eISSN: 2379-6367


Review Article Volume 6 Issue 3
Chemistry Department, Faculty of Science, Al Baha University, Saudi Arabia
Correspondence: Loutfy H Madkour, Department of Chemistry, Faculty of Science, Baljarashi, Al Baha University, Al Baha, Saudi Arabia, Tel 966 533899075
Received: April 30, 2017 | Published: May 8, 2018
Citation: Madkour LH. Applications of gold nanoparticles in medicine and therapy. Pharm Pharmacol Int J. 2018;6(3):157-174. DOI: 10.15406/ppij.2018.06.00172
The stability and dispersity of AuNMs in solution play a key role for the many applications. Most inorganic nanomaterials are not well dispersed in physiological buffers and require function-alization by thiols or surfactants to offer the stabilization forces. Furthermore, sufficient blood circulation time is critical for both imaging and in vivo drug delivery. Localized surface Plasmon resonance (LSPR) is one of the most significant features of AuNMs. The AuNMs as reporters have been broadly applied into lateral flow immunechromatographicalassay (LFICA) and enzyme-linked immunosorbent assay (ELISA), which is a well-established technology for analysis of the target analytes in food safety, clinical diagnosis, environmental monitoring, and medical science and so on. Au based nanomaterials (AuNMs) are known to possess many attractive features such as unique electrical, optical and catalytic properties as well as excellent biocompatibility. In this review, we summarize the current advancement on application of AuNMs in analytical sciences based on their local surface plasmon resonance, fluorescence and electrochemistry properties. AuNMs based imaging and therapy in biomolecules is explained. As one of the most reliable imaging modes, computed tomography (CT), X-ray and SERS imaging has been widely used owing to its high spatial and density resolution. We end the review by a discussion of the conjugation between gold nanoparticles with other kinds of nanoparticles such as other metals and carbon nano structures. Finally, future development in this research area is also prospected.
In 2017 Madkour LH1 published a vision for life sciences: interfaces between nano-electronic and biological systems in order to the prevention and treatment of disease in the human body. In this article, we review the applications of gold nanoparticles in medicine. Generally, the recognition elements, which are applied into colorimetric sensors based on AuNMs, are categorized as follows:
Furthermore, some novel techniques have been tried to improve the selectivity of colorimetric sensing, such as click-chemistry-based assay. Physics, chemistry and biology of Au based nanomaterials (AuNMs) have emerged as a broad and new sub discipline in the community of colloids and surfaces. The specific size and shape dependent physiochemical properties and remarkable bio/chemical inertness of AuNMs have made themselves the ideal candidates for both fundamental and technical study including crystal growth, electron-transfer mechanism, localized electro-magnetic theory, catalysis, DNA assay, bio-imaging and therapy, and so on.2 Among those rich properties, the optical characteristics originated from the giant electromagnetic field near the surface of AuNMs are particularly intriguing and thus broadly applied in analytical science, e.g. colorimetric assay, surface enhanced Raman spectroscopy (SERS) and surface Plasmon resonance (SPR) spectroscopy, as well as bio-imaging. Another interesting optical property of AuNMs is that fluorescence appears with their size shrinkage to below 2 nm, which allows development of luminescence-based analysis. Also, electrochemical (EC) sensors could be constructed based on the redox feature of Au NMs (Figure 1). Analytical science has been playing a primary role in our daily life, for instance, food safety control,3 biomedical diagnosis,4 medicolegal appraisement,5 anti-terrorism alert,6 and environmental pollution monitoring.7 Efficient detection technologies depend greatly on the analytical method in terms of analysis time, signal-to-noise (S/N) ratio, selectivity, and limit of detection (LOD).8 Therefore, a great of efforts should be paid to develop new analytical methods with the advantages of rapid determination, high analysis flux, satisfactory sensitivity and good robustness. Thanks to the above-mentioned optical and electrochemical features, many functionalized AuNMs have been successfully synthesized9–14 for improving performances of analytical techniques. (Figure 1)
Small organic molecules and metal ions
For the sensitive detection of small organic molecules, the AuNMs play an important role in improving sensitivity of the EC sensors. Especially, AuNMs composited with conducting polymers or carbon nanomaterials have been used to increase electro-chemical efficiency. It deserves noting that the decorated carbon nanomaterials with metal NPs are of special interest due to the integrated property of two components with better catalytic activity and enhanced electrical conductivity.15,16 Zhu et al.17 fabricated EC sensors by using spherical AuNPs combined with conducting polymer (poly-[2, 5-di-(2- thienyl)-1H-pyrrole-1-(p-benzoic acid)]).17 To further improve electron transfer, synergistic effect of multiple nano=composite layers, namely, chitosan-AuNPs, graphene-AuNPs and multi-walled carbon nanotubes (MWCNTs)-cobalt phthalocyanine, was exploited by Sun et al.18 Roushani et al.19 fabricated a sensitive and selective EC aptasensorfor detection of cocaine based on the conformational change of the aptamer-functionalized AuNPs onto CNTs-based nanocomposite. Detection of organic small molecules and metal ions is very important for controlling food quality, environmental protection and antiterrorism. In order to realize detection of trace amount of analytes at extremely low concentrations, various efforts for increasing the sensitivity of SPR biosensors have been paid. Most of these methods are based on changing the refractive index at the sensor surfaces by means of a variety of (bio) chemical or NP agents, which are captured by the sensor surfaces subsequent to the binding of the target analyte to the primary biorecognition elements. These methods include the use of secondary and tertiary antibodies, antibodies labeled with enzymes, and dielectric or metallic NPs.20 In particular; AuNMs of diameters ranging from 5 to 40nm have been widely used to enhance the response of SPR biosensors.21,22
Metal ions
Some EC sensor methods based on AuNMs have been developed for detection of heavy metal ions. For examples, Shen et al.23 proposed an EC DNAzyme sensor based on AuNPs for sensitive and selective detection of Pb2+. A label-free EC sensor was developed for the highly sensitive and selective detection of Hg2+ by Tang et al.24 In their study, the catalytic HAuCl4/NH2OH reaction was utilized for formation of AuNPs as signal reporter after capturing Hg2+on the modified electrode surface through the specific thymine-Hg2+-thymine (T-Hg2+-T) coordination. Compared with the traditional metal NPs-based method, this sensor avoided the labeling of the DNA probe with NP tags, and only one unlabeled T-rich DNA sequence was needed, which greatly reduced the cost and simplified the sensing procedure. Thus, a LOD as low as 0.06nM could be obtained for Hg2+. Furthermore, in 2016, Wang et al.25 presented a sensitive, selective and reusable EC biosensor for determination of Hg2+ based on thymine modified AuNPs/reduced graphene oxide nanocomposites (Figure 2).25 Notably, the developed EC sensors afforded excellent selectivity for Hg2+against other heavy metal ions including Zn2+, Cd2+, Pb2+, Cu2+, Ni2+ and Co2+.Wang et al.26 prepared an EC sensor based on graphene and AuNPs for detection of trace Cu2+ with the anodic stripping voltammetry analysis.26 Yang et al.27 constructed a highly sensitive electro-chemical DNA biosensor made of polyaniline (PANI) and AuNPs nanocomposite (AuNPs@PANI) for detection of trace concentration of Ag+.27 In the presence of Ag+, with interaction of cytosine-Ag+-cytosine (C-Ag+-C), cytosine-rich DNA sequence immobilized onto the surface of AuNPs@PANI had a self-hybridization and then formed a duplex-like structure. The prepared EC sensors could detect Ag+ at a wide linear range of 0.01–100nM with a LOD of 10pM (Figure 2).
Traditionally, the tri-sodium citrate-based thermal reduction has been broadly recognized as a universal approach for preparing spherical Au nanoparticles (AuNPs) of varied sizes ranging from 15 to 147nm possessing strong SPR signals.28 Although this preparation method simply involves controlling the ratios of citrate to Au ions,29 the reaction often needs to be carried out on a relatively large scale at the temperature of boiling water and lacks the reproducible preparation of uniform AuNPs with rather large sizes. After the breakthroughs reported by Schmid30,31 and Brust et al.32, a variety of methods have been developed to synthesize AuNMs, and many excellent reviews33 are available now. In general, the reported synthesis methods of spherical AuNMs can be categorized as followed:
In addition, non-spherical nanoparticles (NPs) such as Au nanorods (AuNRs),35 Au nanocages,36 Au cubes,37 Au triangular prisms,38 as well as other unconventional structures like hollow tubes and even branched nanocrystals39 have attracted significant research attention in the past two decades, since the properties of AuNMs are known to be strongly dependent on the size and shape of the particle.40 Recently, a lot of excellent reviews have summarized how to achieve shape and size-defined AuNMs with different techniques including solve thermal synthesis, seed-mediated growth, homogeneous nucleation and electrochemical method.41,42 In this review, we mainly introduce the latest and important advancement of synthesis of the AuNMs, especially on how to control the morphology of AuNMs. A great of efforts have been paid to achieve high-quality and shape-controlled AuNMs for the purpose of highly-sensitive analysis. Unfortunately, most of the synthetic methods are empirical, and understanding their growth mechanism remains a challenging task. Interestingly, introduction of Ag+ in growth solutions significantly improves the yield of AuNRs, and Yang et al.43 prepared AuNRs with controlled aspect ratio by using photochemistry in the presence of Ag+.43 This process itself was highly promising for producing uniform AuNRs, and more importantly it was useful in deciphering the growth mechanism of anisotropic AuNPs due to its simplicity and the relatively slow growth rate of the AuNRs.
Later, a versatile seed-mediated growth method was reported for selective synthesis of single crystalline rhombic dodecahedral, octahedral, and cubic AuNMs.44 These results might provide a basis for gaining mechanistic insight into the growth of shape-and structure-controlled noble metal nanomaterials. An even more complex sample is nanostar, which is characteristic of uneven arms with different lengths. To fully implement the advantageous property of Au nanostars, a precise control over their symmetry and uniformity is highly desired. In 2015, Niu et al.45 obtained Au nanostars with excellent symmetry control by using a solution-phase method in high yield and good monodispersity.45 In their study, icosahedral seeds were used to dictate the growth of the evenly distributed arms in an Ih symmetric manner. Benefiting from their high symmetry, the Au nanostars exhibited much better single-particle SERS performance compared with asymmetric Au nanostars, in terms of both intensity and reproducibility. Apart from large-sized Au particles, Au nanoclusters (AuNCs) with the sizes of less than 2nm formed in dendrimers displays visible luminescence of high quantum yield (QY).46 Ying et al.47 reported a simple, one-pot and “green” synthetic route, based on the reduction and stabilization capability of bovine serum albumin (BSA) proteins. The AuNCs prepared at the physiological temperature (37oC) showed red emission with a QY of ∼6%.47
In order to further control the luminescence property, synthesis of atomically precise AuNCs has ignited numerous research efforts.48,49 In 2014, Yu et al.50 synthesized Au22 (glutathione)18 clusters of the emission peak at ∼665nm with a QY of ∼8%. Upon comparison of their luminescence properties with those of Au (I)-thiolate complexes, the enhanced luminescence of Au22 (SG)18 was ascribed to aggregation-induced emission. Nevertheless, the origin of emission in AuNCs still keeps unclear, posing a major hurdle for development of highly luminescent clusters. In 2015, Pyo et al.51 prepared Au22 (glutathione)18 clusters with a luminescence QY greater than 60% by rigidifying their Au shell with tetraoctylammonium cations.51 This study presents an effective strategy to enhance the luminescence efficiencies of AuNCs by optimizing the shell structure. In order to precisely tune the size and shape of AuNMs, DNA is recently adopted as a powerful programmable tool to realize above goals. Lu’s group systematically summarized morphology controlling, spatial positioning and dynamic assembly of AuNMs with DNA as a powerful tool.52 In some details, their research results demonstrated that DNA could be used to control the morphologies of AuNMs during seed-mediated growth.53 They also developed a novel method based on DNA-encoded tuning to achieve AuNMs from AuNR seeds with controlled geometric and plasmonic properties.54 Furthermore, they suggested that the DNA molecules played important roles via influencing diffusion of the Au precursors to the seed, and therefore the AuNMs growth might be modulated through difference in DNA desorption, density and mobility on the seed surfaces.55
These insights into the mechanism of DNA-guided AuNMs synthesis not only provide deep understanding of the interactions between the DNA and nanomaterials but also allow better control of the shapes and surface properties of many nanomaterials. Ye et al.56 designed a millifluidic reactor to synthesize a wide variety of AuNMs at high concentrations.56 The synthesized method based on millifluidic reactors inspired other researchers to use the analogous reactors for possible mechanism and application study. Recently, Krishna et al.57 demonstrated adoption of a simple millifluidic chip for in situ analysis of morphology and dimension-controlled growth of Aunano and micro-structures with a time resolution of 5ms (Figure 3).57 See from Figure 3, gold structures with ∼3µm in diameter that had corrugated metal-like shapes were formed after a time interval of 5h (Figure 3D). When the time of flow was increased to 9h, these structures further transformed into micro-hemispherical ones (Figure 3C). This study offers a feasible way to obtain dimension-and morphology-controlled AuNMs in a high yield (Figure 3). Noteworthy, some novel synthesis strategies, such as biosynthesis,58,59 2D immiscible oil/water interface,60 levitated leidenfrost drop,61 sunlight-induced synthesis62 and so on, have been reported, and such green and economic synthesis methods for preparation of uniform and controlled AuNMs provide the opportunity for development of low-cost and sustainable analytical techniques. However, the above technologies need to be further improved and standardized.
Owing to the large surface to volume ratio, modification and functionalization of AuNMs surfaces with biomolecules, DNA or chemicals becomes imperative for their application in analytical science. It is well known that free AuNMs have high surface energy and tend to aggregate and fuse. As a result, the intriguing properties observed for the AuNPs would disappear, and the difficulty arises for long term storage, processing, and applications. Therefore, great efforts have been devoted to develop novel strategies to stabilize AuNMs,63 and the most common approach is to coat AuNMs with either organic or inorganic shells. These shells not only endow AuNPs with high stability but also offer them additional functionalities. As an example, in addition to good stability and biocompatibility, the mesoporous silica shells that are currently broadly used have high surface area and tunable pore size and volume, which can accommodate analytes and drug molecules.64
AuNMs have been widely used in SERS based immunoassays of biomolecules such as DNA, protein and cell. However, challenges still remain with amplification of SERS signals due to the extremely small cross-section of Raman scattering.65 Yan et al. intro-duced a new strategy based on nano rolling-circle amplification (nanoRCA) and nano hyper branched rolling circle amplification(nanoHRCA) to increase “hot spot” groups for protein microarrays.66 Li et al.67 constructed a SERS immune sensor for detection of biomarkers.67 In their study, the capture antibody was immobilized on the Au triangle nanoarray chip, while the detection antibody was conjugated with the SERS probe (Au@Ramanreporter@SiO2 sandwich NP). The antigen (analyte) was sandwiched between the capture antibody and the detection antibody. Under light excitation, a lot of “hot spots” were created between the Au triangles in nanoarray, and thus the developed SERS immunosensor could be used for sensitive biomarker detection. Luo et al.68 reported a facile immunoassay for porcine circovirus type 2 (PCV2) based on SERS using multi-branched AuNPs (mb-AuNPs) as substrates. The mb-AuNPs in the immunosensor act as Raman reporters, which were prepared via Tris base-induced reduction and subsequent reaction with p-mercaptobenzoic acid. The modified mb-AuNPs were covalently conjugated to the mono-clonal antibody (McAb) against the PCV2 cap protein to form SERS immuno nanoprobes. These were captured in a microtiterplate via an immuno reaction in the presence of target antigens. Compared to conventional detecting methods such as those based on PCR, the method was demonstrated to be rapid, facile and very sensitive. Tang et al. developed a simple and reproducible SERS chip for highly sensitive and selective screening of active ricin in complex matrices.69 They fabricated single strand oligodeoxynucleotides modified AuNPs in order to amplify recognition and reaction by virtue of their cooperative binding property. To improve sensitivity of this method, a Ag nanoshell was deposited on post-reacted poly (21dA)-AuNPs, which lowered LOD to 8.9 ng mL−1.
The utilization of SERS-based techniques in targeting specific DNA and RNA sequences is generally realized by combination with AuNMs and Raman-active molecules.70,71 Wang et al.72 raised a concept to use enzyme controlled plasmonic coupling as SERS nanosensors for DNA demethylation.72 The nanosensors were constructed by decorating AuNPs with Raman reporters and hemimethylated DNA probes. The enzymatic degradation of DNA substrate probes was utilized to induce aggregation of AuNPs, so the reproducible and sensitive SERS signals were achieved from biological recognition events. Development of SERS-barcoded particles, which contain multiple Raman-active molecules and are insensitive to photo bleaching, is particularly intriguing for high-throughput and multiplex DNA screening.73 Recently, Chenet al.74 prepared a SERS-barcoded nanosensor to detect Bacillusthuringiensis (Bt) gene transformed rice expressing insecticidal proteins.74 The barcoded sensor was designed by encapsulation of AuNPs with silica and conjugation of oligonucleotide strands for targeting DNA strands. The transition between the cry1A (b) and cry1A(c) fusion gene sequence was used to construct a specific SERS-based detection method with a LOD of 0.1pg/mL. In order to build the determination models to screen transgene, a series mixture of Bt rice and normal rice were prepared for SERS assay, and the LOD was 0.1% (w/w) was explored for transgenic Bt rice relative to normal rice. The sensitivity and accuracy of the SERS-based assay were comparable with real-time PCR. Recently, Ye et al.75 proposed an asymmetric signal amplification method for simultaneously detecting multiple biomarkers with significantly different levels.75 The biobarcode consisted of a large number of signals DNA (Cy3-DNA for Cy3-bio barcode or Rox-DNA for Rox-biobarcode) and capture DNA immobilized onto the AuNPs. The AuNPs simultaneously act both as a Raman-signal-enhancing substrate and a Raman signal carrier. Using these bi-functional probes, a linear amplification mode was obtained with high-concentration markers, whereas quadratic amplification mode was responsible to low-concentration markers.
Detection of circulating tumor cells (CTCs) in the blood of cancer patients is significant for early cancer diagnosis, cancer prognosis, evaluation of the treatment effect of chemotherapy drugs, and choice of cancer treatment options.76 In 2011, Wang et al.77 introduced SERS technology for direct detection of target CTCs in human peripheral blood. The LOD of this method ranged from 5 to 50CTCs in 1mL of blood.77 Recently, Wu et al.78 fabricated SERSNPs for direct detection of CTCs in the blood with excellent specificity and high sensitivity.78 The AuNPs were encoded with a Raman reporter molecule, 4-mercaptobenzoic acid (4-MBA), and then functionalized with reductive bovine serum albumin (rBSA) to stabilize the 4-MBA-encoded AuNPs (AuNP-MBA) and decrease the nonspecific interaction with blood cells. Then AuNP-MBA-rBSA-FA composite NPs were constructed with a targeted ligand folic acid (FA) (Figure 4A) (Figure 4B). The FA on the surface of AuNP-MBA-rBSA-FA NPs was recognized by CTCs, which over expressed folate receptor alpha (FRα). The protection layer of rBSA was much thinner than that of the reported PEG, resulting in a stronger SERS signal (Figure 4C). (Figure 4) For detection of the biological macromolecules such as bacteria pathogen and enzyme, SPR with the advantage of label free provides a powerful platform.79,80 There are many successful examples of SPR sensing as a medical diagnostic tool, which have been reported for biomarkers, pathogen detection and hormone analysis with high sensitivity. For example, Mc Phillips et al.81 employed aligned Au nanotube arrays to strengthen performance of refractive index sensors in biomolecular binding reactions.81 Generally, sandwich82 and competitive or inhibition assay83 are two major detection approaches in SPR biosensor. In 2012, Fernandez et al.84 demonstrated that AuNMs combined with sandwich assays could improve detection sensitivity and selectivity of protein detection.84 Jung et al.85 introduced a strategy for enhancement of SPR signals by adopting AuNPs and a SiO2layer on a Au surface. The modified surfaces showed significant changes in SPR signal when biomolecules were attached to the surface as compared with an unmodified Au surface. The LOD of AuNPs immobilized on a SPR chip was 0.1ngmL−1 for the prostate specific antigen (PSA), a cancer marker. The enhanced shift of the absorption curve resulted from coupling of the surface and particle plasmon by the SiO2 layer and the AuNPs on the gold surface. Now, there were a lot of reports of SPR sensors based on spherical AuNMs for detection of biomolecules.86,87 The anti-E.coliO157:H7 polyclonal antibodies (pAb) were labeled with AuNPs, which were used as secondary antibodies. The E.coli O157:H7 was detected using direct assay and enhancing sandwich assay based on the two channels SPR biosensor. By introducing AuNPs-PAb compound, the LOD was determined to be 10cfu/mL. The sensitivity was 100 times higher than that of direct detection.88 Compared with spherical AuNMs, AuNRs labels are more favorable for the Au-amplified SPR biosensors because of the tunable longitudinal plasmonic peak enables an effective plasmonic coupling between sensing film and NPs. Law et al.89 revealed the potential of applying this “perfectly matched” nanotag in a well-established SPR sensing system and immunoassay. Through detection of tumor necrosis factor alpha antigen, 40-fold sensitivity enhancement using wavelength-matched AuNRs was observed.89 Recently, Xiang et al.90 developed a SPR DNA biosensor array based on target-primed rolling circle amplification (RCA) for isothermal and rapid detection of two pathogenic mycobacteria, mycobacterium tuberculosis complex (MTBC) and mycobacterium avium complex (MAC) (Figure 5). AuNPs were directly assembled on to the surface of the sensor chip via hexanedithiol (HDT) for enhancement of sensitivity as a label-free detection system. Experimental results showed that the signal enhancement by the target-primed RCA together with AuNPs-embedded surface caused at least 10-fold increased sensitivity as compared with conventional RCA on bare SPR chip method (Figure 5).
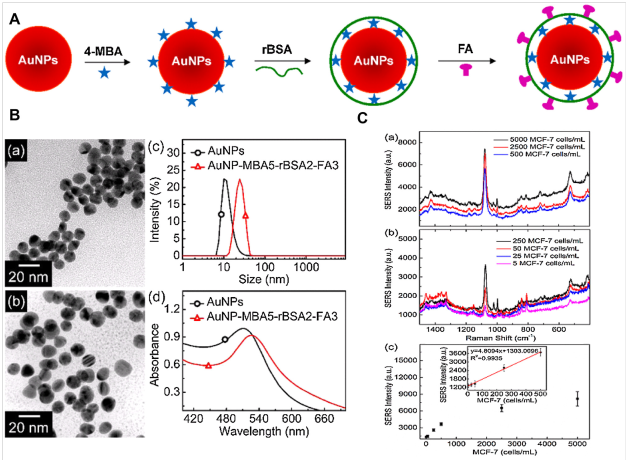
Figure 4 (A) Schematic Illustration for the Design of SERS Nanoparticles. (B) Characterization of the AuNPs (control) and SERS nanoparticles. (a) TEM images of AuNPs. (b) AuNP-MBA5-rBSA2-FA3. (c) Size distributions of AuNPs and AuNP-MBA5-rBSA2-FA3 in Milli-Q water at room temperature. (d) UV-vis spectra of AuNPs and AuNP-MBA5-rBSA2-FA3. (C) Detection sensitivity of the AuNP-MBA5-rBSA2-FA3 nanoparticles for MCF-7 cells in the rabbit blood.78
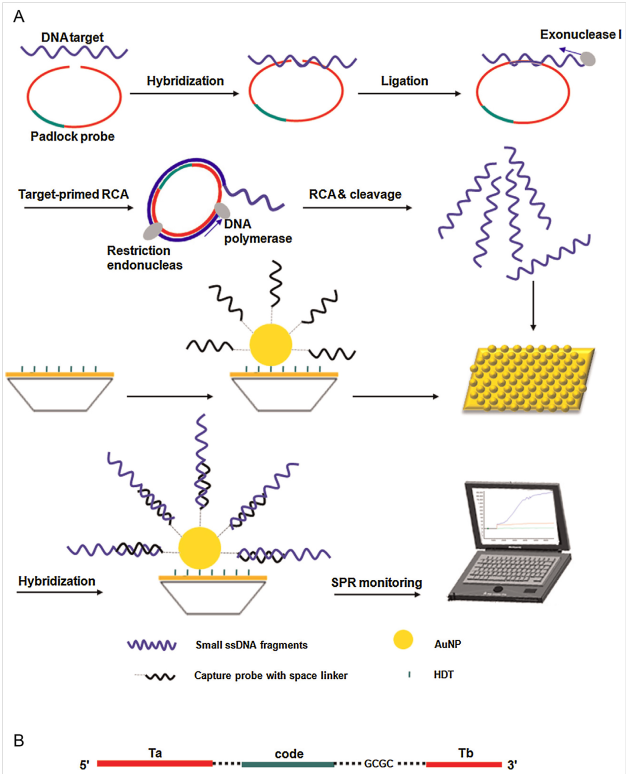
Figure 5 (A) Principle of target-primed RCA-cleavage reaction-based AuNP-embedded SPR assay. (B) Mode of the padlock probe. Ta and Tb are asymmetric target complementary regions in padlock probe. Each padlock probe contains a unique code sequence for multiplex hybridization. The dotted line represents the bases in the linker sequence.90
Besides, the chemical vapor deposition (CVD) method has been adopted by Ruoff’s group for large scale preparation of high-quality graphene sheets.91 The strategy based on AuNMs combined with graphene sheets could improve the conductivity of SPR.92 In 2013, Zhang et al.93 employed Au-graphene oxide composite inthe wavelength modulation SPR biosensor for rabbit IgG detection.93 In their study, the staphylococcal protein A (SPA) modified Au-graphene oxide composite was directly immobilized onto SPR chips without any additional chemical treatment. The biosensor with the SPA modified Au-graphene oxide composite as the enhanced sensing platform exhibited a satisfactory response to rabbit IgG. The LOD obtained with the composite was 16 times lower than that obtained with the SPA modified chip. This study provided a simple and effective approach for fabrication of sensitive SPR immunosensors and extended the application of the Au-graphene oxide composite in immunoassays. Molecularly imprinted polymers (MIPs) have become a competitive tool in the field of molecular recognition, owing to their low cost, physical robustness, thermal stability and easy preparation over biological receptors and other functionalized materials.3 High surface-to-volume ratio makes AuNPs a competitive candidate as matrix material for novel nano sized MIPs. Xie et al. showed a surface molecular self-assembly strategy for MIP in electro polymerized aminothiophenol (ATP) membranes at the AuNP-modified glass carbon electrode for electrochemical detection of pesticidechlorpyrifos.94 Yu et al.95 constructed an EC sensor for determination of dopamine based on core-shell composite of AuNPs andSiO2MIPs through sol- gel technique.95 Similarly, Xue et al.96 prepared a highly sensitive and selective biomimetic EC sensor for amperometric detection of trace dopamine (DA) in human serums by AuNPs doped MIPs (Figure 6). The LOD of the proposed EC sensor for DA was 7.8 nmol/L.96 Sun et al.97 presented an EC sensor for detection of 3-chloro-1, 2-propandiol (3-MCPD) that was usually used as surfactant. They fabricated an AuNPs-modified glassy carbon electrode coated with an MIP film via electro polymerization. The LOD reached 3.8×10−18mol/L. impressively, the sensor showed high sensitivity, good selectivity, excellent reproducibility and stability during the quantitative determination of 3-MCPD (Figure 6).
Ultrasensitive detection of biomolecules (especially for disease markers) is very important for early disease diagnosis. To realize ultrasensitive detection of biomolecules, development of novel and robust detection methods for signal amplification of EC sensors is essential. In past work, a lot of photo or electro-active indicators, such as ferrocene,98 methylene blue99 and enzymes,100 were immobilized onto AuNPs to realize the detection signals amplification. Zheng et al.101 prepared an ultrasensitive EC sensor based on network-like thiocyanuric acid/AuNPs for detection of thrombin.101 Zhao et al.102 fabricated an ultrasensitive and highly specific EC aptasensor for thrombin based on amplification of aptamer-AuNPs-horseradish peroxidase conjugates.102 He et al.103 reported a label-free and sensitive signal-on electrochemical assay for MTase quantification and activity analysis using AuNP samplification.103 In 2016, an ultrasensitive DNA biosensor was developed through in situ labeling of electroactive melamine-Cu2+ complex (Mel-Cu2+) on the end of hairpin-like probe using AuNPs as the signal amplification platform by our group.104 In this work, the highly conductive AuNPs were applied as the loading platform of the Mel-Cu2+ tags. The proposed biosensor showed high selectivity, wide linear range and low LOD for target DNA. Except for the aforementioned amplification strategies, rolling circle amplification (RCA) is one of the most popular and important amplification strategies. Therefore, the dual amplification strategies combined RCA with DNA-AuNPs probe could tremendously improve the sensitivity of bacterial detection. Zhu et al.105 reported an electrochemical sensing strategy for ultrasensitive and rapid detection of Salmonella by combining the RCA with DNA-AuNPs probe.105 As shown in Figure 7, the target DNA could be specifically captured by probe 1 on the sensing interface. Then the circularization mixture was added to form a typical sandwich structure. In the presence of dNTPs and phi29 DNA polymerase, the RCA was initiated to produce micrometer-long single-strand DNA. Finally, the detection probe (DNA-AuNPs) recognized RCA product to produce enzymatic electrochemical signal. The developed method was successfully applied to detect Salmonella with lower LOD of 6CFUmL−1in real milk sample (Figure 7). Both of graphene based materials and AuNPs are the frequently used nanomaterials in the field of EC biosensors for detection of biomolecules due to their excellent electrical signal amplification and the versatile functionalization chemistry. Accordingly, hybrid nanomaterial sensors based on AuNPs distributing on the surface of graphene oxide (GO) or reduce GO (rGO) also attract much scientific interest.106,107 The most frequently used technique refers to reaction of AuCl3 with GO under reductive condition for in situ anchoring AuNPs to GO or rGO. This method, however, often lacks fine control over the size, uniformity and density of AuNPs on the GO sheets in the reaction process.108 AuNPs can be decorated to GO surface based on NH-Au binding109 or SH-Au binding.110 DNA111 and proteins112 have been also used as the molecular linkers between AuNPs and GO. These methods have greatly increased the uniformity and density of AuNPs. Liu et al.113 presented stable label-free EC sensor for detection of cardiac troponin-I (cTnI) in the early diagnosis of myocardial infarction based on AuNPs and GO nanocomposites.113 The EC sensor demonstrated good selectivity and high sensitivity against human-cTnI, and was capable of detecting cTnI at concentrations as low as 0.05ngmL−1, which was 100 times lower than that by conventional methods. Karaboga et al.114 described a simple and disposable immunosensor based on indium-tinoxide (ITO) sheets modified with AuNPs to sensitively analyze heat shock protein70 (HSP70), a potential biomarker that could be evaluated in diagnosis of some carcinomas.114 Wang et al.115 constructed an AuNPs/polyaniline/chitosan-graphene sheets based electrochemical DNA sensor with functional hairpin probe for detection of BCR/ABL fusion gene in chronic myel-ogenous leukemia.115 Recently, the interest in layered transition metal chalcogenides, especially MoS2, has been growing quickly because they share many impressive physicochemical properties of graphene. Su et al. have prepared an EC sensor based on MoS2 layer for sensitive detection of proteins.116 In subsequent work, they further fabricated a MoS2-based EC aptasensor for simultaneous detection of thrombin and adenosine triphosphate (ATP) based on AuNPs-MoS2 nanocomposites.117
Development of novel ultrasensitive EC sensors provides a great opportunity for quick and specific sensing of cancer molecular markers (CMMs), which early detection is of great importance for the success of cancer therapy.118 Johari-Ahar et al.119 modified an Au electrode with mercaptopropionic acid (MPA), and then consecutively conjugated with silica coated AuNPs (AuNPs@SiO2),CdSe QDs and anti-CA-125 monoclonal antibody (mAb). Successive conjugation of AuNP@SiO2, CdSe QD and anti-CA-125mAb onto the Au electrode resulted in sensitive detection of CA-125 with aLOD of 0.0016UmL−1. Serafin et al.120 presented an immuno reagent label-free strategy to construct a dual EC immunosensor using car-bon nanotube screen printed electrodes modified with AuNPs and PEDOT NPs for the multiplexed determination of human growth hormone and prolactin.120 The ECL of luminol promoted by AuNMs was reported by Cui et al.121 The ECL intensity of luminol was found to be enhanced by ∼2 to 3 orders with catalysis of AuNPs. Li et al.122 proposed a simple and sensitive sandwich-type ECL immunosensor for detection of cancer antigen 125 on a nanoporous Au modified glassy carbon electrode.122 Gao et al.123 prepared an ultrasensitive luminol ECL immunosensor by using carboxyl group functionalized MWCNTs as platform and glucose oxidase supported on AuNPs decorated MWCNTs as labels.123 The results indicated that this developed sensor exhibited sensitive and stable response for detection of α-1-fetoprotein (AFP), ranging from 0.0001 to 80ngmL−1 with a LOD down to 0.03pgmL−1. Gui et al.124 constructed a simple and sensitive ECL biosensor for determination of β-lactamase with Ru (phen)2(cpaphen)2+ linked-ampicillin (Ru-Amp).124 In their work, Ru-Amp complex act not only as a specific recognition element for β-lactamase but also as the ECL luminescent reagent. The biosensor displayed excellent sensitivity of a concentration variation from 50pgmL−1 to 100ngmL−1 with a high sensitivity of 37pgmL−1. Beside the organic dye, noble metal NPs could greatly affect ECL behavior of semiconductor QDs.125 The ECL enhancement by LSPR of AuNPs was investigated and termed as surface enhanced ECL (SEECL).126 The LSPR of AuNPs was found to increase both the excitation rate and the emission factor of luminophores, and thus the ECL intensity was enhanced greatly. Based on this SEECL effect, a method was developed for ultrasensitive detection of Hg2+ in drinking water.127 Moreover, a SEECL sensor was prepared for detection of carcinoembryonic antigen (CEA). In this sensor, Ru (bpy)32+-doped SiO2NPs (Ru@SiO2) act as ECL luminophores, while AuNPs were used as LSPR source to enhance the ECL signal. Two different types of aptamers specific to CEA were modified on the surface of Ru@SiO2and AuNPs, respectively. A LOD of 1.52× 10−6ng/mL of CEA in human serum was achieved.128 Electrochemical kinase analysis is of special interest in monitoring biological activity in process of life. Measurements of phosphorylation reactions based on oxidation current of electroactive species such as tyrosine,129 ferrocene,130 etc., which are conjugated on the substrate during the phosphorylation processes, have been reported.131 To simplify the detection procedure, AuNPs labeled phosphorylation process for the kinase assay was designed by measuring the redox currents of AuNPs.132 However, the sensitivity of the developed method was relatively low. In order to improve the analytical performance, a DNA-based strategy was also described by the chronocoulometric response of [Ru(NH3)6]3+ absorbed on the DNA-AuNPs that linked with the phosphorylated peptide by Zr4+.133 Despite the improvement of these methods, it still remains great challenge to develop sensitive, accurate and rapid methods for the profiling of kinase activity and inhibition. Most recently, Wang et al.134 built a highly sensitive EC sensor for monitoring kinase activity based on DNA induced AuNPs polymeric network block signal amplification.134 In this strategy, the current signal of EC sensor was significantly amplified to afford a highly sensitive electrochemical analysis of kinase activity, due to its excellent electro activity and high accommodation of the DNA AuNPs polymeric network block for [Ru(NH3)6]3+. Notably, the proposed EC sensor presented a low LOD of 0.03UmL−1 for protein kinase A (PKA) activity and excellent stability even in cell lysates and serum samples. Jing et al.135 developed a sensitive and selective electrochemical method for detection of DNA methylationas well as determination of DNA methyltransferase (MTase) activity and screening of MTase inhibitor.135 In this work, methyleneblue (MB) was employed as electrochemical indicator and DNA-modified AuNPs were used as signal amplification unit because the DNA strands in this composite had strong adsorption ability toward MB (Figure 8). The differential pulse voltammetry signal demonstrated a linear relationship with logarithm of DNA methylation concentration ranging from 0.075 to 30U/mL, achieving a LOD of 0.02U/mL (Figure 8). Overall, the LSPR feature and excellent biocompatibility of AuNMs provide great chance for improving the analytical performance of EC sensors. Currently, only spherical AuNPs have been extensively applied into fabrication of EC sensor. Certainly, other shaped AuNMs possesses specific physiochemical property, and may offer additional application option in signal amplification of ECL or EC sensors.
Recently, AuNMs have been highlighted in biological imaging as a contrast agent due to their unique optical property.136 Biological imaging with simultaneous diagnosis and therapy will provide the multimodality needed for accurate targeted therapy.137 AuNMs have been considered as one of the best contrast agents for disease diagnosis, and functionalization of AuNMs becomes essential for application of AuNMs in computed tomography (CT), X-ray and SERS imaging.138 As one of the most reliable imaging modes, CT has been widely used owing to its high spatial and density resolution. For CT imaging, contrast agents are generally required to increase the density of the area to be imaged to improve the diagnostic accuracy. The commercially available CT imaging agents used in clinics are usually iodinated small molecules (e.g. omnipaque) that have severe drawbacks such as renal toxicity at a high concentration, short imaging time and non-specificity.139 For improved CT imaging, some inorganic NPs such as AuNPs140,141 have been used as contrast agents because of their higher X-ray attenuation coefficients than those of iodine-based small molecular agents. AuNPs radio sensitization represents a novel technique in enhancement of ionizing radiation dose and its effect on biological systems.142 From the first demonstration of AuNPs as a radiation contrast agent, AuNPs radio enhancement/sensitization has become an increasing area of investigation as an approach to increase the effectiveness of ionizing radiation in biological systems.143 For example, dendrimers of well-defined structure have been used as templates or stabilizers to form dendrimer-entrapped AuNPs144,145 for CT imaging applications, especially for blood pool imaging and tumor imaging. Peng et al. reported a facile approach to synthesize the dendrimer-stabilized AuNPs through use of amine-terminated fifth-generation poly (amidoamine) (PAMAM) dendrimers modified by diatrizoic acid as stabilizers for enhanced CT imaging.146 Li et al.147 constructed Au-coated iron oxide (Fe3O4@Au) nano roses to integrate five distinct functions including aptamer-based targeting, magnetic resonance imaging (MRI), optical imaging, photo thermal therapy and chemotherapy into one single probe.147 Zhang et al.148 introduced design and synthesis of branched polyethyleneimine (PEI)-stabilized AuNPs modified with polyethylene glycol (PEG) for blood pool, lymph node and tumor CT imaging.148
Despite recent advances on the different shapes of AuNPs in improving CT attenuation differences or radiotherapy damage of tumors, few studies have systematically investigated the size effect of spherical AuNPs on CT imaging and radio sensitization.149,150 In most cases, AuNPs concentration effect was examined for evaluating enhancement performances.151 Some published results indicated that great difference existed even with the same concentration of AuNPs with different size.152 In 2016, Dou et al.153 explored and confirmed the distinctive size-dependent effects based on a series of spherical AuNPs for enhanced CT imaging and radiotherapy (Figure 9).153 The result indicated that AuNPs had great size-dependent enhancement on CT imaging and radiotherapy (RT) in the size range of 3–50nm. Interestingly, AuNPs with asize of ∼13nm could simultaneously possess superior CT contrast ability and significant radioactive disruption. AuNPs have been studied as potential contrast agents for X-ray imaging, because they are nontoxic and have a higher atomic number and X-ray absorption coefficient compared with typical iodine-based contrast agents.154 Rand et al.155 proposed that the enhanced sensitivity of the X-ray scatter imaging technique over that of typical absorption based X-ray imaging could reduce the amount of AuNPs required for visible contrast.155 Therefore, they developed an imaging technique for the early diagnosis of hepatocellular carcinoma that utilized surface-modified AuNPs in combination with X-ray imaging. Tissues labeled with these electron-dense particles showed enhanced X-ray scattering over normal tissues, distinguishing cells containing AuNPs from cells without Au in X-ray scatter images. This approach could enable in vivo detection of tumors as small as a few millimeters in size. In 2012, Chien et al.156 used AuNPs as high-resolution X-ray imaging contrast agents for analysis of tumor-related micro-vasculature.156 AuNMs hold great interest in imaging field thanks to their characteristics such as mono dispersity, stability, minimal toxicity and excellent contrasting for transmission electron microscopy (TEM) analysis, which enable tracking with unprecedented resolution on dynamic of uptake, intracellular sorting and potential secretion.157,158
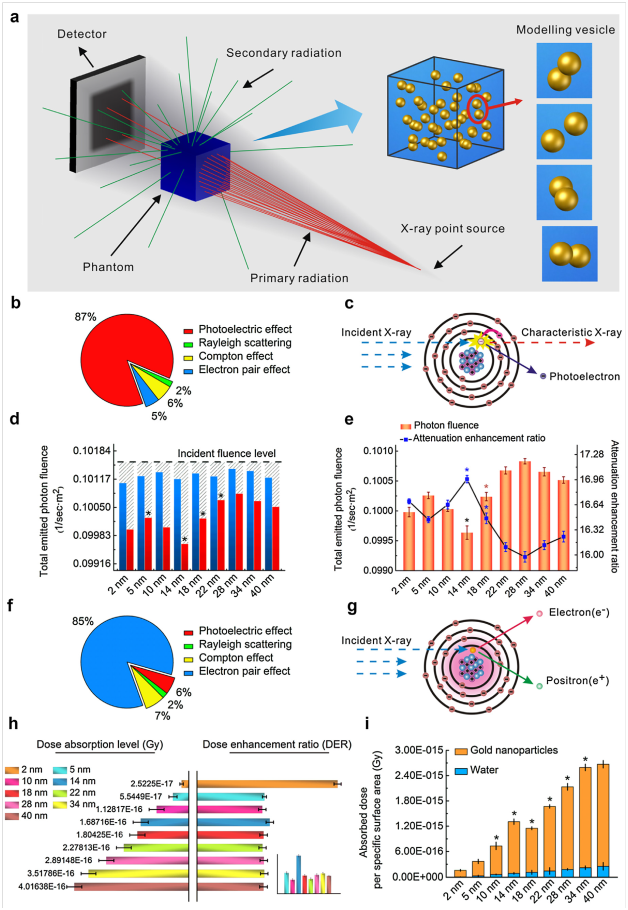
Figure 9 Monte Carlo simulation to evaluate size-dependent enhancement. (A) Scheme showing a phantom filled with a AuNP aqueous suspension that may trigger completely different secondary radiation depending on primary radiation energy irradiated from the X-ray point source for CT detection or radiotherapy. A simulated “modeling vesicle” containing two particles randomly distributed, representative of the system inhomogeneity based on particle sizes. (B) X-ray attenuation mainly coming from a photoelectric effect under the kilo voltage energy radiation for CT imaging, with little influence by other interactions including Rayleigh scattering, Compton scattering and electron pair effect. (C) Photoelectric effect generation. Inner-shell electrons receive energy from the incident X-rays, which are subsequently ejected from the atom as a photoelectron,
Recent results gathered on the interaction of AuNPs with different synthetic lipid membranes159 or cell lines demonstrate that not only properties such as size, charge and chemical functionality, but also the arrangement of organic ligands on NP surface, dictate the internalization route.160 Moreover, surface functionalization may influence the fate of AuNP upon cell uptake, for instance, HUVEC cells treated with AuNPs coated by different peptides display distinct exocytosis profiles.161 Recently, Marchesano et al.162 observed inward and outward trafficking of AuNPs at whole animal level using the small invertebrate Hydra vulgaris.162 AuNPs are also excellent candidates as contrast agents for SERS imaging. Yigit et al.163 synthesized conjugates of AuNMs and 3,3-diethylthiatricarbocyanine iodide (AuNM-DTTC) that were used as a bimodal contrast agent for in vivo MRI and Raman spectroscopy.163 The probe consisted of MRI-active super paramagnetic iron oxide NPs, stably complexed with AuNM-DTTC. The Au component served as a substrate for a Raman active dye molecule to generate a SERS effect. The synthesized probe produced T2 weighted contrast and was simultaneously used as a SERS active material both in silico and in vivo. Kong et al.164 prepared metal carbonyl based biotags by combining osmium carbonyl clusters and AuNPs, as an example of an organometallic-AuNPs (OM-AuNPs) conjugate used to cell SERS imaging.164 It showed clearly the advantage of transition-metal carbonyl compounds, for which the CO stretching vibration signal was well separated from other molecular vibrational modes of the cells in live-cell imaging.
A big disadvantage of using AuNMs as the optical contrast agents is their high photo thermal conversion efficiency under resonance excitation, which may perturb or even damage the biological species being imaged.165 Thus, to minimize unwanted heat-ing in bio imaging applications, laser irradiation time is typically set to be in the range of 0.1–10S, which either lowers the SERS signal contrast or is simply not suited for image-guided tumor resection in intraoperative settings, considering the longer timescales (1–2h) associated with such procedures.166,167 While resonant nanostructures are certainly desirable for the ranostic applications, having the ability to tune the photo thermal effect of SERS probes while preserving their high SERS activity is critical to avoid unwanted heating during imaging. Oh et al.168 designed and synthesized plasmonic AuNPs with ultra small (typically, ∼1nm) interior gap using thiolated DNA and obtained the relationships between these noble metal nano gap structures and plasmonic signals from these structures.168 In 2015, Tian et al.169 demonstrate bio enabled synthesis of a novel class of ultra-bright SERS probes with built-in and accessible electromagnetic hotspots formed by densely packed satellite NPs grown on a plasmonic core.169 Through the rational choice of the shape of the core, the LSPR wavelength of Au superstructures was tuned to be either off or on-resonant with the NIR excitation without sacrificing their high SERS activity. Consequently, the photo thermal efficiency of these ultra-bright SERS tags could be tuned to realize either contrast agents with minimal heating and perturbation, or multifunctional the ranostic agents that imaged and photo thermally killed the targeted cells.
None.
The authors declared there is no conflict of interest.

©2018 Madkour. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.