eISSN: 2377-4304


Research Article Volume 17 Issue 1
1Clínica Hexsel de Dermatologia, Porto Alegre, Brazil
2Guarnieri & Munia Médicos Associados, São Paulo, Brazil
3Clínica Dermatológica Dra Meire Parada, São Paulo, Brazil
4Healthy Laser & Skin Care Assistência Médica São Paulo, Brazil
5Galderma, São Paulo, Brazil
Correspondence: Cristhine Kamamoto, MD, PhD, Galderma, 14261, Nações Unidas Ave. 4º andar - Torre A, Ala A - Condomínio WT Morumbi - Vila Gertrudes – ZIP CODE: 04730-090, São Paulo - SP, Brazil, Tel +55 11 93315-7523
Received: December 25, 2025 | Published: January 21, 2026
Citation: Hexsel D, Guarnieri CM, Parada MB, et al. Retrospective study on the effectiveness and safety of poly-L-lactic acid (PLLA-SCA) for skin flaccidity in the labia majora and mons pubis. Obstet Gynecol Int J. 2026;17(1):10-14. DOI: 10.15406/ogij.2026.17.00815
Background: Poly-L-lactic acid (PLLA-SCA) is a biostimulator used for the treatment of sagging skin. In recent years, there has been a growing demand for dermatological procedures in the genital region, primarily attributed to the loss of firmness, which can be improved with PLLA-SCA injection.
Purpose: Evaluate the effectiveness and safety of PLLA-SCA for skin flaccidity in the labia majora and mons pubis.
Methods: This retrospective, single-arm, clinical trial involved the analysis of medical records from nine women who underwent treatment with PLLA-SCA for skin flaccidity on the labia majora and/or mons pubis. The endpoints included improvements in skin flaccidity, functional symptoms, and overall aesthetic appearance, which were assessed by independent investigators using the Global Aesthetic Improvement Scale (GAIS), as well as the frequency of adverse events. Clinical trial registration: NCT05940038.
Results: The participants were Caucasian women, with a median (p25-p75) age of 59 years (48–71). All underwent three PLLA-SCA sessions in the indicated region, using one vial per treatment session. Only two had previously undergone treatment with PLLA-SCA in other areas, and none had undergone any other treatment in the labia majora and mons pubis. The labia majora were treated in 78% of the cases, while 22% were treated in other external genital areas. GAIS evaluation showed improvement in 100% of the subjects, both in skin flaccidity and overall appearance. Regarding functional symptoms (e.g., discomfort in sports, difficulties in sexual intercourse, perception of flaccidity), 86% reported complete or partial improvement. Four patients (44%) experienced adverse events after the procedure (pain, hematoma, and hypersensitivity in the labia minora), however, all were mild and transient.
Conclusion: Procedures involving the injection of PLLA-SCA in the mons pubis and labia majora are well tolerated and effective for reducing flaccidity and enhancing skin firmness. Moreover, these procedures also have the potential to mitigate functional complaints associated with genital aging.
Keywords: aging, labia majora, poly-L-lactic acid, mons pubis
Aging is an inherent process that accompanies individuals across all stages of their development, and the external female genitalia are not exempt from its effects.1 Menopause induces a gradual reduction in female hormones, leading to a decline in hyaluronic acid, collagen, and fat. This contributes to a loss of volume and the onset of sagging skin throughout the body, including the external female genitalia, significantly impacting the quality of life for women.2,3
In the ever-evolving world of aesthetic medicine, individuals are increasingly seeking options beyond traditional facial rejuvenation or body contouring. One area gaining attention is genital beautification, a term encompassing various procedures designed to enhance the appearance and function of the genital region.4 One common concern among individuals seeking genital beautification is dissatisfaction with the size, shape, or symmetry of the external genitalia, especially labia majora hypotrophy and labia minora redundancy. Non-surgical options, such as dermal fillers, have also gained popularity for labial and pubis enhancement. These minimally invasive procedures offer temporary volumizing effects and natural results with short downtime, providing an alternative for those hesitant to undergo surgical interventions.5,6
Beyond aesthetics and self-esteem, the labia majora serve a crucial function in safeguarding the labia minora, the upper portion of the vestibule, and the posterior part of the vaginal introitus. This protective role helps to prevent undesired friction of the labia minora, especially friction-related discomfort from activities such as sports and during sexual intercourse. Hypotrophy of the labia majora contributes to chronic vulvitis and vulvovaginal dryness, leading to discomfort, itching, and dyspareunia.7–9
Hormonal and non-hormonal treatments are part of the therapeutic options for treating the genital area of menopausal and perimenopausal women. Non-hormonal treatment options encompass lifestyle changes, pelvic physiotherapy, moisturizers, lubricants, lasers, and other energy-based therapies like radiofrequency, as well as minimally invasive procedures such as fat grafting. The application of injectable hyaluronic acid-based fillers and collagen biostimulators on the skin of the labia majora and mons pubis represents a minimally invasive approach to enhance local skin volume and address skin flaccidity.10,11
PLLA-SCA is an implant of poly-L-lactic acid, biodegradable, sterile and biocompatible polymer. PLLA-SCA is a collagen biostimulator that should be injected into the deep dermis or subcutaneous plane to improve skin laxity resulting from intrinsic and extrinsic aging processes and to correct volume loss in depressed areas, such as grooves, wrinkles, skin depressions, atrophic scars, and changes resulting from facial lipoatrophy, including in people living with the human immunodeficiency virus (HIV).12 It functions by stimulating the production of skin collagen through the activation of fibroblasts. Upon injection into the skin, PLLA-SCA triggers a controlled local and subclinical inflammatory response, leading to the gradual formation of collagen and elastin around each particle and a subsequent increase in dermal thickness.13–15
Previous reports involving injectable hyaluronic acid or calcium hydroxyapatite in the external female genitalia have reported satisfactory results and mild, reversible adverse events. However, research specifically addressing the use of PLLA-SCA in collagen biostimulation for the pubic region is limited.2,3,11,16–20 Hence, this study aims to assess the effectiveness and safety of PLLA-SCA by examining data from the medical records of participants who underwent treatment for skin flaccidity in the labia majora and mons pubis.
Study design
This was a retrospective, open-label, single-arm clinical trial that collected medical record data to evaluate the safety and effectiveness of applying PLLA-SCA for the treatment of skin flaccidity in the labia majora and mons pubis.
Study population
The study involved the analysis of medical records from women who underwent injections of PLLA-SCA to treat the labia majora and mons pubis after January 1st, 2022 in the authors' clinical settings, with a follow-up period of up to 12 months.
Medical charts from adult women (>18 years old) were selected from regular patients in the last two years who visited four private clinics in Brazil. Subjects with any clinical condition, previous surgery, or recorded laboratory alterations in the medical records that could compromise participation in the trial, those who had undergone any procedure on the skin of the labia majora and/or mons pubis less than 12 months before the first administration of PLLA-SCA, or those who had participated in another clinical study less than 30 days before inclusion in this trial were not included.
Since this was a retrospective study, patients whose medical records were selected were contacted to obtain consent and sign the Consent Form. The study protocol was approved by the Research Ethics Committee of the Hospital Moinhos de Vento (#69127223.3.0000.5330), and all the participants gave written consent for data and image publication. All procedures in the study were conducted in accordance with the Declaration of Helsinki (as revised in 2013). The clinical trial registration number is NCT05940038.
Outcomes
The following demographic data were recorded: age, sex, ethnicity, and body mass index (BMI). Medical evaluation included standardized photographs of the skin of the labia majora and/or mons pubis taken before and after the treatments, from at least one PLLA-SCA session.
The following parameters were evaluated: the number of sessions performed, number of vials per session, total volume per session; reconstitution details such as total volume of diluent (mL), use of lidocaine or other ingredients, product reconstitution time; areas treated per session; assessment of skin flaccidity and global aesthetic improvement of the labia majora and mons pubis using GAIS by comparing clinical photographs before and after treatment, improvement in functional symptoms, and frequency of adverse events.
An independent investigator assessed skin flaccidity and the global aesthetic aspect in labia majora and mons pubis using the GAIS (Global Aesthetic Improvement Scale) scale score after reviewing the photographic records. GAIS is a 5-point scale used in aesthetic medicine to assess the overall improvement in a patient's appearance after a specific treatment or procedure, and it ranges from "worse" to "very much improved".21
Safety assessment
The safety assessment involved recording adverse events related to the product or the injection procedure, documented in medical records during follow-up visits within a period of up to 12 months after the initiation of the procedure with PLLA-SCA. Adverse events were classified as expected or unexpected; mild, moderate, or severe; definite, possible, probable, or unlikely cause; and fully resolved, resolved with sequelae, in progress of resolution, or resulting in subsequent death.
Data analysis
Statistical analysis was conducted using IBM SPSS software version 29. Continuous variables were presented as median and interquartile intervals (p25-p75), and categorical or ordinal variables as their percentiles.22–24
A total of nine patient records were assessed, all of whom were caucasian, with a median (IQR) age of 59 years (48─70). The median (IQR) BMI was 24 (23─25), and none of the patients were smokers. Each of the nine patients received three PLLA-SCA sessions in the labia majora and mons pubis.
Regarding the treatment location, the labia majora was the chosen region in 7 (78%) of the participants, while 22% involved other external genital areas, although specific details were not specified in the records.
Sterile water was the diluent used in all cases, varying only in the dilution volume: 45% used 15 mL, 33% diluted in 14 mL, and 22% in 8 mL, resulting in a final volume of 16 mL in 78% of the cases and 10 mL in 22% of the participants. In all instances, the final volume was attained by adding lidocaine 2% to ensure adequate anesthesia in accordance with physicians practice.
All cases utilized one PLLA-SCA vial per session, and all subjects underwent three identical sessions within one year (minimum interval of 3 weeks). The reconstitution (hydration) time for PLLA-SCA vials was immediate in 5 (56%) of the subjects evaluated, and 24h for the other four women.
Regarding the injection plane, the treatment with PLLA-SCA was administered in the superficial subcutaneous plane in 8 (89%) participants, while the data for one woman was not retrieved from the files. Among the cases, 67% were treated using a 22G cannula, 22% with a 15G cannula, and data for one patient were not specified in the archives.
The procedures of the nine participants are summarized in Table 1.
|
Case |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
|
Diluent |
Sterile Water |
Sterile Water |
Sterile Water |
Sterile Water |
Sterile Water |
Sterile Water |
Sterile Water |
Sterile Water |
Sterile Water |
|
Diluent volume (mL) |
14 |
14 |
8 |
8 |
15 |
15 |
15 |
15 |
14 |
|
Hydration time |
immediate |
immediate |
immediate |
immediate |
24h |
24h |
24h |
24h |
immediate |
|
Anesthetic use |
Lido 2% |
Lido 2% |
Lido 2% |
Lido 2% |
Lido 2% |
Lido 2% |
Lido 2% |
Lido 2% |
Lido 2% |
|
Anesthetic volume (mL) |
2 |
2 |
2 |
2 |
1 |
1 |
1 |
1 |
2 |
|
Final dilution volume (mL) |
16 |
16 |
10 |
10 |
16 |
16 |
16 |
16 |
16 |
|
Number of vials per session |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
1 |
|
Number of sessions |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
3 |
|
Interval S1-S2 (weeks) |
3 |
5 |
7 |
7 |
5 |
5 |
5 |
5 |
3 |
|
Interval S2-S3 (weeks) |
6 |
5 |
4 |
4 |
5 |
5 |
5 |
5 |
6 |
|
Treated areas |
LM / MP |
Missing* |
LM |
LM |
LM |
LM |
Missing* |
LM |
LM |
|
Injection plane |
SSC |
Missing** |
SSC |
SSC |
SSC |
SSC |
Missing** |
SSC |
SSC |
|
Injection device |
Cannula |
Missing** |
Cannula |
Cannula |
Cannula |
Cannula |
Cannula |
Cannula |
Cannula |
|
Gauge (G) |
22G |
Missing** |
25G |
25G |
22G |
22G |
22G |
22G |
22G |
Table 1 Summary of the treatments with PLLA-SCA for addressing skin flaccidity in the labia majora and mons pubis
LM, Labia Majora; MP, Mons pubis; SSC, superficial subcutaneous; Lido 2%, lidocaine 2%; S1, S2, S3, first, second and third sessions.
*Not informed in the medical chart if treatment of LM only or both LM and MP.
**Although not informed in the medical chart, the injection plane, injection device and its gauge were defined in accordance with the authors’ clinical practice
Upon the assessment through GAIS, all subjects exhibited improvements in both skin flaccidity and overall appearance (Figure 1), reaching a 100% improvement rate (Table 2).
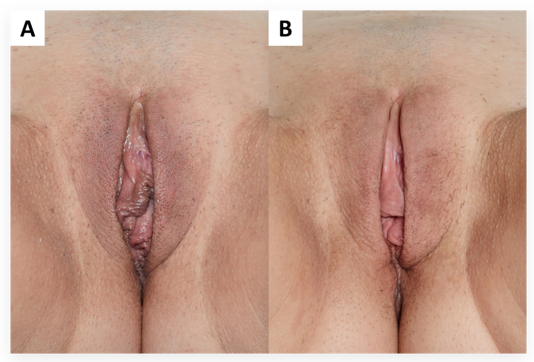
Figure 1 Labia majora overall appearance (A) before and (B) six months after treatment with PLLA-SCA. Patient #6 (table 1); 48 years old. Improvement of the skin flaccidity, roughness, and overall appearance of the labia majora after the treatment with PLLA-SCA.
|
|
Flaccidity |
Global improvement |
|
Very much improved |
0 (-) |
0 (-) |
|
Very improved |
7 (78%) |
7 (78%) |
|
Improved |
2 (22%) |
2 (22%) |
|
Neutral |
0 (-) |
0 (-) |
|
Worse |
0 (-) |
0 (-) |
Table 2 Evaluation of standardized images concerning skin flaccidity and overall improvement following three treatments with PLLA-SCA in the labia majora and mons pubis (n = 9
The functional symptoms and their evolution one year after the completion of the procedures are presented in Table 3. Five (56%) women reported complaints related to skin flaccidity, and 86% of the reported symptoms either completely (++) or partially (+) improved after three treatments. Only vaginal dryness was not improved after treatment with PLLA-SCA.
|
Complaint |
N (%) |
Improvement |
|
Flaccidity |
2 (22%) |
++ |
|
Loss of volume |
1 (11%) |
+ |
|
Discomfort in sports |
1 (11%) |
+ |
|
Difficulties in sexual intercourse |
2 (22%) |
++ |
|
Vaginal dryness |
1 (11%) |
- |
|
No symptoms |
2 (22%) |
|
Table 3 Functional complaints and their evolution one year after the completion of the treatment with PLLA-SCA in the labia majora and mons pubis (n = 9)
(++) complete improvement; (+) partial improvement; (-) no improvement.
Four women (44%) experienced adverse events after the procedure, including increased sensitivity in the labia majora (22%), mild pain in the labia majora (11%), and hematoma in the region of the labia majora (11%). There were no moderate or severe adverse events. No serious adverse events were reported. All adverse events were considered mild and transient; however, recorded data was available only for the mild pain in the labia majora, which resolved within three days.
In this retrospective trial, aesthetic and functional improvements were achieved after three treatments of the labia majora and/or mons pubis with PLLA-SCA. The mild and transient adverse effects reinforce the safety and tolerability of the product for this region.
PLLA-SCA has earned widespread recognition for its consistent effectiveness in facial rejuvenation, especially in combating volume loss, skin flaccidity, atrophic scars, and changes due to lipoatrophy in the treated area, with extensive evidence of its effectiveness and safety.25,26 Nevertheless, the versatility of PLLA-SCA extends beyond the face, with reports documented for areas such as the hands, neck, arms, abdomen, buttocks, thighs, and more.25,27–32
Aesthetically, female genitalia varies significantly among women, and while beauty standards for female genitalia remain ill-defined, the growing interest in genital beautification reflects a broader cultural shift towards body positivity and self-expression.33–35 Individuals are increasingly empowered to make choices that align with their personal preferences, challenging societal norms and expectations. Therefore, genital beautification represents a nuanced and evolving field within aesthetic medicine, offering individuals the opportunity to enhance both the appearance and functionality of the genital region.5,6
Injectable hyaluronic acid fillers and collagen biostimulators in the skin of the labia majora and mons pubis have gained attention in recent years as therapeutic options for enhancing skin volume and addressing skin flaccidity.3,11,16–18 In this study, we demonstrated that three sessions of PLLA-SCA treatments resulted in an improvement in flaccidity, firmness, and signs and symptoms associated with the aging of the labia majora and mons pubis in all participants.
Publications involving aesthetic and functional improvements following procedures on female genitalia emphasize the significance of research investigating aesthetic procedures, like PLLA-SCA, in this region. A retrospective study evaluating the efficacy and safety of injectable hyaluronic acid for augmenting the labia majora included 37 participants with a mean age of 45 years, all experiencing hypotrophy of the labia majora. Following the treatment, significant improvements were observed. Moderate elasticity was noted in 84% of patients, and sufficient elasticity of the labia majora was observed in 14%, marking a clear enhancement compared to the pre-procedure evaluation. The initial assessment revealed little elasticity in 70% of participants and a lack of elasticity in 16% of cases. Aesthetic satisfaction was also measured before and after treatment, with 81% of participants reporting being very satisfied and 19% expressing high satisfaction. These findings underscore the positive impact of injectable interventions in addressing aesthetic concerns and enhancing patient satisfaction.21
A systematic review examining the therapeutic approaches to augmenting the labia majora of the external female genitalia from 2006 to 2017 revealed that the most frequently employed technique was fat grafting, as reported in four articles encompassing 183 participants, despite surgical techniques were also prevalent.2 Additionally, two articles within the same review highlighted the use of hyaluronic acid. Notably, no significant differences in adverse events were reported among the observed techniques, underscoring the safety of these procedures and indicating a growing trend in their implementation.2
Adverse events associated with PLLA-SCA treatments are typically non-specific and may include pain, bleeding, erythema, edema, and allergic reactions, among others. Late complications, such as chronic inflammation and nodule formation, can occur, often attributed to improper product reconstitution or injection.36,37 In our study, there were no reported events involving nodule formation, irrespective of the dilution volume and hydration time. However, further investigations exploring these complications and reconstitution (hydration) times are warranted to enhance the safety profile of these procedures.38
This study has limitations inherent to retrospective design such as some missing data, the absence of a control group, and a relatively small sample size. Nevertheless, considering that genital aging is not expected to naturally reverse without intervention, the observed improvement in all participants serves as compelling evidence for the effectiveness of the treatment.39
The treatment of extra facial areas, including buttocks, abdomen, and arms, for enhancing body contour has become a common request in aesthetic practice. Minimally invasive procedures are preferable due to reduced downtime and minimal risks.40 However, additional research is required to gain a deeper understanding, particularly regarding the longevity of PLLA-SCA in non-facial areas such as the female genitalia. Additionally, there is a need to establish injection protocols aimed at maximizing optimal aesthetic results for patients in these specific areas.41
Procedures involving the injection of PLLA-SCA in the mons pubis and labia majora are well-tolerated and effective for reducing flaccidity and enhancing skin firmness. Additionally, these procedures may also have the potential to mitigate functional complaints associated with genital aging. Further prospective studies on collagen biostimulators in mons pubis and labia, using randomized controlled designs and larger sample sizes should be performed to maximize the results on genital beautification.
The patients in this study who gave written informed consent to the publication of their case details and images. Medical writing and editorial assistance were provided by Dr. Hélio Miot (Unesp, Botucatu-SP, Brazil) and was supported by Galderma Brasil Ltda., according to Good Publication Practice guidelines.
Informed-consent statement: Written informed consent was obtained from patients to publish their data and images in this paper.
Ethics statement: The study was approved by the Research Ethics Committee of the Hospital Moinhos de Vento (CAAE: 69127223.3.0000.5330). All procedures in studies involving human participants were performed in accordance with the Declaration of Helsinki (as revised in 2013).
Clinical trial registration: NCT05940038
Data sharing statement: The raw data from this study is available under contact with the corresponding author and a suitable reasoning.
Competing interests
Drs Hexsel, Del Nero, Parada, and Guarnieri are speakers, consultants, advisors, and investigators for Galderma Brazil. Dr. Kamamoto and Dr. Tomaz Gomes are Medical Managers of Galderma Brazil, BSc.Dr. Nogueira is the Medical Director of Galderma Brazil.
Galderma Brasil Ltda funded the trial development and the medical writing assistance. Despite this support, the study’s protocol and this manuscript were conducted with autonomy and independence by the authors.
The authors has no conflict of interest.

©2026 Hexsel, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.
 On the occasion of the International Day to End Obstetric Fistula (May 23rd), we join the global effort
to raise awareness about obstetric fistula, a preventable childbirth injury that continues to affect women worldwide. Often resulting from
prolonged, obstructed labor without timely medical care, obstetric fistula leads to severe physical and social consequences, yet it is both
preventable and treatable.
To highlight the importance of maternal health and to support this vital campaign, we are offering a 40% discount on any article submitted
to OGIJ. Together, let’s raise awareness, advocate for safe childbirth practices, and champion the rights of women to live with dignity and
access quality healthcare.
On the occasion of the International Day to End Obstetric Fistula (May 23rd), we join the global effort
to raise awareness about obstetric fistula, a preventable childbirth injury that continues to affect women worldwide. Often resulting from
prolonged, obstructed labor without timely medical care, obstetric fistula leads to severe physical and social consequences, yet it is both
preventable and treatable.
To highlight the importance of maternal health and to support this vital campaign, we are offering a 40% discount on any article submitted
to OGIJ. Together, let’s raise awareness, advocate for safe childbirth practices, and champion the rights of women to live with dignity and
access quality healthcare.