eISSN: 2379-6367


Research Article Volume 11 Issue 4
Facultad de Medicina de la Universidad Autonoma del Estado de Morelos, Mexico
Correspondence: Vera L Petricevich, Facultad de Medicina-Universidad Autónoma del Estado de Morelos, Calle Iztaccihuatl Esq. Leñeros, Col. Volcanes, 62350 Cuernavaca, MOR, México
Received: October 20, 2023 | Published: October 27, 2023
Citation: Cortezano MC, Petricevich VL. Anti-inflammatory effect of Ethanolic Extract of Bougainvillea x buttiana on LPS-induced J774A.1 macrophages. Pharm Pharmacol Int J. 2023;11(4):130-135. DOI: 10.15406/ppij.2023.11.00411
Introduction: Bougainvillea is used in Mexico to treat of various disorders. It is largely used in folk medicine as an infusion to gastrointestinal and respiratory disorders to treat cough, asthma, bronchitis and as laxative. To know the direct activity of this plant species on immune system cells the present study evaluated the immunostimulant activities of ethanolic extract from Bougainvillea x buttiana (Orange) (BxbOE) in murine macrophage cell line J774A.1.
Methods: BxbOE extract was tested on J774A.1 cells to prove their immune-stimulant effect as determines by the viability, phagocytic index and mediators production. Methylthiazolydiphenyltetrazolium bromide (MTT) and Griess assays were performed to evaluate cell viability and nitric oxide (NO) production, respectively. The phagocytic activity was determined by non-opsonized zymosan was addition and the amount of engulfed zymosan. While the cytokines levels were tested by enzyme-linked immunosorbent assay (ELISA) method.
Results: Concentrations of BxbOE at 200 to 400 μg/mL had cytotoxicity on J774A.1 cells LPS- stimulated, however, while in cells treated with lower concentrations (from 0.01 to 100 μg/mL) did not produce significant cytotoxic effects and not exhibited an inhibitory effect phagocytic index. Our results showed that the BxbOE can inhibit the pro-inflammatory mediators production including nitric oxide and cytokines comprising IL- 1β, TNF-α and IL-6, and an increment of IL-10 levels.
Conclusion: Our results help to aggregate for literature data on the immunostimulation activity of this plant species, cell from the immune system involved in the innate and acquired immune response, the macrophages.
Keywords: Bougainvillea x buttiana, nitric oxide, cytokines, phagocytosis, anti-inflammatory
BxbOE, ethanolic extract Bougainvillea x buttiana; FBS, fetal bovine serum; FST, DMEM Dulbecco’s minimal essential medium; IL, interleukin; LPS, lipopolysaccharide; TNF, tumor necrosis factor; WHO, World Health Organization
Inflammation is a natural process of the innate immune system triggered after any injury or exposure to infectious agents, with the purpose to protects the body against infection by bacteria, virus and fungi,1,2 and injury and is characterized by the activation of immune cells numerous, including monocytes, macrophages and neutrophils. The primary cells of the immune system are the macrophages which have an active participation in the inflammatory response by their functions, such as antigen presentation, phagocytosis of agents and immunomodulation allowing clearance to maintain homeostasis.3,4
Macrophage is one of the representatives of innate immune cells and together with neutrophils, eosinophils and natural killer cells are performing first line defense during infection initiates.2,3 The macrophages are found in almost all tissues and play an important role the maintenance of tissue homeostasis and are responsible for detecting, engulfing, and destroying pathogens such as the phagocytosis.5-7 Another important function of macrophage such as immunomodulation that including the production of inflammatory mediators such as nitric oxide, prostaglandins and cytokines, growth factors, reactive oxygen species (ROS) and nitric oxide (NO) in response to immune stimuli leading to immune cascades.8-11 The inflammatory process is characterized by increased vascular permeability as well as cell migration. The macrophages and neutrophils are related to the synthesis of prostaglandins and cytokines, as well as increased nitric oxide release. The studies carried out by Cezarotto et al.,12 showed that the decrease in neutrophil migration is associated with anti-inflammatory activity. The overproduction of NO contributes to tissue illness inflammatory and infectious diseases.13-15 However, overproduction of NO is related to septic shock, vascular collapse and death.16 Another macrophage function includes the vasoregulation, neurotransmission and effect on the cytotoxicity of tumor cells.17-20 In the course of the inflammatory process, the production of cytokines, such as IL-1β, IL-6, and TNF-α by macrophages take place to repair injured tissues.9 TNF-α, a cytokine involved in acute inflammatory processes, regulates the levels of other pro-inflammatory cytokines like IL-6, which is involved in the induction and perpetuation of early inflammation.21
IL-1β is another pro-inflammatory cytokine that plays an important role in host-defense responses against injury induced by infections and pain progression.22 IL-1β increases tissue damage during chronic diseases.23 IL- 10 is an anti-inflammatory cytokine that limits the host immune response by preventing damage to the host and contributing to the maintenance of tissue homeostasis.9 Thus, the inhibition of pro-inflammatory cytokines (IL-1, IL-6, and TNF-α), and the increase in the production of immunomodulatory like cytokine IL-10. This cytokine acts as a strong obstruction of macrophage activity by blocking of pro-inflammatory cytokines synthesis for example, TNF-α, IL-6 and IL-12 and by failing the disastrous effects of excessive activation of this cell during inflammation.24,25 Though over activated immune system damages host’s homeostasis, weakened immune system is also significantly harmful to human health. The overproduction of pro-inflammatory cytokines promote systemic inflammation and include interleukin-1 (IL-1), interferon-gamma (IFN-γ), and tumor necrosis factor (TNF). By contrast, anti-inflammatory cytokines that include IL-4, IL-10, and IL-13 are those responsible for regulating and maintaining homeostasis.26-29
Herbal medicines as a therapeutic tool have been used for centuries to treat different illnesses and numerous medicinal plants and their derivatives are used as the inflammatory diseases treatment.30-34 Much research has been undertaken to identify the active compounds and mechanisms of action of plants used in traditional medicine.30 Bougainvillea x buttiana is one of genera of the Nyctaginaceae family widely used in Mexico traditional medicine. It is largely used in folk medicine as an infusion to gastrointestinal and respiratory disorders to treat cough, asthma, bronchitis and as laxative.35,36 The analgesic, antioxidant, and anti-inflammatory properties of this plant have been shown in various studies.36,37 In our previous study, we described the immunomodulatory activity of the B.xbuttiana ethanolic extract on peritoneal macrophages.37 Little is known about the effect of Bougainvillea x buttiana on the cells from the immune system. We previously showed the immunomodulatory effect of the ethanolic extract of Bougainvillea x buttiana on peritoneal macrophages.33 Thus, the aim of this study was to expand understanding of the anti-inflammatory activity of ethanolic extract from B. xbuttiana in macrophages, J774A.1 stimulated by LPS. Their effects on immune related nitric oxide and cytokines production (TNF-α, IL-1β and IL-6), and phagocytic activity were investigated.
Materials and reagents
Griess reagent, dimethyl sulfoxide (DMSO), lipopolysaccharide (LPS, E. coli 0111:B4), Thiazoyl blue tetrazolium bromide (MTT), Fetal bovine serum (FBS), Dulbecco´s modified Eagle´s medium (DMEM) and DMSO were purchased from Sigma-Aldrich (St. Louis, MO, USA). All capture and detection antibodies of cytokines (TNFα, IL-1β, IL-6 and IL-10) were purchased from BD Biosciences. (San Jose, CA, USA). All other reagents were analytical grades.
Extraction of Bougainvillea x buttiana
Bougainvillea x buttiana were cultivated in Cuernavaca (Morelos, México). The specie was examined by Herbarium HUMO, CIByC-UAEM, under the number certificate specimen (23683). The method for extraction of Bougainvillea x buttiana was conducted in detail as describe in Patent MX/a/2011/813522.37 In brief, the Orange flowers collected were dried and pulverized and the sample through by maceration with ethanol 50% for 72 hours at 25°C. The crude 50% ethanol extract obtained was filtered and subjected to evaporation under reduced pressure in a rotary evaporator. Bougainvillea x buttiana ethanolic extract was referred as BxbOE.
Cell culture
Murine macrophage cell line J774A.1 (ATCC TIB67™) was cultured in Dulbecco’s minimal essential medium (DMEM) with 10% FBS, and antibiotic mixture (constituted by penicillin and streptomycin 100 units/mL), under a humidified 5% CO2 at 37°C.
Cytotoxicity assay
Cytotoxicity of BxbOE was performed in murine macrophages J774A.1 by the MTT assay. Cells were cultured in DMEM supplemented with 10% FBS, 2 mM glutamine, 100 μg/mL streptomycin and 100 U/mL penicillin, and incubated at 37°C, in a humidified 5% CO2. Macrophages J774A.1 (1 X 104 cells) were seeded in 96 well plates and incubated for 24 hours before they were treated with or without BxbOE at given concentrations of 0.01, 0.1, 1, 10, 50, 100, 200 and 400 μg/mL for 24 and 48 hours the same conditions. The cells incubated with only culture medium was considered as negative controls. After 24 and/or 48 hours, 5 mg/mL of MTT solution was added to each well and incubated for 4 hours. Subsequently, the supernatants were eliminated and DMSO was added for dissolving formazan. The absorbance was evaluated with a microplate reader at 540 nm. All assays were performed in quadruplicate.
Nitric oxide production
The nitric oxide levels were determined in supernatants by the Griess assay.38 Briefly, J774A.1 cell was seeded in a 96 well plate (1 x 105 cells/well) and incubated with the BxbOE at concentrations of 0.01, 0.1, 1, 10, 50 and 100 μg/mL for 1 hour. As a reference anti-inflammatory drug was used Dexamethasone (50 nM). After 1 hour of treatment, the cells were stimulated with LPS (10 μg/mL) and maintained in culture for 24 hours in a 5% CO2 at 37°C. The absorbance was evaluated at 540 nm using microplate reader. The amount of nitrite was determined by a standard curve of NaNO2. Minimal detection for nitrite was 1.25 nM.
Enzyme-linked immunosorbent assay (ELISA)
J774A.1 cells were treated with BxbOE at given concentrations (0,01, 0.1, 1, 10, 50 and 100 μg/mL) and/or treated with LPS at 10 µg/mL and/or Dexamethasone 50 nM, for 24 hours, their supernatants concentrations of TNF-α, IL-1β, IL-6, and IL-10 were measured by an ELISA.39 All assays were measured according to manufacturer’s instructions. Absorbance was read at 405 nm on a microplate reader and compared with the specific recombinant cytokines. The results were expressed in pg/mL and the minimum levels of these cytokines detected were 10 pg/mL.
Phagocytic activity
The phagocytic ability of J774A.1 cells was evaluated by the method described by Zebedee et al.40 Cells were seeded in 96-well plate at 1 x 105 cells/well and incubated at 37°C with 5% CO2 for 24 h. After this time the cells were treated with BxbOE (0.01, 0.1, 1, 10, 50 and 100 μg/mL), complete DMEM was used as a negative control and LPS (10 μg/mL) was for positive control. At the end of 24, hours of incubation, the supernatants were discarded and a solution containing of opsonized yeast 1:5 was added for 1 hour. After this incubation the supernatants were substituted by adding safranin solution for 40 seconds. The phagocytic index (PI) was evaluated by employing of optical microscope at 40x the phagocytic index (PI) was estimated with following formula: number of macrophages with internalized yeast/100.
Statistical analysis
The data is reported as the mean ± standard deviation (SD) and was statistically analyzed by two-tailed Student t- test using Microsoft Excel 2016. P values < 0.05 were considered significant.
Physicochemical analysis of bract from Bougainvillea x buttiana variety orange
GC-MS analysis of ethanolic extract from Bougainvillea x buttiana has been previously reported by Arteaga- Figueroa et al.37 This analysis revealed the presence of 9 compounds such as 2-Propenoic acid, 3-(2- hydroxyphenyl), (E)-, 3-O-Methyl-d-glucose, Tetradecanoic acid, n-hexadecanoic acid, Isopropyl palmitate, Squalene, Diisooctyl maleate and 1,2-Benzenedicarboxylic acid, diisooctyl ester.
Effect of extract BxbOE on cell viability
To determine the toxicity induced by BxbOE at different concentrations were tested on J774A.1 macrophage cell line for 24 and 48 hours. Figure 1 expressed as percentage of viable cells. The concentrations of 400 μg/mL of BxbOE had a cytotoxic effect, indicating significant reduction of the surviving cells 50% dead cells (Figure 1). The cells treated with a BxbOE concentration of 100 and 50 μg/ mL the cytotoxic effect was 10% and 8%, respectively, whereas other concentrations tested were non-toxic, at cell viability higher than 90%, for the groups of cells treated with 10 and 1 µg/mL the viable cells were 96 and 99.8%, respectively while in groups of cells treated with 0.1 and 0.01 μg/mL of BxbOE presented 100% viable cells (Figure 1). The cytotoxic effect of BxbOE was dose dependent.
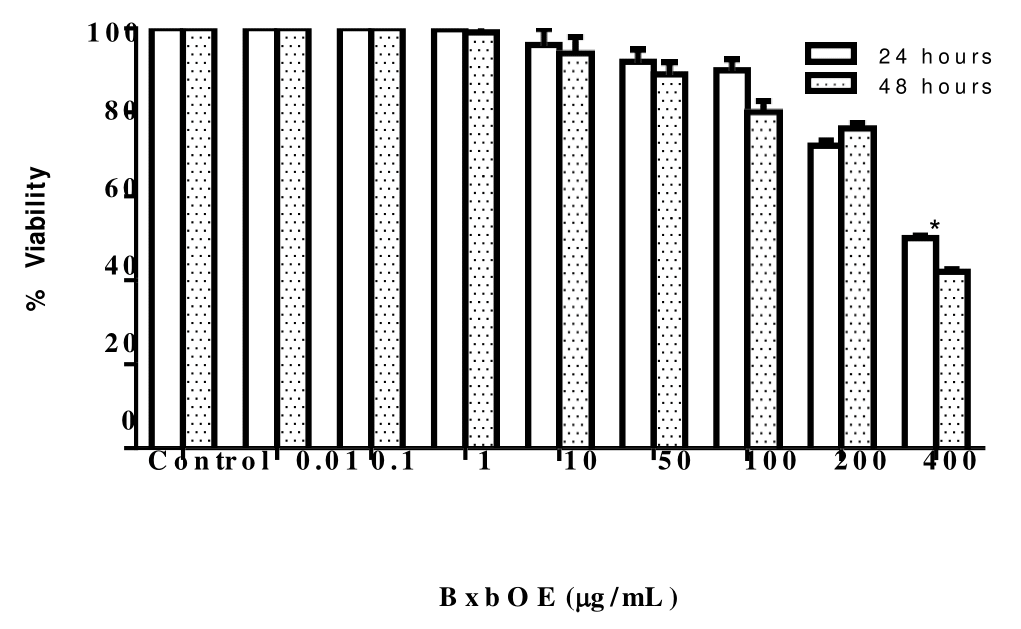
Figure 1 Effect of BxbOE on J774A.1 macrophage viability. The cytotoxic effects of BxbOE at the concentrations of 0.01 to 400 µg/mL on J774A.1. Macrophage cell line was incubated with different concentrations of the BxbOE. After 24 and/or 48 hours, 5 mg/mL of MTT solution was added to each well and incubated for 4 hours, and then the absorbance was measured with a microplate reader at 540 nm. The viability was expressed as the percentage compared with the untreated cell group. All assays were performed in quadruplicate, and the values are presented as mean ± SD. *Indicate statistically significant difference (P < 0.001) between stimulated group (LPS+) and non-stimulated group (LPS-).
Effect of extract BxbOE on nitric oxide production in LPS-stimulated J774A.1 macrophages
Extract concentrations that showed a percentage of cell viability below 80% were chosen as non-toxic, the concentrations of 200 to 400 µg/mL were excluded from further anti-inflammatory analysis. To investigate the ability of BxbOE to modulate the nitric oxide production on J774A.1 macrophages stimulated with LPS at different concentrations of 1, 5, 10 µg/mL were used to stimulate J774A.1 macrophages. Under the conditions used to carry out this work, the concentration chosen was the one that caused a toxicity lower than 10% which was 10 µg/mL of LPS (Data not shown). BxbOE effect on NO production in LPS-stimulated macrophages is described in Figure 2. In groups of J774A.1 macrophages treated with LPS the production of NO was significantly higher when compared with control cells (P < 0.005). For groups of J774A.1 stimulated with LPS and treated with Dexamethasone, a significant reduction in NO levels was observed (P < 0.005) (Figure 2). Groups of cells J774A.1 treated with different concentrations of BxbOE demonstrated potent inhibitory activity against LPS stimulation. The highest reductions in NO levels were observed in cultures treated with 10, 50 and 100 µg/mL of BxbOE, being 57%, 46.2% and 35.8%, respectively (Figure 2). With these concentrations of BxbOE have suppressed the NO production of almost 2.5-fold when compared to the LPS- induced group (Figure 2).

Figure 2 BxbOE effect on NO production in LPS-stimulated J774A.1 macrophages. J774A.1 macrophages LPS-stimulated were treated with different concentrations of BxbOE. The NO level was studied by observing the nitrite level production by the Griess reagent. The nitrite production was expressed in µM. All assays were performed in quadruplicate, and the values are presented as mean ± SD. *Indicate statistically significant difference (P < 0.005) between stimulated group (LPS+) and non- stimulated group (LPS-).
Effect of BxbOE on cytokine production in LPS-stimulated J774A.1 macrophages
To examine the ability of different concentrations of BxbOE to modulate the cytokines production on J774A.1 macrophages stimulated with 10 µg/mL of LPS. The cytokine production in LPS-induced macrophages was checked using ELISA assay. The effect of BxbOE on cytokine production in LPS-stimulated macrophages is described in Figure 3. In groups of macrophages J774A.1 treated with BxbOE and induced by LPS the production of cytokines IL-1β, IL-6 and TNF-α was significantly reduced in a dose-dependent manner when compared with cells stimulated by LPS (P < 0.005) (Figure 3). The highest reductions in TNF-α, IL-1β and IL-6 levels were observed in cultures treated with 10, 50 and 100 µg/mL of BxbOE (Figure 3). In groups of J774.A1 macrophages stimulated by LPS and treated with 10, 50 and 100 µg/mL of BxbOE, the TNF-α were reduced 2.1, 2.8 and 3.35-fold, respectively. The reduction in IL-1β production was 1.6, 2.0 and 2.0-fold in macrophages cultures induced by LPS and treated with 10, 50 and 100 µg/mL, respectively. The greatest reductions in IL-6 levels observed in the macrophage treated with 10, 50 and 100 µg/mL were 2.9, 3.3 and 4.9-fold, respectively. In contrast, the groups of J774A.1 stimulated with LPS a significant increment in IL-10 levels was observed when compared to those levels observed in the J774A.1 groups induced with LPS for 24 hours (P < 0.05) (Figure 3).
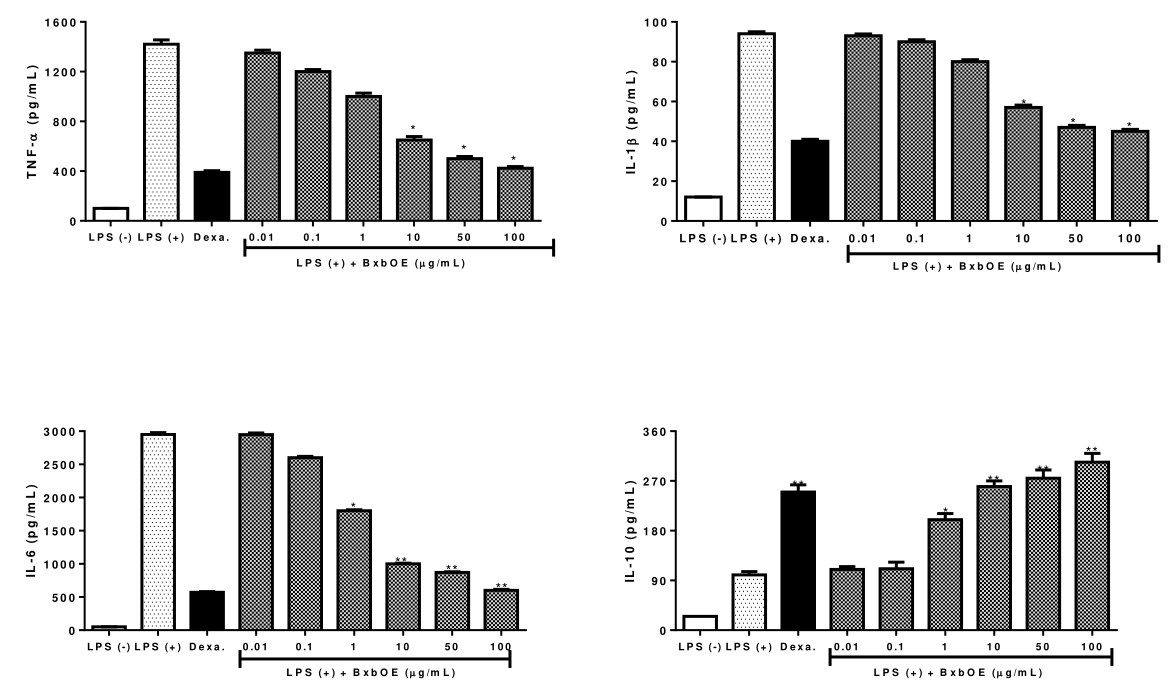
Figure 3 Effect of BxbOE on cytokine production in LPS-stimulated J774A.1 macrophages. J774A.1 cells induced by LPS were treated with different concentrations of BxbOE. The cytokine production was determined by ELISA. All assays were performed in quadruplicate, and the values are presented as mean ± SD. * Indicate statistically significant difference (P < 0.05) and ** (P< 0.005) between stimulated group (LPS+) and non-stimulated group (LPS-).
Effect of BxbOE on phagocytic activity in LPS-stimulated J774A.1 macrophages
To investigate the effect of different concentrations of BxbOE were tested on phagocytic activity in J774A.1 macrophages stimulated with 10 µg/mL of LPS. The effect of BxbOE on the phagocytic activity in LPS-stimulated macrophages J774A.1 is described in Figure 4. All concentrations of BxbOE used were able to increase phagocytic capacity when compared with the groups of cells without treatment (Figure 4). Another observation is that the percentages of phagocytosis obtained in groups of macrophages J774A.1 treated with BxbOE were like those observed in groups of cells induced by LPS (Figure 4).
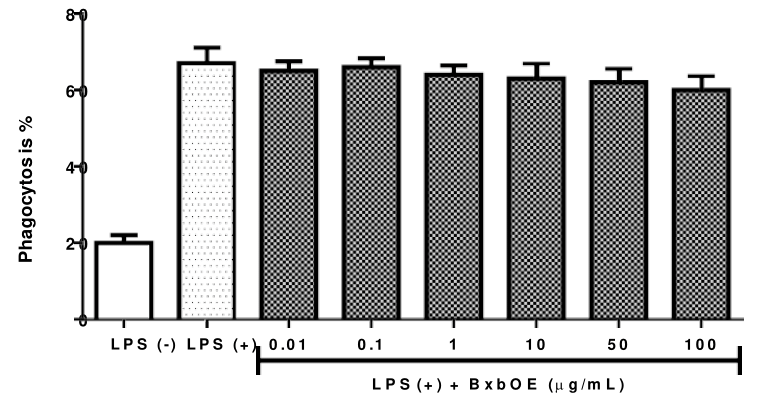
Figure 4 Effect of BxbOE on phagocytic activity in LPS-stimulated J774A.1 macrophages. J774A.1 cells were treated with different concentrations of BxbOE and stimulated with LPS. The phagocytosis percentage was determined as described in Materials and Methods. All assays were performed in quadruplicate, and the values are presented as mean ± SD.
In previous studies we show the chemical analysis by GC-MS of Bougainvillea x buttiana (Variety Orange) ethanolic extracts the existence of 9 compounds such as the phenolic compounds like the 2-Propenoic acid, 3-(2- hydroxyphenyl) that is recognized as strong antioxidants by their high redox activities.41,42 Among the chemical group of the carbohydrates, the 3-O-Methyl-d-glucose that has the protective activity.43 Chemical group of fatty acid a) Tetradecanoic acid presented cytotoxic effect,44 b) n-hexadecanoic acid present the follows activities such as anti-inflammatory, antioxidant, and antinociceptive45 and c) Isopropyl palmitate that show the antioxidant activity.46 Chemical group of terpenes the squalene demonstrated the antioxidant, antitumoral and antifungal activities.47 Chemical group ester Diisooctyl maleate Chemical group of alkene the 1,2-Benzenedicarboxylic acid, diisooctyl ester with showed antifungal, antioxidant and anti-inflammatory activities.48-51
Macrophages are considered important cells in inflammatory responses due to their functions such as presentation of antigen, phagocytosis and immunomodulation. The main function of macrophages in inflammation is to promote the secretion of mediators and cytokines.52 The suppression caused by these cells, or their mediator derivatives is the key which allows the host to reconstruct the tissue damage resulted from inflammatory process. In the present study the effects of BxbOE on inflammatory mediators secreted in J774A.1 macrophages stimulated by LPS were investigated. The effects of different concentrations of BxbOE on cell viability were analyzed. At concentrations of 0.01 to 100 µg/mL of BxbOE they did not present a cytotoxic effect on macrophages.
The groups of macrophages stimulated-LPS induce a nitric oxide production. Nitric oxide is considered an important biological mediator and regulator of inflammation.53 In this study the effect of BxbOE on the nitric oxide production was analyzed. The results obtained showed that BxbOE reduced NO production in a dose-dependent manner in cultures of J774A.1 macrophages stimulated by LPS. Different studies that have shown that extracts obtained from different plants can reduce NO production.53-55 Our results are in agreement with other studies and suggest that the BxbOE has pharmacological action on macrophages.
Both cytokines and mediators play a vital role in the inflammatory response due to their chemotactic and vasoactivating properties. With the purpose, the increment of the permeability of the blood vessel for both cells and fluids, to recruit more inflammatory cells to the damaged region, and providing localization in the infected area and removing the harmful agent. Mediators and cytokines are extremely important for maintaining the internal environment against harmful agents. For this reason, they must be adjusted back to their original level. When its expression is elevated over a prolonged period, it become harmful and can develop into chronic inflammation.
Our results showed that BxbOE was able to cause a significant reduction in the levels of TNF-α, IL-1β and IL-6 in J774A.1 macrophages stimulated by LPS. The reductions observed for the groups of macrophages treated with BxbOE were dose dependent. TNF-α produced by macrophages is involved in the acute phase of inflammation through the neutrophils recruitment and activation of arachidonic metabolism.56 The role of IL-1β in inflammatory responses is due to infections and illnesses.56 In experimental models have been showed that the IL-1β inhibitors can be used to diminish the inflammation caused by arthritis. Another pro-inflammatory cytokine involved in the generation and propagation of inflammation is IL-6.57 Our results showed that BxbOE was able to reduce IL-6 levels in a dose-dependent manner. With current knowledge, it is known that the inflammatory response can be resolved through the elimination of inflammatory cells or by the release of endogenous anti- inflammatory mediators, such as IL-10, which is a late cytokine produced by several types of cells that can limit collateral damage.58 Thus, the effect of BxbOE on the production of IL-10 from J774A.1 macrophages induce by LPS was evaluated. In groups of macrophages J774A.1 induced by LPS low levels of IL-10 were detected. In contrast, the BxbOE induced the IL-10 production at all concentrations tested. Nonetheless, the anti-inflammatory activity of the BxbOE is complicated and up to now is not completely understood.59
The present study showed that BxbOE has strong anti-inflammatory activity and suggest a general inhibitory effect on macrophage activation and that the activity can be attributed to the synergism between the different molecules present in the extract.
The results obtained in this study show that ethanolic extract of Bougainvillea x buttiana proved for its anti- inflammatory activities. This activity was confirmed by the maintenance of phagocytic activity, reduction in the levels of NO and the cytokines such as TNF-α, IL-1β and IL-6 and increment in IL-10 levels in macrophage cultures. The chemical analysis of BxbOE, describing the main constituents, associates these metabolites with the observed anti-inflammatory activity. Both mediators, as well as signaling pathways and transcription factors expressed in activated macrophages in the inflammatory process can be targets of future studies to better understand the pharmacological action of the extract and the main compounds from Bougainvillea x buttiana Variety Orange.
This study received financial support from the Secretariat of Public Education (SEP-PRODEP), Mexico, and the National Council of Science and Technology (CONACYT, Mexico).
No conflict of interest associated with this work.

©2023 Cortezano, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.