eISSN: 2379-6367


Research Article Volume 11 Issue 3
Department of Pharmacology and Toxicology, University of Puerto Rico Medical Sciences Campus, Puerto Rico
Correspondence: José G Ortiz PhD, Pharmacology and Toxicology Department, University of Puerto Rico-Medical Sciences Campus, PO Box 365067, San Juan, Puerto Rico 00936-5067
Received: June 12, 2023 | Published: July 6, 2023
Citation: Rosa-Falero C, Peña-Jiménez JA, Joubert-Miranda D, et al. Anticonvulsant interactions and anxiolytic effects of Citrus aurantium leaf infusion in the zebrafish. Pharm Pharmacol Int J. 2023;11(3):87-91. DOI: 10.15406/ppij.2023.11.00405
Many cultures use Citrus species to address various conditions, notably gastrointestinal dysfunctions, vascular disorders, and central nervous system disturbances. Citrus auratium L preparations have anticonvulsant properties. We examined the interactions of C aurantium (CA) extracts with phenytoin (PHT) or valproic acid (VPA) in pentylenetetrazole-induced seizures in zebrafish. Adult zebrafish were pretreated with the CA extract, the anticonvulsant, or their mixture and challenged with a convulsant dose of pentylenetetrazole. Co-administered C. aurantium 14 mg/mL with 8.0 mM PHT significantly increased seizure latency by 198%. The mixture of C. aurantium 14 mg/ml with VPA 3.0 mM and 6.0 mM showed a significantly higher seizure latency when compared to C. aurantium (107% and 167%, respectively) or VPA (57% and 90%, respectively). Higher doses are toxic. CA extract, 14 mg/mL, potentiates the effects of either PHT or VPA. In contrast, toxicity was more prominent with combinations of C. aurantium 20 mg/ml or 28 mg/ml and VPA. Anxiolytic effects of CA extracts were also observed.
Keywords: Citrus aurantium, seizures, zebrafish, valproic acid, phenytoin
PTZ, pentylenetetrazole; VPA, valproic acid; PHT, phenytoin; TPM, topiramate; CA, Citrus aurantium; ASM, antiseizure medications
Interactions of natural products with conventional drugs are difficult and remain understudied,1 yet epilepsy patients are willing to try natural products and dietary supplements.2,3 Adjuvant or complementary therapy could fill the gaps in the currently available antiseizure medications or reduction of their notorious side effects, resistance, and cost.
Citrus' essential oils are widely used in perfume, food, and beverage industries and as aromatherapy and medicinal agents.4-7 Among its multiple effects, Citrus aurantium extracts have anticonvulsant properties.8-10 In addition, Citrus aurantium formulations are used to treat anxiety.11-14
The zebrafish-PTZ model is validated behaviorally and electrographically.15 We examined possible interactions of CA with PHT or VPA by co-administering different concentrations to zebrafish exposed to the convulsant pentylenetetrazole. We also examined C. aurantium extract's anxiolytic effects using open-field behavior. CA extract, 14 mg/mL, potentiates the anticonvulsant effects of PHT or VPA.
In contrast, toxicity was more prominent with combinations of C. aurantium 20 mg/ml or 28 mg/ml and VPA. These results suggest CA and PHT exhibit synergy with therapeutical potential. In contrast, CA and VPA combinations are to be avoided.
Animals
Adult wild-type zebrafish (Danio rerio), 3-6-month-old males and females, and approximately 0.25 ± 0.04 g in weight were obtained from Caribe Fisheries, Inc. Lajas, Puerto Rico. Animals were housed in aquaria covered with blue contact paper to reduce stress caused by laboratory traffic. Animals were kept at a density of approximately 5 animals per liter of water. These tanks were maintained with deionized tap water, supplemented with 60 mg/L Instant Ocean® Sea Salt (Spectrum) to achieve the appropriate water chemistry. Fish tanks were maintained with constant filtration systems (mechanical and biological). The water temperature was maintained at 25–27°C using water heaters. Illumination was provided by ceiling-mounted fluorescent light tubes on a 14-light/10- dark cycle. Fish were allowed a week in quarantine to acclimate to their new environment and determine health conditions before any experiment was performed. Animals were hand fed at least once a day and fed by an automatic dispenser twice a day with Tetramin® Tropical Flake Food (Tetra Co.) or Wardley Tropical Fish Flake food. All experimental manipulations were performed between 8 am and 5 pm on a designated bench. Animals showing signs of distress either in the housing tanks or during experiments were humanely euthanized following institutional IACUC regulations. The Protocol was approved by the Institutional Animal Care and Use Committee of the University of Puerto Rico, Medical Sciences Campus (Protocol 3180110).
Chemicals
Pentylenetetrazole (PTZ), and Valproic acid (VPA) were obtained from Sigma-Aldrich. Phenytoin solution (50mg/ml) (PHT) was obtained from the Parke-Davis Division of Pfizer Inc. All stock solutions were prepared in deionized water or ethanol. Topiramate (TPM) was obtained from Neolpharma (Mexico). All final dilutions were prepared in deionized water.
Preparation of the C. aurantium leaf infusion
C. aurantium leaves were obtained from local trees in Eastern and Northern Puerto Rico. A voucher specimen was deposited at the herbarium of the Botanical Garden of the University of Puerto Rico (voucher 008253). Before extract preparation, the leaves were gently cleaned using a moist paper towel to remove dust and dirt, then heated in ultrapure water at 32°C for 1 hour to obtain an aqueous infusion was filtered through a 12.5 cm Whatman Qualitative no. 1 filter.
Dose-response curves for antiepileptic drugs
Randomly selected untreated zebrafish were placed for one hour in a small chamber containing 15 ml of a solution of either Phenytoin (PHT) or Valproic acid (VPA), prepared in aquarium water. Following ASM exposure, animals were transferred to a clear tank with a final volume of 100 mL of PTZ (3mg/mL/22mM) prepared in aquarium water. The time elapsed between exposure to PTZ and loss of coordination and swimming axis after a wild jump is considered the seizure latency. Experiments were recorded using an Olympus FE340 camera.
Herb-drug interactions
Randomly selected untreated zebrafish were placed for one hour in a small chamber containing 15 ml of a mixture of C. aurantium and either Valproic acid or Phenytoin, prepared in aquarium water. After this, animals were transferred to a clear tank with a final volume of 100 mL of PTZ (3mg/mL, 22mM) prepared in aquarium water. Changes in seizure latency caused by the mixture of ASM and C. aurantium extract were measured to determine the effectiveness of the extract. Experiments were recorded using an Olympus FE340 camera.
Open field
Behavioral testing took place in groups of three fish during the light cycle between 9:00 and 17:00 h. Groups of three zebrafish (total 12-15 fish) were placed in the center of an acrylic chamber 36.5 cm by 36.5, divided into 6.0 cm by 6.0 cm squares and recorded for 5 minutes. Zebrafish movements and monitored to assure that the animals were active and not sedated.
Statistical analysis
Data were normalized by the control mean and expressed as mean values ± standard error of the mean (SEM) of at least three independent experiments (N =12-15). Differences between the experimental groups were tested for significance using one-way analysis of variance (ANOVA) followed by the Tukey multiple comparisons test, with p<0.05 calculated, or Welch correction 2-tailed t-test using the GraphPad Prism software (https://www.graphpad.com/ v9.4.1).
Figure 1 shows the dose-response curves for Phenytoin (PHT), Valproic acid (VPA), or topiramate (TPM). Phenytoin was tested at concentrations of 2 mM, 4 mM, and 8 mM. There was a significant increase in seizure latency at 8 mM PHT. Valproic acid concentrations ranging from 3.0 mM to 18.0 mM significantly increased seizure latency. In contrast, topiramate (TPM) at 0.003 mM and 0.03 mM significantly reduced seizure latency. Seizure latency gradually increased with TPM doses ranging from 0.15 mM to 2.9 mM with a significant increase in seizure latency at 14.7 mM compared to PTZ-treated animals. Topiramate was not tested further as no clear dose-response was observed.
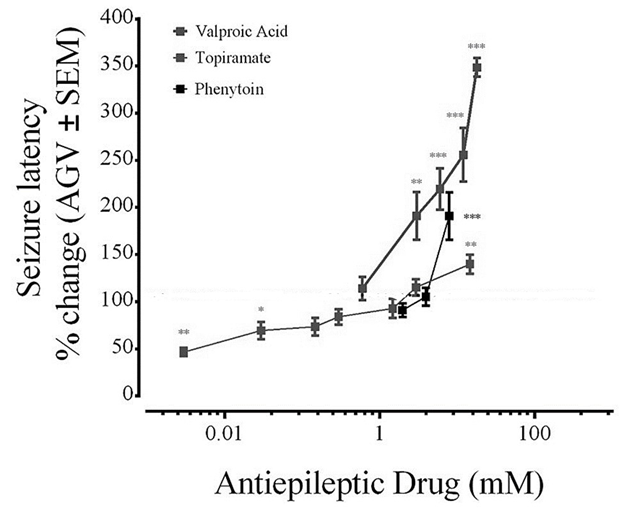
Figure 1 Dose-dependent effect of Valproic acid, Phenytoin, and Topiramate on seizure latency to PTZ in zebrafish. Zebrafish were exposed to solutions containing the AED of interest prepared in aquarium water. VPA, ranging from 3mM to 18mM, and PHT 8mM significantly increased seizure latency. TPM at a dose of 20.6 mM significantly increased seizure latency. Values were normalized against naïve animals and results are shown as average ± SEM of at least three experiments, n: 6-9 per concentration. * vs Naive P < 0.05; ** P < 0.01; ***P < 0.001
Co-administration of C. aurantium 14.0 mg/mL with PHT 2.0 mM or 4.0 mM had no significant effect on seizure latency when compared to PTZ-treated zebrafish or PHT (2.0 mM or 4.0 mM) alone as shown in Figure 2A. In contrast, PHT 8.0 mM co-administered with C. aurantium 14.0 mg/mL, significantly increased seizure latency compared to PTZ-treated zebrafish or 8.0 mM PHT. C. aurantium, 20 mg/mL, administered with PHT 2.0 mM and 4.0 mM significantly increased seizure latency by 185% and 106% compared to PHT alone but not when compared to C. aurantium 20 mg/mL alone (Figure 2B). The effect of its combination with PHT 8mM does not affect seizure latency when compared to PHT or C. aurantium alone although it did the animal showed erratic swimming patterns that suggest toxicity of the mixture. Figure 2C co-administration of C. aurantium 28 mg/ml co-administered with PHT 4mM significantly reduces the seizure latency compared to C. aurantium alone although seizure latency was still higher than that observed with PHT 4 mM alone.
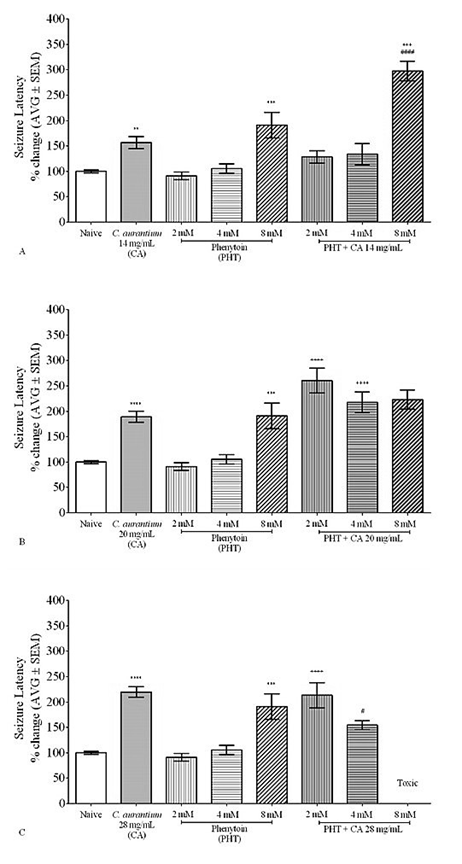
Figure 2 C. aurantium 14mg/mL potentiates the anticonvulsant effect of Phenytoin 8mM. Animals were exposed to a mixture of C. aurantium and PHT and then challenged with PTZ to assess changes in seizure latency caused by the mixture. The mixture of C. aurantium 14mg/ml with PHT 8mM significantly increases seizure latency when compared to Naïve, C. aurantium 14mg/ml, and PHT alone (A). C. aurantium 20mg/ml combined with PHT had no further effect on seizure latency than that of C. aurantium alone (B). C. aurantium 28mg/ml combined with PHT 4mM significantly reduces the seizure latency when compared to C. aurantium alone (C). Values were normalized against naïve animals and results are shown as average ± SEM of at least three experiments, n: 6-9 per concentration. * vs Naïve, + vs PHT, # vs CA.
Figure 3A shows that zebrafish exposed to the mixture of C. aurantium 14mg/ml with VPA 3 mM and 6 mM showed a higher seizure latency when compared to C. aurantium 14 mg/ml or VPA alone. Figures 3B and 3C illustrate the toxicity that resulted from the mixture of the higher dose of the C. aurantium extract (20 mg/ml and 28 mg/ml) with VPA 3 mM and 6 mM. C. aurantium, 28 mg/ml combined with VPA resulted in high levels of toxicity with animals losing their swimming axis, and difficulty breathing (opercular movement) after 10-30 min of absorption.
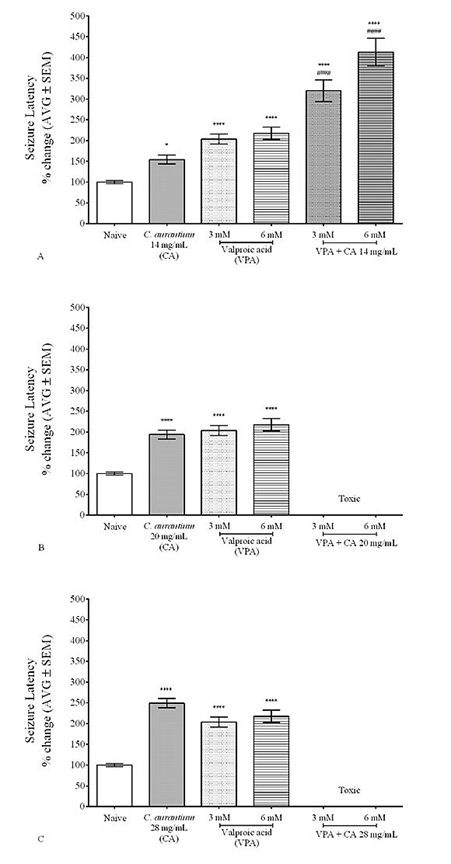
Figure 3 C. aurantium 14mg/mL potentiates the anticonvulsant effect of Valproic acid 3mM and mM. Animals were exposed to a mixture of C. aurantium and VPA and then challenged with PTZ to assess changes in seizure latency caused by the mixture. The mixture of C. aurantium 14mg/ml with VPA 3mM and 6mM significantly increased seizure latency when compared to C. aurantium 14mg/ml or VPA alone (A). The mixture of C. aurantium 20mg/ml (B) and C. aurantium 28mg/ml (C) resulted in high levels of toxicity. Values were normalized against naïve animals and results are shown as average ± SEM of at least three experiments, n: 6-9 per concentration. * vs Naïve, + vs PHT, # vs CA.
The percentage of time spent in the center of the open field arena is shown in Figure 4. Exposure to CA extract leads to an increase in the time spent in the central area. (*** p, 0.001, **p, 0.01 Welch correction 2-tailed t-test).
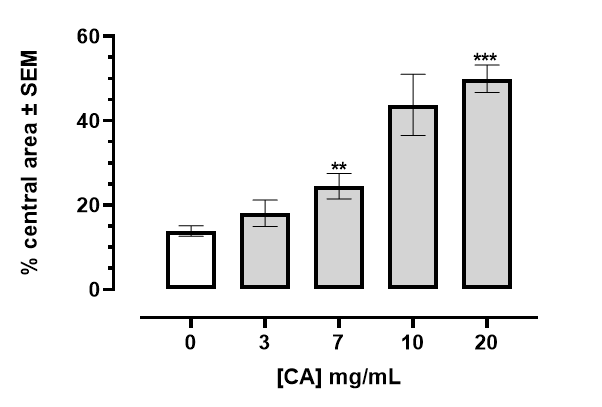
Figure 4 Percentage of time spent in the center of the open field arena of an acrylic chamber 36.5 cm by 36.5 cm, divided into 6.0 cm by 6.0 cm squares central area. Exposure to CA extract leads to an increase in the time spent in the central area. (*** p, 0.001, **p, 0.01 Welch correction 2-tailed t-test).
Citrus auratium's extracts have gained notoriety as a replacement for ephedra (Ephedra sinica) in dietary supplements.16 Citrus aurantium leaves share many of many flavonoids present in the fruit , however, synephrine or related bioactive amines were not detected in our CA extract.17
Citrus aurantium L peel and the dried unripe fruit (Aurantii Fructus, Zhiqiao, ZQ in Chinese) are commonly used for gastrointestinal motility regulation, anti-gastric ulcer effect, regulating blood pressure, cardioprotective effects, anti-atherosclerotic activity, anti-vascular damage activity, anti-depression activity, anti-obesity activity, anti-inflammatory, anti-oxidant, anti-tumor, immunomodulatory activity and affecting enzyme activities.6
Many Citrus species have anticonvulsant properties.8,10,11,18,19 It is unclear if such effects are mediated by the same constituent(s) or mechanism(a). The anticonvulsant effects of CA blossom appear to be mediated via GABAa,8 yet, we could not alter the anticonvulsant effects of CA extracts by antagonizing GABAa receptors.9 The anticonvulsant effects of our CA leaves' extracts appear mediated via NMDA and mGluR groups I and II.9
We observed anxiolytic effects at concentrations that have no anticonvulsant or sedative effects.9 The anxiolytic effects of several Citrus species and formulations have been documented in experimental models as well as, in several human studies.5,11,14,20
Pharmacological or toxicological interactions between natural products and conventional drugs cannot be underestimated.21 The short duration of our experiments and the excess of ASM or citrus extract reduce the likelihood that pharmacokinetic parameters (i.e., metabolism) played a significant role in our observations. The narrow concentration range in which the ASM and CA extracts could be tested prevented isobolographic analyses.
CA extracts have anxiolytic effects at doses below its anticonvulsant dose.9 There are clear interactions between VPA or PHT and CA leaf infusions. With PHT, there is a marked potentiation by PHT, while with VPA, the potentiation produces severe sedation.
The authors would like to thank Bianca Torres-Hernández, Yolimar Santiago, and Kiara Serrano for their help with the experiments and handling and animal care. Lizmarie Tirado-Castro should also be an author but could not be contacted. This research project is in partial fulfillment of the doctoral dissertation of Coral Rosa-Falero.
This work was supported in part by the Research Centers for Minority Institutions (RCMI/NIH Grant no. G12 RR03051) and the Minority Biomedical Research Support Research Initiative for Scientific Enhancement (MBRS RISE Grant no. R25GM061838).
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Data curation, Coral Rosa-Falero; Formal analysis, Rigel Rígel, and Jose Ortiz; Investigation, Coral Rosa-Falero, Damaris Joubert-Miranda, Rigel Rígel, and Jose Ortiz; Methodology, Coral Rosa-Falero; Supervision, Jose Ortiz; Writing – original draft, Coral Rosa-Falero; Writing – review & editing, Coral Rosa-Falero, and José G. Ortiz.

©2023 Rosa-Falero, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.