain should be gently cut. Brain tissue must be cleaned with dissection medium using forceps gently and keeping all brain structures intact. Once isolated, the brain is placed in a 15ml conical tube with 5 ml of clean condition neurobasal medium (Dulbecco’s Modified Eagle Medium: Nutrient Mixture F-12 (DMEM/F12) containing 1% (Penicillin-Streptomycin antibiotic).
Subculture process
Primary brain cells subculturing was implemented as described in.13 Briefly, zebrafish brains located in a 15mL conical tube with 5 mL of clean condition medium (Neurobasal plus medium containing 10μL of primocin). Brain tissues are then centrifuged for 2 minutes at 2.0 rpm and condition medium extracted by gently pipetting with a 5mL pipette, without touching brain tissue. Add 5mL of washing solution to rinse the rest of the condition medium. Centrifuge and remove the washing solution again with a 5mL pipet without touching the tissue. Add 3 mL of digestion solution and incubate the brain tissue at 37°C for 10 min. After incubation, gently dissociate the brain tissue by slowly pipetting up and down 10 times with a 10mL pipette; Incubate brain tissue again at 28°C for 10 minutes and then pipette up and down 10 to 13 times with 5mL pipette; Add 2ml of PBS to remove the digest solution and centrifuge the cell suspension. Carefully decant the supernatant and resuspend the pellet in the washing solution. Pipette up and down with a 1,000μL micropipette. At this stage, verify under the microscope that a single cell suspension has been obtained. Use a syringe (19Gx1 ½ “- 1.1x38mm) to break up residual brain tissue if needed; Centrifuge the cell suspension again, remove the supernatant and resuspend the pellet with 1ml of conditioned medium. Cultures are incubated at 28°C for 24 hours before treating or exposing for toxicity assays.
GI50 determination
For 50% growth inhibition (GI50) determination, zebrafish primary brain cells were cultured (approximately, 10-30,000 cells per well) on a black 96-well plate with a final volume of 200 µL in each well (plates BRAND cell Grade, Sigma Aldrich, MO). Cells were exposed to DEHP from 10 to 100 µM (in triplicates) for 48 hours with respective vehicle and background (Neurobasal media plus cells) controls. Analysis of viability was determined by fluorescence generation (indicative of cell viability) with a FluoStar OMEGA (BMG labtech, Cary, NC) with the 544/590nm filters.
Apoptosis induction (Annexin V)
For apoptosis induction determination, as part of the cell death mechanism, the annexin V assay was implemented. The annexin V assay is used as an indicator of early stages of apoptosis.20 This assay allows for detection of phosphatidylserine (PS) migration to the exterior surface of cells, a hallmark in apoptotic cells. Experimental plates of approximately 10,000 – 30,000 primary brain cells per well were cultured in a black 96-well plate and exposed to controls and experimental compounds for 48 hours at 28ºC with 5% CO2. Once the exposure period was completed, the zebrafish primary neural cells were stained with 2.5 µL of FITC Annexin V Conjugate, 50µL of Annexin Binding Buffer, and 2 µL Propidium Iodide (Sigma, APOAF-20TST). Plate was then incubated for 10 minutes in a plate shaker at 500 rpm at room temperature. After incubation, plates were placed in a FLUOstar OMEGA at a wavelength of 544 nmEx/590 nmEm using a green filter to measure the fluorescence following the protocol provided by the manufacturer. A one-way ANOVA was performed to determine statistically significant results.
Caspase 3/7 activity
For determination of caspases 3/7 activation involved in apoptosis cultures of approximately 10,000 – 30,000 zebrafish primary brain cells per well were cultured in a white 96-well plate and exposed to controls and experimental compounds for 48 hours at 28ºC with 5% CO2. After 48 hours of exposure, 50 µL of the Caspase 3/7 reagent was added to each well. Plates were incubated at room temperature in a dark room for 3 hours following the manufacturer’s protocol. Luminescence was measured at 485 nmEx/520 nmEm in the FLUOstar OMEGA following the specification of the protocol provided by the manufacturer. A one-way ANOVA was performed to determine statistically significant results.
Mitochondrial membrane damage
To determine the mitochondrial membrane damage on zebrafish primary neural cells, the Promega Mitochondrial ToxGlo™ Assay Kit (G8000) was applied. Brain primary cell were exposed to negative control (DMSO), positive control (Cisplatin), and the experimental compound (DEHP) in a white 96-well plate with approximately 10,000-30,000 cells per well. After 48 hours of exposure, 10 µL of the 5X Cytotoxicity Reagent (A mixture of bis-AFF-R110 substrate and an assay buffer) was added to each well, except for the blank. The plate was then placed in a plate shaker for 1 minute at 500 rpm and incubated for 30 minutes at 28ºC with 5% CO2. After the incubation period, the plate was placed in the FLUOstar OMEGA to measure the fluorescence at 485 nmEx/520-530 nmEm. A one-way ANOVA was performed to determine statistically significant results.
Autophagy cell death detection
To determine autophagy as part of the cell death process induced by exposure to DEHP, zebrafish primary brain cells were exposed to monodansylcadaverine (MDC) from Sigma Aldrich was implemented following the protocol provided by the manufacturer. Cultures of approximately 10,000-30,000 cells per well were cultured and exposed to DMSO, Cisplatin, and DEHP in a black 96-well plate for 48 hours and incubated at 28ºC with 5% CO2. After the exposure period, 1.67µL of MDC (autophagy indicator) was added to each well and incubated for 30 minutes at room temperature in a plate shaker at 500 rpm. Plates were then analyzed in the FLUOstar OMEGA (at 355 nmEx/520 nmEm). A one-way ANOVA was performed to determine statistically significant results.
Statistical analysis
The statistical analysis was performed with GraphPad PRISMA v.6. The statistical comparison was completed using the one-way ANOVA with multiple comparisons. For the GI50 Determination Assay, the MARS software data analysis in the FLUOstar OMEGA instrument was used for curve fitting results and the effective concentration dose determination of DEHP.
GI50 determination
The growth inhibition effect of DEHP on zebrafish primary neural cells was determined at 48 hours of exposure using a range concentration from 10 to 100 µM. Each 96-well plate consisted of a blank column (media and cells) and a negative column (media, cells, and DMSO). After the exposure period, the fluorescence analysis was completed and the averaged determined effective concentration of DEHP was 62.9 µM (Figure 1). The GI50 was based on an average of three replicates.
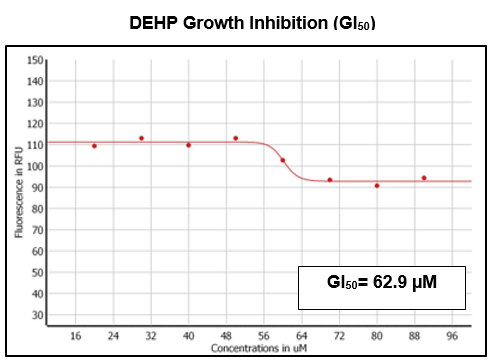
Figure 1 GI50 Determination of DEHP on zebrafish primary brain cells after 48 hours of exposure. An average GI50 of 62.9µM was determined based on three triplicates.
Annexin V
Annexin V assay was implemented as an indicator of zebrafish primary brain cells at an early stage of apoptosis by the detection of phosphatidylserine (PS) migration to the exterior membrane of cells. The zebrafish primary neural cells were treated with controls (DMSO and Cisplatin) and the experimental compound (DEHP) at the GI50 concentration. The fluorescence analysis was completed and the statistical analysis using a one-way ANOVA with the experimental compound DEHP demonstrated a significant difference between the negative and positive controls (Figure 2). Results demonstrate that apoptosis has been activated by exposure to DEHP.
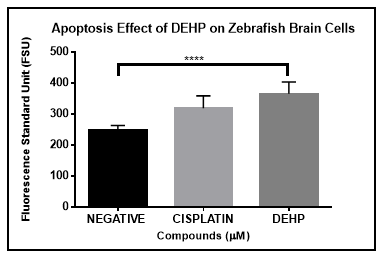
Figure 2 Annexin V Assay on Zebrafish brain primary cells exposed to DEHP after 48 hours. One-way ANOVA analysis demonstrate statistically significant difference (p<0.05) between the negative and experimental compound.
Caspase 3/7 activation
The caspase family plays an important role during apoptosis cell death. The caspases family are classified into two groups: the initiators and effectors caspases. One of our specific aims was to determine the caspase activity, in particular Caspase 3 and Caspase 7 as a way to describe the apoptosis pathway. Results demonstrated that exposure of zebrafish primary neural cells in culture to DEHP for 48 hours resulted in activation of caspases 3/7. Based however on a one-way ANOVA analysis this activation was not significantly different in comparison to the negative control (vehicle). On the other hand, the positive control (Cisplatin) demonstrated significant difference in the induction of caspase activation in zebrafish primary neural cells in comparison to the negative controls (Figure 3).
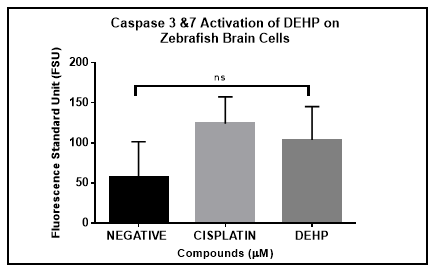
Figure 3 Caspase 3/7 activation on Zebrafish brain primary cells after 48 hours. One-way ANOVA analysis demonstrate statistical significance between (p<0.05) negative control and positive control. Activation was also observed with experimental compound.
Mitochondrial membrane damage
The mitochondria are known as the key organelle for energy generation (ATP) in the cell. When the cell death is mediated by apoptosis, the mitochondrial membrane is disrupted, and all the proteins located inside the mitochondria are released to the cell cytoplasm migrating to other organelles of the cell. Cultures of zebrafish primary neural cells were treated with controls and experimental compound for 48 hours using a 96-well plate. Mitochondrial membrane damage based by the implementation of 5X Cytotoxicity Reagent demonstrated that DEHP exposure strongly disrupted the mitochondrial membrane. This observation is consistent with an apoptosis cell death process. The one-way ANOVA analysis implemented demonstrated that DEHP mitochondrial disruption is significantly stronger compared to the negative control and even more than the positive control, cisplatin (Figure 4).
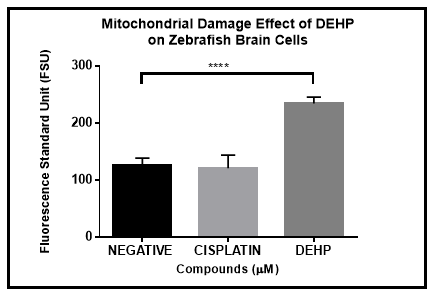
Figure 4 DEHP Mitochondrial Damage induction on zebrafish brain primary cells in culture after 48 hours. Results demonstrated statistical significance by a one-way ANOVA analysis (p<0.05) between DEHP and both negative and positive controls.
More than one cell death process can take place as result of exposure to DEPH. In addition to apoptosis, autophagy is also a key cell death process. In autophagy, the cell death is mediated by lysosomes (lysosomal degradation). Apoptosis however is known as programmed cell death that takes place by activating intracellular proteins. To evaluate the autophagy induction on zebrafish primary neural cells in culture, cells were exposed to the negative (DMSO), and positive (Cisplatin) controls, and DEHP for 48 hours. An indicator of autophagy, Monodansylcadaverine (MDC) is a fluorescent reagent that can accumulate on of vacuoles inside cells undergoing autophagy. After exposure, cells were stained with MDC according to the protocol established by the manufacturer. The results demonstrated that DEHP did show a significant difference in the induction of autophagy compared to the controls using multiple comparisons one-way ANOVA analysis (Figure 5).
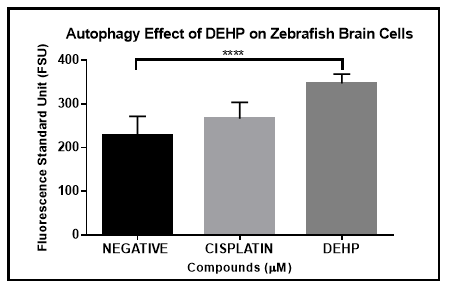
Figure 5 DEHP autophagy induction analysis by Monodansylcadaverine (MDC). Zebrafish brain primary cells in culture were exposed for 48 hours to DEHP and negative and positive controls (vehicle and cisplatin). A one-way Anova was performed and demonstrate the significant difference between DEHP and negative and positive controls.
The main goal of the study was to analyze the neurotoxicity effects of DEHP on zebrafish primary neural cells in culture as well as what types of cell death mechanisms were activated during the exposure period. During the past years, there has been an alarming concern over different types of phthalates, especially DEHP, because it is commonly used in the production of toys, homes, and medical equipment. Also, phthalates are frequently released by industrial waste, manufacturing runoff, solid waste, sewage drainage, and the degradation of phthalate-containing items which magnifies the sources of exposure to such chemicals.
Multiple studies have demonstrated the effect of phthalates on human as well as other species such as rodents. Among phthalates, DEHP is the most used and it is best known as an endocrine disruptor (ED).21 As an endocrine disrupter it can alter the function(s) of the endocrine system and consequently causes adverse health effects in an intact organism, its progeny, or (sub) populations.22
Apoptosis is the programmed cell death most frequently studied because it has an important role in different types of processes, such as normal cell turnover, and chemical-induced cell death.20 Therefore, the analysis of Annexin V is significant because it is designed to detect phosphatidylserine in the membrane when the cell is in early apoptosis. It is significant to mentioned that our results clearly demonstrated that apoptosis induction occurs after 48 hours in zebrafish primary neural cells as it was observed in human neuroblastoma cells23 after 24 hours. Furthermore, apoptosis has two key pathways toward cell death: the intrinsic and the extrinsic pathway. In both pathways, different types of caspases are activated to complete the apoptosis death. Caspases 3 and 7 known as executioners, have a crucial role in the intrinsic pathway, and are used to expand the analysis of apoptosis in chemical-induced cell death research.24 A previous study showed that after 24 hours of exposure to DEHP, the caspase 3 activity increases in rat pheochromocytoma cells.25 In our study, caspases 3 & 7 activity was observed (Figure 3) however it was not significantly different compared to the negative control. This low significance can be explained by the variability of the primary cultures that resulted from brain isolated by multiple adult zebrafish and not an established cell line.
In terms of mitochondrial damage, when cells are exposed to stress, they are led to the accumulation of dysfunctional mitochondria, and they are more prone to mitochondrial membrane permeabilization and apoptosis. Therefore, when apoptosis is induced, the pro-autophagic proteins are inactivated and the inhibition of autophagy is promoted. But when apoptosis is not induced and the accumulation of dysfunctional mitochondria is elevated, the mitochondria can control the autophagic induction.26 In our study, we observed that after 48 hours of exposure to DEHP, mitochondrial damage in zebrafish primary neural cells was evident. This observation is consistent with a significant autophagy cell death activation when compared to the apoptosis induction. Following the previous statement, studies, have shown that DEHP induces autophagy in mouse GC-1 spg cells via oxidative stress.27
As we know, the mitochondria play an important role during the energy production and cell death events as autophagy and apoptosis. In a study realized by Ko et al.28, using adult male mice to prove that DEHP exposure in prenatal stages could be affecting neurobehavior and recognition memory, in particular the cells of the hippocampus and to prove the possibility that the cells located in this important region of the brain are affected by oxidative damage. The hippocampus is considered one of the most important areas of the brain and is related with learning and memory processes. When the mitochondrial membrane disruption occurs, the organelle could be increasing the oxidative damage in macromolecules such as DNA, proteins, lipids and see an increase in ROS (Reactive Oxygen Species) production. To identify the oxidative damage in the hippocampal cells, they used two DNA markers: 8-OHdG and TG, each marker has the advantage of labeling the OH- radicals. These markers are identifying the affected cells of the hippocampus. They conclude and support the hypothesis established by Hamilton et al.29, the long exposure of DEHP affect the cells and some areas of the hippocampus increase the neurodegeneration and promote neurological diseases such as Alzheimer and Parkinson.
In other studies, involving neurodegenerative diseases like Alzheimer’s and Parkinson’s Disease, mitochondria and oxidative stress are known to play a key role in the pathogenesis of these afflictions.30 Accumulation of damaged mitochondria brings about an activation of mitophagy, to degrade the dysfunctional organelles and improve synaptic and cognitive functions. However, it’s been found that in neurodegenerative diseases as these mentioned, there’s a decrease in mitophagy activity.30 These studies provide evidence that dysfunctional mitochondria and defective mitophagy contribute to degeneration of vulnerable genes in the brain, leading to these types of diseases in humans, as shown in the utilized mouse models.30,31 Certain environmental contaminants, such as metals and phthalates have been discovered to be involved in linking oxidative stress to Alzheimer’s disease, the findings have associated DEHP marking an increase in oxidative stress and neurotoxic effect in C. elegans, resulting in pathological signs pertaining to AD.32 These statements relate to our study, where mitochondria membrane permeabilization was significantly induced by DEHP (Figure 4), as such, this can indicate a degeneration to the development of the nervous system in the utilized zebrafish neural cells. This can serve as a further connection from the zebrafish model to the human nervous system, to better understand the phthalates effects on the human nervous system.
In conclusion, our study demonstrates toxicity of DEHP after 48 hours exposure on zebrafish primary cells in culture involving activation of both cell death mechanism apoptosis and autophagy as well as mitochondrial damage. These results support the necessity to better control the liberation of phthalates to water systems that can impact multiple ecosystems, as well as the nervous system of humans.
The authors thank Gloria Carrasquillo, MSEM, Miguel Calderón and Nicole Dávila former members from the ChEMTox Lab for their help and support. We also want to thank Dr. Carlos A. Rosado-Berrios from Texas A& M for technical advice in this process. This research was funded by the National Science Foundation under the NSF sub-award 2018-00-1109 and by the PR-INBRE award, National Institute of Health (NIH) Grant No. 5P20GM103475-19 to the Biotesting CORE Laboratory at University Ana G. Méndez, Cupey Campus in San Juan, Puerto Rico.
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.

©2022 Acevedo, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.