eISSN: 2377-4304


Case Series Volume 12 Issue 6
1Plastic & Reconstructive Surgery, USA
2Hoag Gynecologic Oncology, USA
3Plastic surgeon, USA
4Newport Irvine Surgical Specialists, USA
Correspondence: Brian P Dickinson, MD, 351 Hospital Road, Suite 415, Newport Beach, CA 92663, USA, Tel 949-612-8632, Fax 10-861-1478
Received: October 26, 2021 | Published: November 15, 2021
Citation: Prajapati KP, Vu MB, Li ED, et al. Reconstruction of pelvic exenteration and gynecologic tumor defects: a pictorial essay for a systematic method. Obstet Gynecol Int J. 2021;12(6):364-373. DOI: 10.15406/ogij.2021.12.00607
Background: Reconstruction of the defects created by extirpative surgery or radiation for gynecologic cancer can be challenging for both patients and surgeons. In the subset of patients who present with recurrent gynecologic cancer after previously successful treatment and radiation therapy, wounds to irradiated tissue are difficult to heal, requiring a collaborative effort between the gynecologic oncologists and the plastic and reconstructive surgeons. Other factors that may hinder successful repair include comorbidities, history of radiation, ongoing tobacco use, and overall protein malnutrition. To maximize repair success, decrease recurrence, and optimize aesthetic outcome, gynecologic oncologists and plastic and reconstructive surgeons need to communicate the anticipated anatomic and physiologic obstacles to each other and to the patient.
Purpose: To create a systematic approach from our current method of gynecologic oncologic reconstruction that facilitates communication among general surgeons, gynecologic oncologists, and plastic and reconstructive surgeons to optimize gynecologic oncologic reconstructive and aesthetic outcomes while minimizing wound complications.
Methods: A retrospective chart review was conducted on patients who underwent gynecologic oncologic surgery for vulvar, vaginal, cervical, and uterine cancer over a 10-year period. A step-by-step method and categorization for gynecologic reconstruction was generated from our experience, and a pictorial essay was created to demonstrate this method and highlight the most common complications.
Results: The pictorial essay serves as a template for gynecologic oncology or plastic and reconstructive surgeons to follow and effectively close gynecologic tumor defects and facilitate patient care. The most common complications were seroma and distal skin necrosis, managed by early excision and closure without consequence. Satisfaction with surgical outcomes was high among the patients.
Conclusion: Successful repair of primary and recurrent gynecologic tumors requires communication between the gynecologic oncologist and the plastic and reconstructive surgeon. The procedures are challenging and require resilience from both the patient and the surgeons. Incision placement is important for adequate flap reconstruction to prevent wound and skin breakdown. Wound breakdown is likely and requires preoperative counseling. High protein nutritional stores are important for expeditious healing.
Keywords: oncoplastic, gynecologic, oncologic, cancer, reconstruction
Gynecologic oncologists treat malignancies of the vulva and female reproductive tract with surgical excision and/or radiation. While effective excision is important, it is equally important to take into account the patient’s desire to maintain sexual and/or reproductive function and to have an aesthetically pleasing result following reconstructive surgery. Loss of libido, dyspareunia, or disfigurement to the external genitalia after gynecologic resection of tumors can potentially pose psychological challenges to the patient and affect their interpersonal relationships.1 Patients may develop body image disorders, as well as anxiety or depression, following surgery.1
Pelvic exenteration and reconstruction are procedures in which gynecologic oncologists, colorectal surgeons, and plastic and reconstructive surgeons work closely together to both treat the gynecologic cancer and reconstruct the genitourinary, intestinal, and perineal structures. Pelvic exenteration and reconstruction are a major undertaking for both patients and surgeons, as oncologic and aesthetic outcomes must be optimized. Not only are multiple organs removed from the pelvis, but the procedure may also be the patient’s first experience with gynecologic cancer. Advanced stage disease may have created wounds or fistulas in the urinary, gastrointestinal, or integumentary systems, and patients may be at the “end of their rope,” yet need to undergo a significant surgery to treat their cancer.2
Alternatively, some patients may already have started treatment for cancer and have organ systems that have received radiation prior to surgery. Medical optimization of these patients is paramount, as organ systems will need to be reconstructed in a previously treated field, and any intra-abdominal surgery will require at least an initial period of NPO status prior to the return of bowel function. Furthermore, protein optimization is critical, as there are often multiple anastomoses performed, as well as creation of a significant amount of dead space. Hypoalbuminemia due to poor protein intake impedes wound healing, contributes to increased edema in the postoperative period, and elevates risk for seroma formation in the subcutaneous and intra-abdominal spaces.3,4
To maximize repair success and decrease postoperative complications, gynecologic oncologists and plastic and reconstructive surgeons must communicate the foreseeable anatomic and physiologic challenges and review CT-scans to formulate successful flight plans for surgery. Careful preoperative planning of the incision location, assessment of the blood supply to the flap, and patient education on wound healing can help achieve patient goals while successfully eradicating the cancer.5,6 Such collaboration and planning can therefore optimize the patient for success, even in the face of previous radiation, comorbidities, or potential postoperative obstacles.5,6
Significant postoperative complications include bowel obstruction and intra-abdominal urine leak from ureteral-ileal conduit anastomosis. Providing soft tissue bulk to the pelvis can prevent the bowel from adhering to a denuded pelvic floor and also provide a stable structure for ureteral-ileal conduits to rest on, limiting tension and migration and ultimately preventing urine leak.7
Tumor reductive surgery is not a typical component of plastic and reconstructive surgeons’ curriculum. A plastic surgeon in training may spend significant time training in microvascular or hand surgery, with little interaction with academic gynecologic oncologists. It has been the senior author's experience that oncoplastic reconstruction of the breast has made significant gains in the past several years, which has improved oncologic and aesthetic outcomes of women who have breast cancer.8–10
The impetus for writing this paper comes from the senior author's experience closing defects for a high-volume gynecologic oncology practice and finding reproducible and teachable techniques to approach the most commonly encountered gynecologic malignancies. This paper can hopefully serve as a guide for plastic and reconstructive surgeons and/or gynecologic oncologists to resect and reconstruct the female reproductive tract in a way that maintains the sexual and psychological self-esteem of the patient and minimizes complications to maximize quality of life. The plastic and reconstructive team of this paper appreciates all they have learned from the gynecologic oncologist and colorectal surgeon on this manuscript.
A retrospective chart review was conducted on all patients who underwent gynecologic reconstruction following extirpative surgery by the primary author between 2008 and 2021. Charts were reviewed and categorized by the most commonly presenting clinical scenarios and then surveyed for the most common challenges affecting the gynecologic oncologic reconstruction. Additionally, preoperative, intraoperative, and postoperative photos were examined.
The preoperative and postoperative courses of complex cases were analyzed and grouped into clinical scenarios. Intraoperative methods and maneuvers were evaluated alongside the postoperative results. The techniques and management of pelvic exenteration, as well as the methods used to address commonly encountered problems, were further described in a photographic demonstration, so that the procedure could be easily reproduced by an oncologic reconstructive team.
Satisfaction was determined by patient response and communication with the gynecologic oncologists who performed surveillance examinations postoperatively.
In the twelve-year period, over 50 patients underwent local flap reconstruction or locoregional flap reconstruction following surgery for a gynecologic or colorectal malignancy.
The most common categories of reconstruction were:
Patient satisfaction was high among those who underwent primary reconstruction of the gynecologic defect. Satisfaction was also high for patients with recurrent disease; this subgroup of patients had the opportunity to revise anatomy that had become distorted or contracted after radiation.
The most common complications postoperatively were seroma and partial skin necrosis, which was treated with early debridement and closure. In the previously irradiated group, the rate of flap separation was high, but the wounds healed with proper nutrition, wound revision, and hyperbaric oxygen. In the primary cancer group treated with surgical excision, there were no significant treatment delays in those patients who needed additional therapy.
Gynecologic tumors can be challenging for both the surgical team and the patient. When recurrence occurs, the patient may become frustrated, and it is important for the treating physician to counsel and to continue to instill confidence in the patient. When planning these complex surgical procedures with possible abdominal wall morbidity, following a systematic method optimizes safety, the restoration of perineal and genital health, and aesthetic outcomes.
Prior to surgery, the most common postoperative complications, such as seroma and wound dehiscence, and risk factors, such as elevated BMI, are explained to the patient. In general, in this operative field and in a patient population that may have high BMI and/or history of radiation, wound separations are common.8 The perineum is an area with motion, dependency, and moisture, and in many cases, the cancer or planned flap is in an intertriginous location. Fortunately, these wounds will usually heal with local wound care, hyperbaric oxygen, and wound revision. It is important to communicate preoperatively with patients that wound separations are common and that additional surgery may be required. Although the decision tree to close oncologic defects can be complex,9 the stepwise method detailed below offers a consistent approach.
Step 1: Identifying the location and size of the tumor
Often, the gynecologic oncologist has examined the patient in the office in the lithotomy position and has a direct understanding of the location and palpable size of the gynecologic malignancy. The plastic and reconstructive surgeon may not have an office setup with the same physical exam capabilities, e.g. stirrups, speculum, etc., but it may be possible for the participating surgeons to examine the patient as a team in the gynecologic oncologist’s office. It is ideal that the plastic and reconstructive surgeon has the same understanding of the location and size of the tumor as the gynecologic oncologist. Attention should be directed towards the laxity of the surrounding tissue in the perineum, which can change depending upon the patient’s history of radiation. It has been the primary author's experience that irradiated skin of the perineum can be rotated with wide based flaps or thick subcutaneous flaps to facilitate wound closure of defects.
After determining the size and location of the tumor, the following general rules are utilized to guide the reconstructive process:
Step 2: Review of the preoperative CT-scan
Review of the CT-scan is important in pelvic reconstructive surgery. The dimensions of the pelvis must be noted, as this is the area to be filled by the VRAM flap to prevent small bowel from adhering to the denuded pelvis. The pelvic inlet should be measured, and the configuration should be transferred to the skin paddle on the abdominal wall. This flap is used to close the pelvic outlet, support the bowel, and prevent a perineal hernia.
In addition to assessing the dimensions and caliber of the rectus muscle, identifying the presence of any concurrent abdominal hernias is crucial. Removal of the rectus muscle and the rectus fascia can make other defects of the abdominal wall much larger. The location and dimensions of the hernia, usually the widest part, should be determined. Hernias close to the umbilicus can often be easily closed, as there is a greater degree of laxity of the abdominal wall in the periumbilical region, compared with the costal margin or iliac regions.
The presence of an ostomy at the abdominal wall can make closure of the abdominal wall much more difficult. Not only is there less room for the placement of sutures, but the excessive tension near the ostomy can also inadvertently obstruct the opening or kink the bowel, affecting the patency of the ostomy.
Step 3 Procedural steps
Group 1: Reconstruction of the Labia
Case 1: (Figure 1) When tumors occur in the posterior labia and when there is redundant tissue anteriorly, the procedure performed to close the defect is identical to an aesthetic labiaplasty. Once the tumor is excised from the posterior labia, the posterior surface of the redundant anterior labia is excised, and the anterior labia are sutured to the defect on the posterior labia. The resultant suture line lies posteriorly.
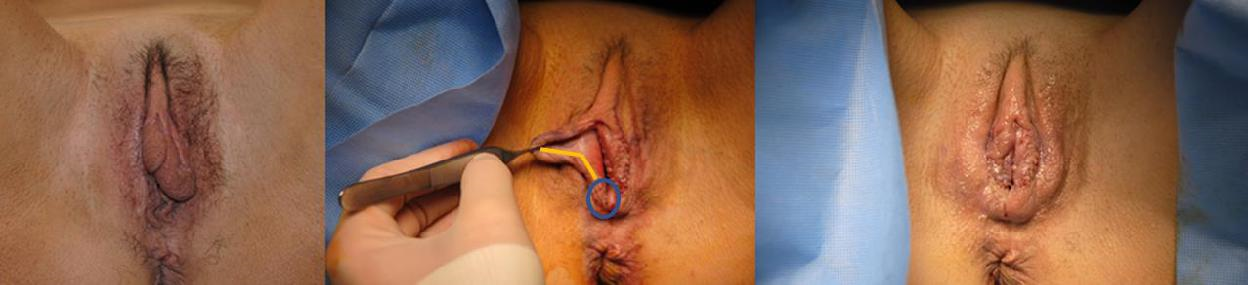
Figure 1 Tumors of the posterior labia can be reconstructed with redundant anterior or mid labia, and the pattern is identical to the pattern used in an aesthetic labiaplasty. The blue circle on the right labia indicates the location of the tumor. The yellow line indicates the portion of the labia to be excised and sutured to the posterior defect. A labiaplasty is then performed on the contralateral labia for symmetry.
Group 2: Tumors of the distal vaginal canal and labia in patients without previous radiation
Case 2: (Figures 2a–2d) Tumors of the distal vagina may require rotation advancement flaps to prevent contracture of the vaginal vault; direct closure of the vaginal defect often results in vaginal stricture and dyspareunia after radiation therapy. The labia minora may not be large enough to rotate into the vaginal vault. When perineum has not been previously irradiated, a medial thigh rotation advancement flap is an effective option for closure. In the setting of previous radiation therapy, a medial thigh rotation advancement flap can still be considered if the skin of the medial thigh is of good quality. These flaps have a robust blood supply, and wound dehiscence secondary to blood supply is not common. However, seroma formation or wound dehiscence associated with maceration or motion may necessitate a return trip to the operating room, so that chemotherapy or radiation therapy can be initiated in a timely manner, as needed. These wound dehiscences heal quickly with repeat closure with chromic or Vicryl sutures.
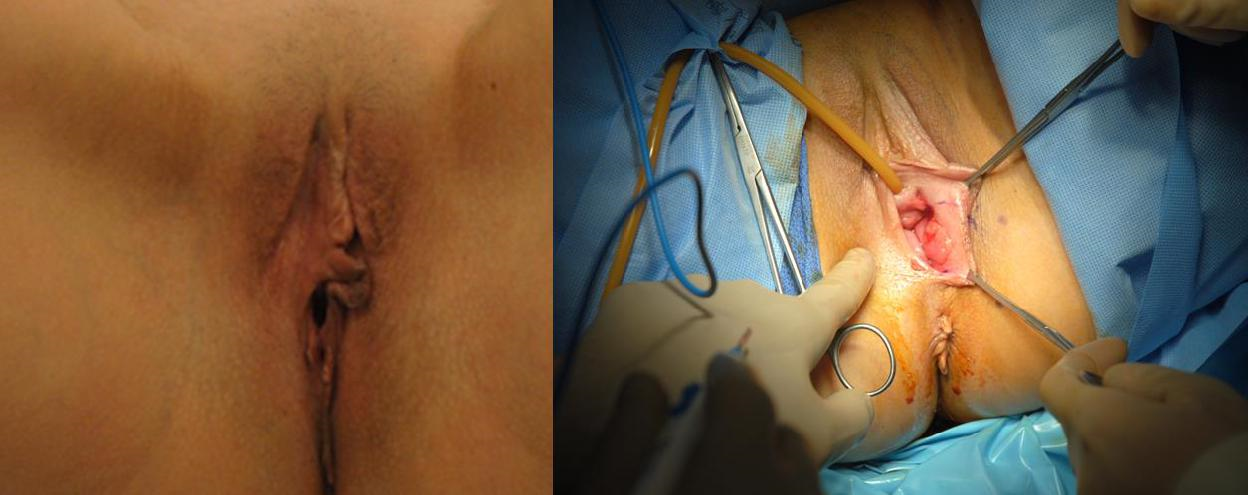
Figure 2a Tumors of the distal vaginal canal can be reconstructed with medial thigh skin. The tumor can be readily seen on the left wall of the vagina.
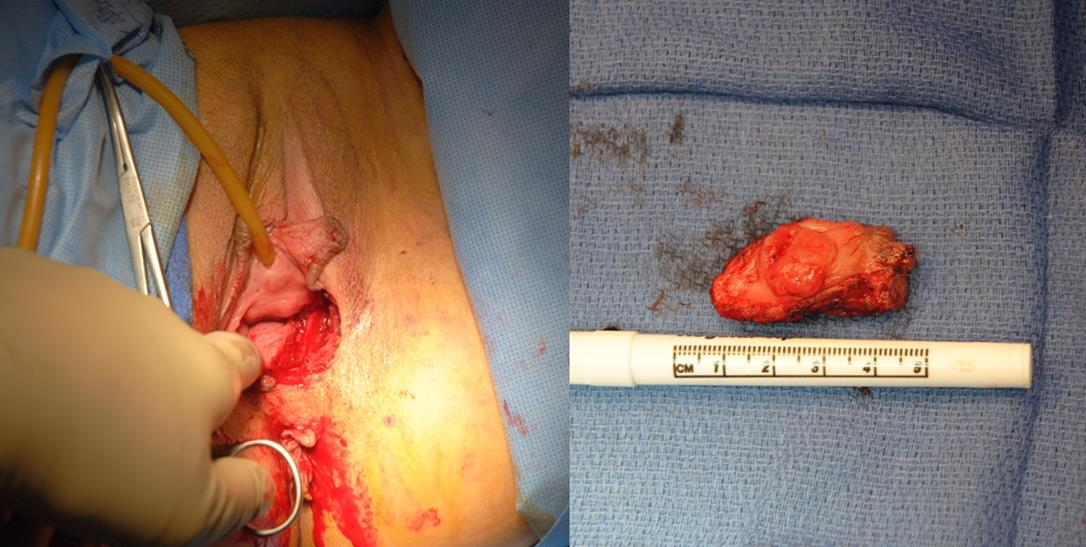
Figure 2b After the tumor has been resected, small areas of the defect that can be closed primarily without distorting the anatomy are closed. This often restores normal anatomical landmarks.

Figure 2c A right labiaplasty is performed to match the oncologic resection of the left side. Once symmetry of the labia minora has been established, a flap is drawn from the left medial thigh to correct the vaginal defect.
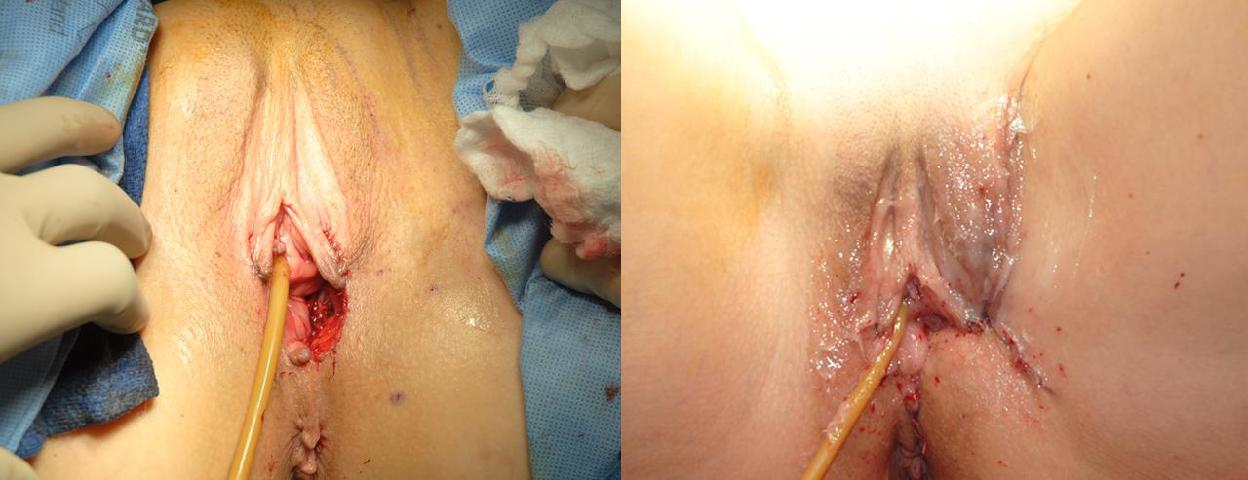
Figure 2d Arterial and venous perforators are located with handheld Dopplers at the base of the medial thigh flap. The thigh flap is elevated and then rotated into the vaginal defect. This prevents circumferential scar formation. The donor site is then closed in a straight line.
Case 3: (Figures 3a–3k) Tumors of the distal vagina that do not include the labia can also be repaired with the medial thigh rotation advancement flap. After the tumor is resected, the defect is measured, and the dimensions are transposed onto the medial thigh flap. When the vulva is not involved, the flap can be deepithelialized and tunneled beneath the vulva and into the distal vagina, so as to not disrupt the appearance of the external genitalia.
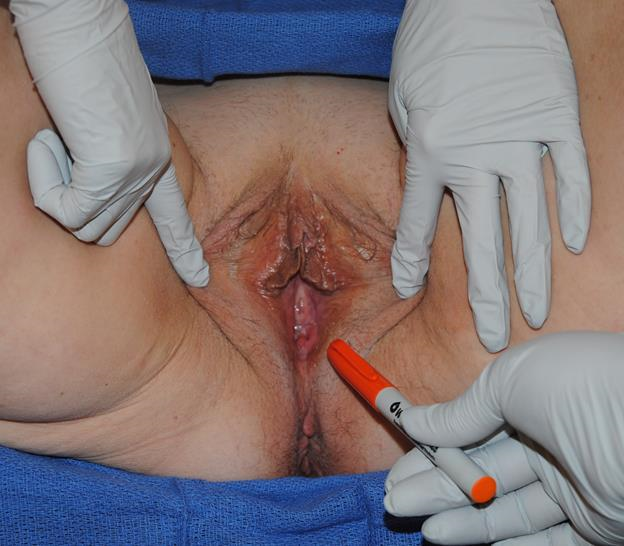
Figure 3a A squamous cell carcinoma is present at the distal vaginal canal. Excision of the squamous cell and direct closure would cause significant stenosis of the vaginal canal. The patient has redundant tissue of the medial thigh that can be rotated into the vaginal defect.
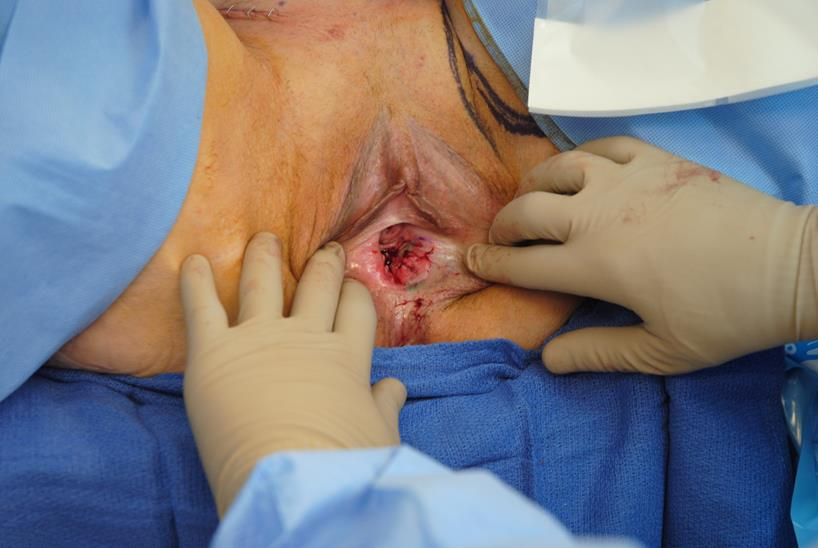
Figure 3b Upon closer examination, the lesion is seen extending proximally into the vaginal canal. Therefore, an extended medial thigh flap is needed to close the defect.
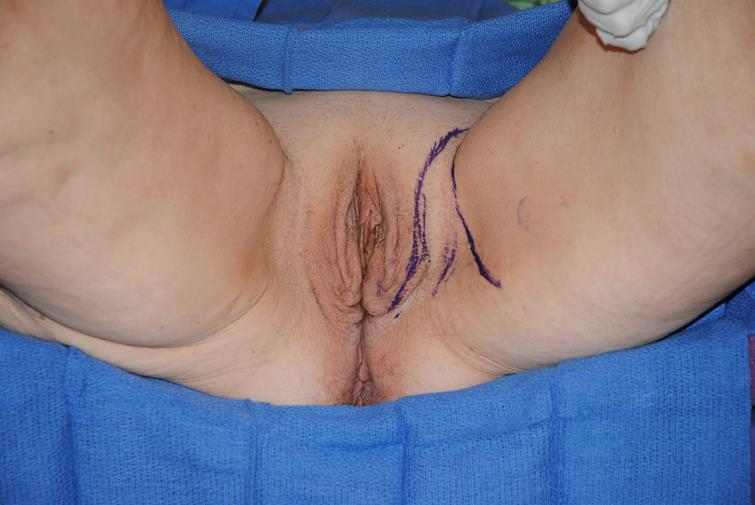
Figure 3c A medial thigh rotation advancement flap is drawn out on the inner aspect of the medial thigh, to be elevated and transferred into the vaginal canal.
Figure 3f The bilateral groin incisions are closed without impacting the medial thigh rotation advancement flap.

Figure 3h The medial thigh rotation advancement flap is incised along its periphery, preserving vascular perforators at the base.
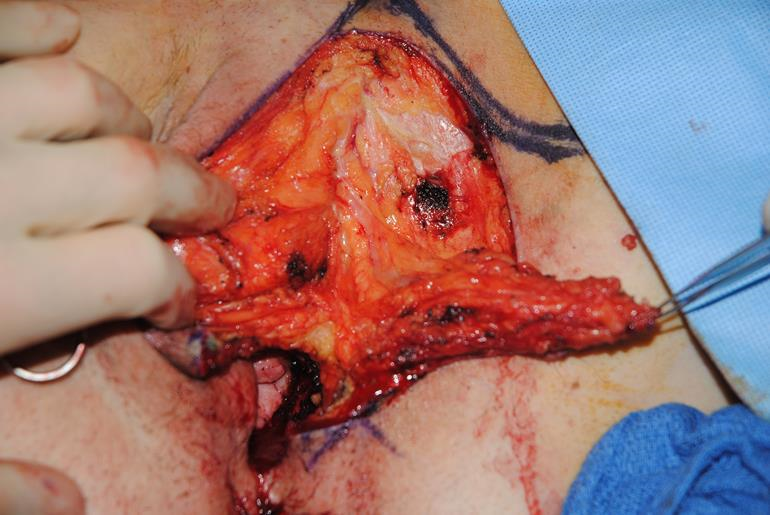
Figure 3i The flap is dissected away from the perineal soft tissues with the perforators at the base of the flap kept intact.
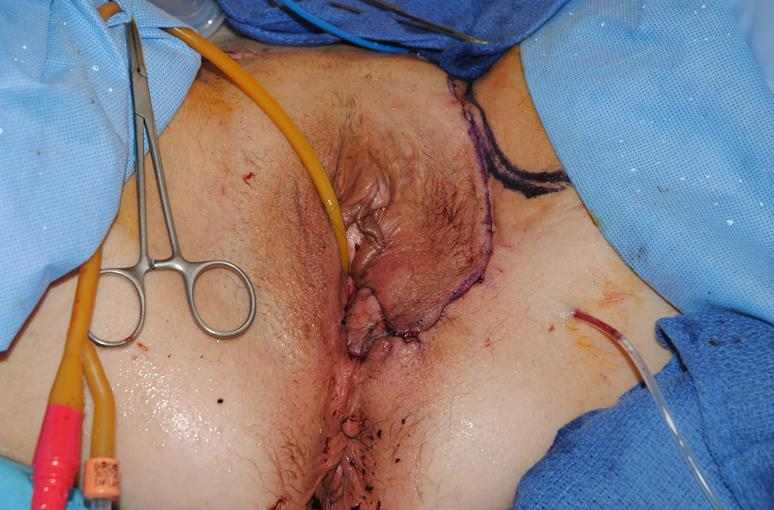
Figure 3k The flap is deepithelialized and tunneled into the vaginal canal for wound closure. The donor site is then closed.
Case 4: (Figures 4a–4c) The medial thigh rotation advancement flap can fix defects that extend laterally with respect to the vulva. When designing the flap, keeping the flap wide at its base incorporates more vascular perforators and results in a heartier blood supply. The inferiorly based flap is useful because venous drainage of the flap is dependent when the patient is supine, thus preventing venous congestion.

Figure 4a The medial thigh rotation advancement flap is kept wide at its base to incorporate a greater number of perforators.
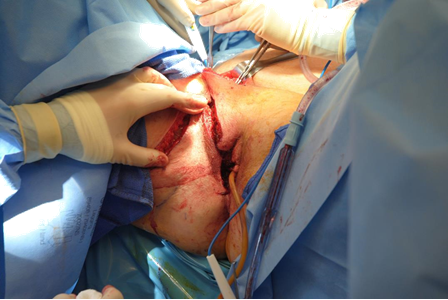
Figure 4b The entire flap base is rotated into the vaginal defect. Excess bulk initially is beneficial in patients who are projected to undergo radiation afterward. Suction assisted debulking of the flap can be performed one year after completion of radiation therapy, if needed.
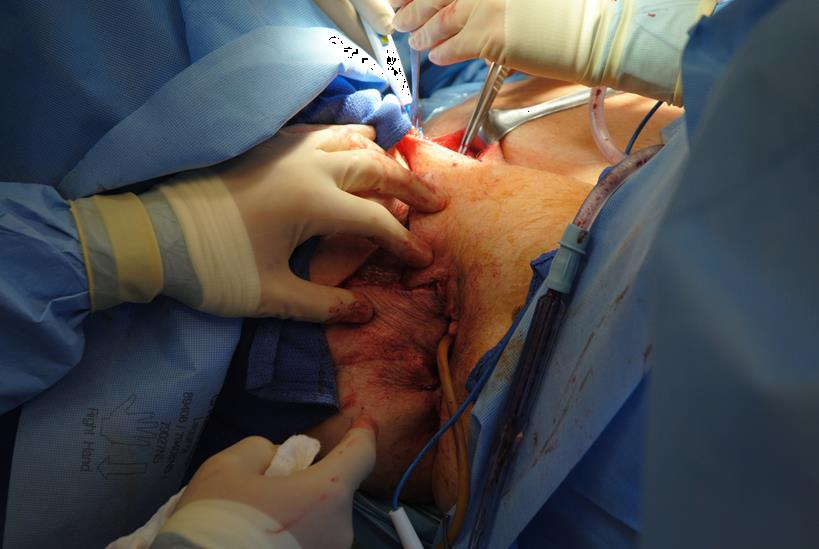
Figure 4c A closed suction drain or penrose drain is placed in these positions to prevent seroma formation and local infection.
Group 3: Tumors of the distal vaginal canal and labia in the setting of previous radiation or posterior extension
Case 5: (Figure 5a–5c) In patients who are status post excision and radiation for previous gynecologic malignancies, the adjacent skin may not have adequate elasticity and/or blood supply to be rotated into the soft tissue defect. In these cases, the gracilis myocutaneous flap can be rotated into the vaginal defect. The gracilis myocutaneous flap is a robust flap that is out of the irradiated field, has a lax donor site for easy closure, and provides ample tissue to reconstruct the entire length of the vaginal canal or only a segment if needed. The gracilis muscle is in the same operative field when the patient is in lithotomy position and can be easily harvested to reconstruct vaginal defects.
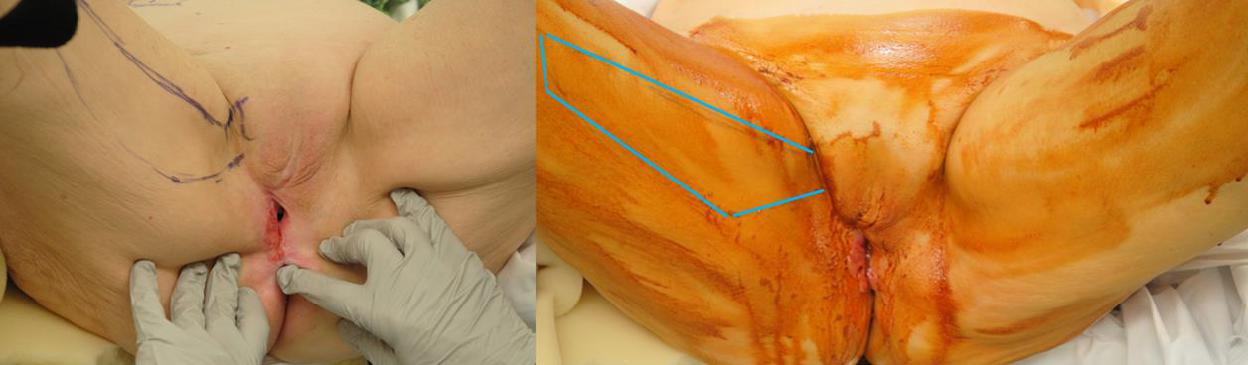
Figure 5a The gracilis muscle is palpated along the length of the medial thigh. The blue outline demarcates the gracilis myocutaneous flap.

Figure 5b The tumor is resected from perineum. The remaining local tissue is taut with poor elasticity. The gracilis myocutaneous flap marked in blue on the patient’s right side is then elevated and used to cover the vaginal canal, as well as the outer perineal skin defect.
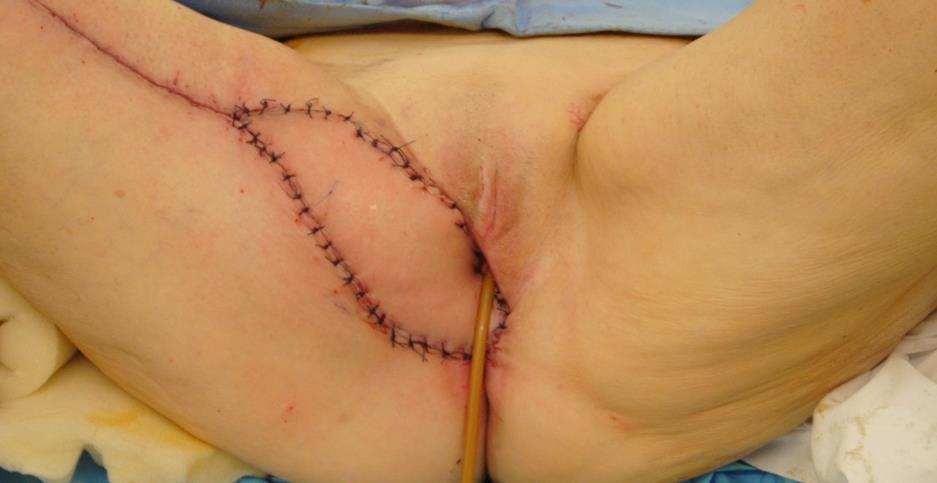
Figure 5c The donor site on the medial thigh is closed primarily. The gracilis myocutaneous flap demonstrates excellent capillary refill.
Group 4: Extensive recurrent disease
Case 6: (Figures 6a–6h) In patients who have undergone previous vulvar excision and perineal radiation, closure of defects is challenging. In some patients, the skin has tolerated radiation well, while in others, elasticity and blood supply are diminished, increasing the risk of wound dehiscence.

Figure 6a Rotation of irradiated tissue is challenging, as the skin does not have normal elasticity and blood supply, and the rate of wound dehiscence is quite high.
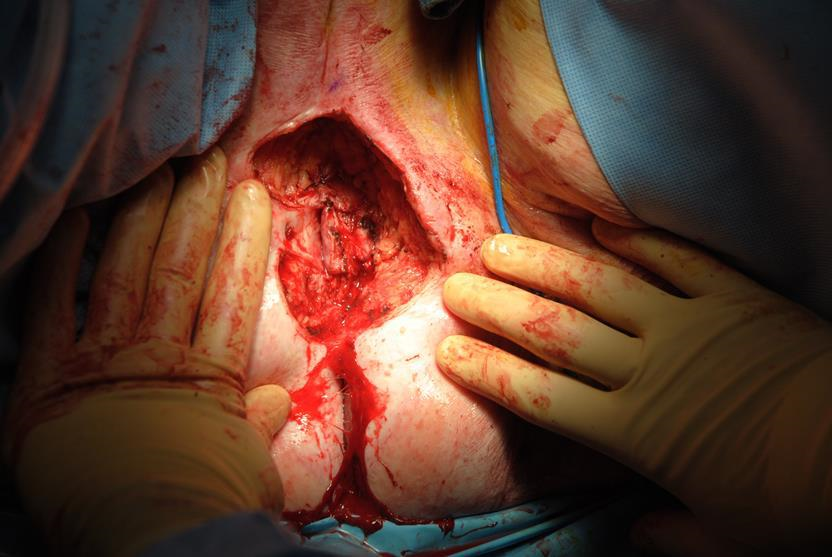
Figure 6d In irradiated tissue, when the skin and tumor are excised, the contraction forces on the wound bed become apparent and the resulting wound is much larger than originally marked.
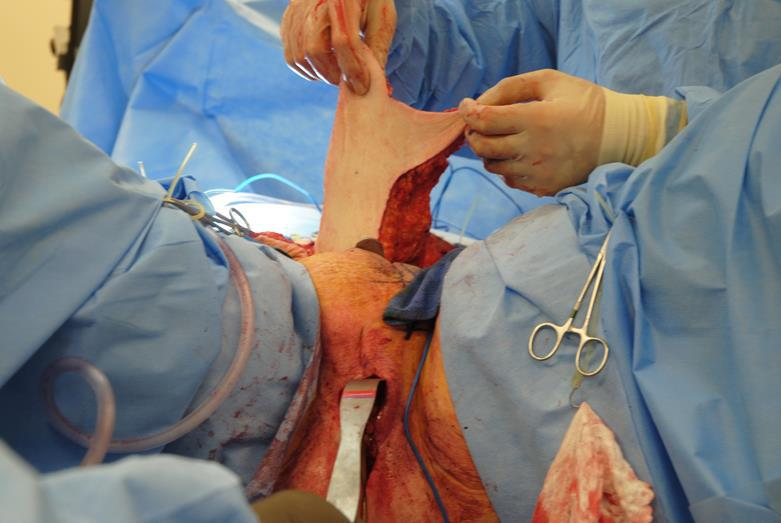
Figure 6f The vertical abdominis rectus myocutaneous flap is then tunneled anterior to the pubic symphysis to the vulvovaginal defect.
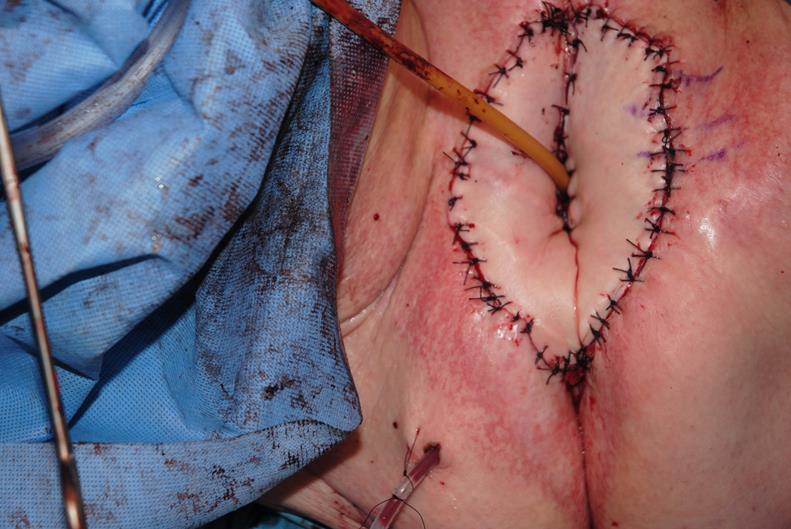
Figure 6h Control of the urine stream is helpful for preventing maceration and wound breakdown in the perineal region. This is accomplished with a Foley catheter, a suprapubic catheter, or bilateral nephrostomy tubes. Urinary diversion is important for patients who undergo abdominoperineal resection or pelvic exenteration where the bladder is maintained, as urinary continence may be temporarily or permanently affected.
Group 5: Pelvic exenteration
Case 7: (Figures 7a–7p) Pelvic exenteration is commonly performed for patients with advanced uterine or cervical cancer that involves the bladder or other structures of the pelvis. In patients requiring this procedure, the most important aspect is to obliterate the dead space of the pelvis to prevent the migration of small bowel into the denuded pelvis and lower the risk of bowel obstruction. The abdominal flap can also support a ureteral anastomosis and help prevent urine leak.

Figure 7a The vertical rectus abdominis myocutaneous flap is drawn on the abdominal wall. The flap is then tubed to reconstruct the vagina.
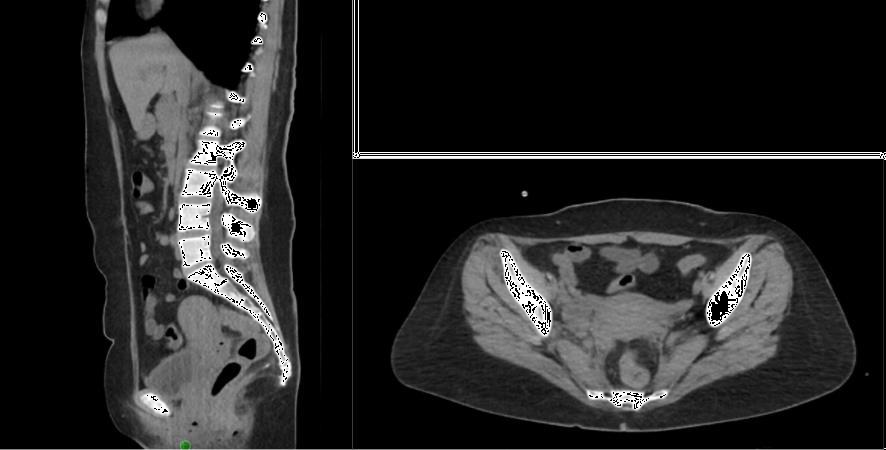
Figure 7b The width of the flap to be harvested is determined by measuring the diameter of the pelvic inlet.
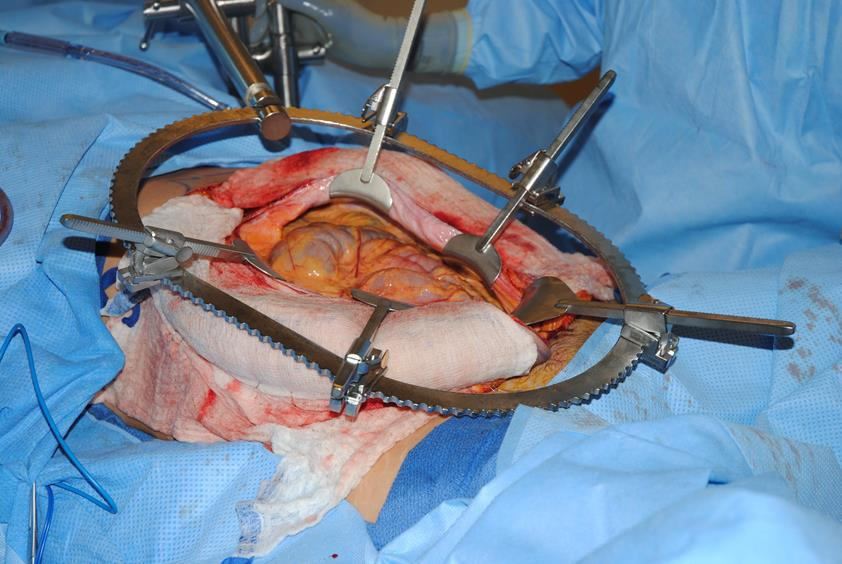
Figure 7f The flap is tucked out of the way laterally beneath a Buckwalter retractor. The posterior rectus sheath is opened, and the gynecologic resection is conducted via a longitudinal incision in the posterior sheath, which provides excellent exposure.
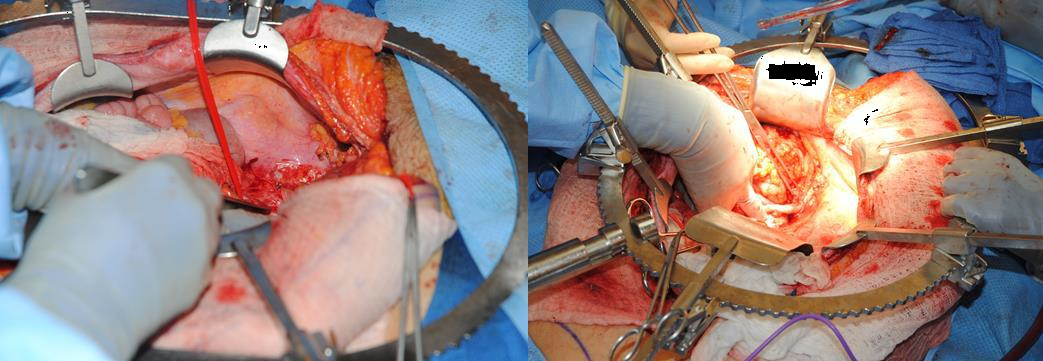
Figure 7g The pelvic exenteration procedure begins. The peritoneum is denuded from the pelvis, and a raw surface is created, which can promote bowel wall adhesions to the pelvis.

Figure 7h The specimen is resected, and the uterus and vagina are removed from the vulvar opening, creating an open communication to the pelvic cavity.
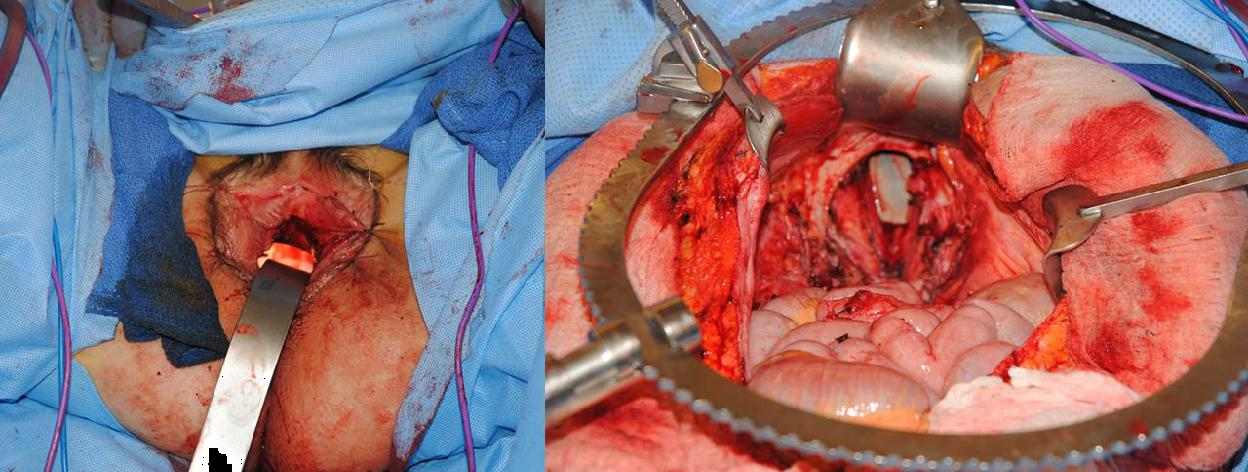
Figure 7i The pelvic cavity and vulvar opening are irrigated, and the flap is then prepared on the abdominal wall. The skin component of the myocutaneous flap is used to reconstruct the vaginal opening, while the muscle, adipose tissue, and deepithelialized component of the flap fill the pelvic space.
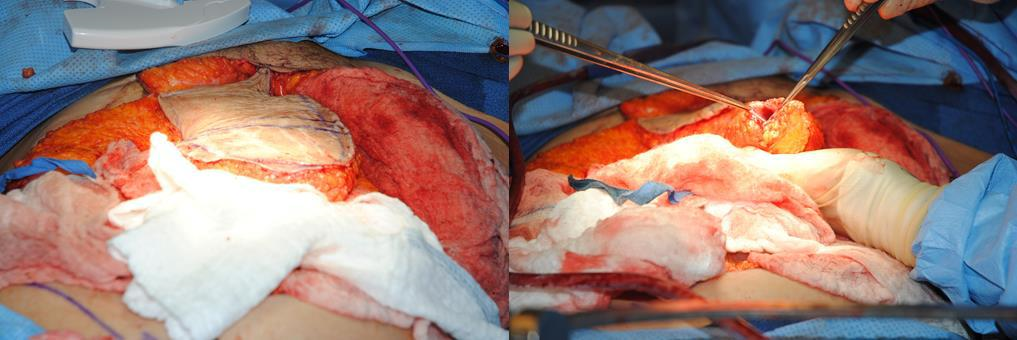
Figure 7j The flap is folded upon itself to create a tube, becoming the introitus and vaginal canal. The most superior aspect of the flap becomes the inferior end of the vaginal canal. The opening of the neo-vaginal canal is shown in the right image, held with Debakey forceps.
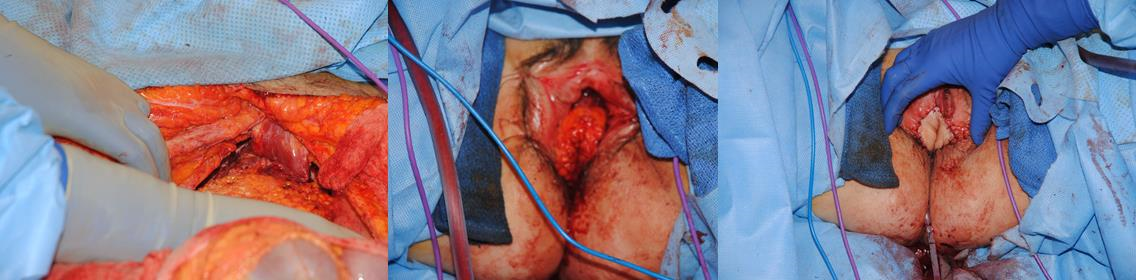
Figure 7l The flap is delivered into the pelvis. The rectus muscle folds upon itself, and the muscle and fat are placed into the pelvis and vaginal canal. The skin component is then sutured to the vulvar opening.

Figure 7m The ureters are isolated and sutured into the ileal conduit. The ureteral-ileal anastomosis can rest upon the subcutaneous portion of the flap. By stabilizing and taking tension off the anastomosis, the risk of urinary leak is decreased.

Figure 7n The abdominal wall is closed, and drains are placed in the subcutaneous space. Care is taken with closure of the abdominal wall fascia to prevent hernia and bulge formation.
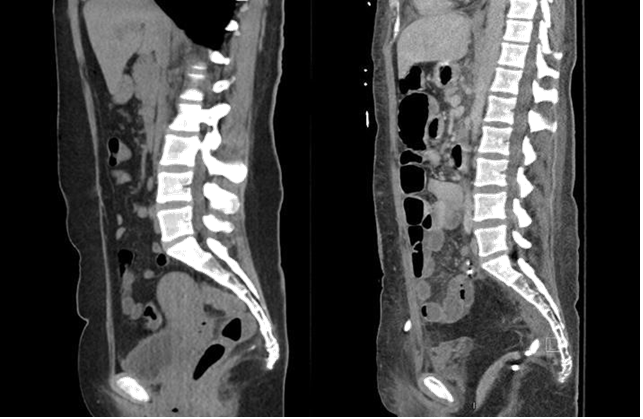
Figure 7o Comparison of the preoperative and postoperative CT-scans indicates that the abdominal fat nicely obliterates the pelvis and prevents the egress of bowel into the pelvis.
Case 8: (Figures 8a–8e) Anorectal cancers, when aggressive, can extend to the posterior vaginal wall. The treatment for these cancers is a variation of pelvic exenteration which may or may not include removal of the bladder. When the bladder is not involved, the vertical rectus abdominis myocutaneous flap can be used to close the anorectal defect. When the bladder does require resection, the flap demonstrated in the previous case can be utilized.
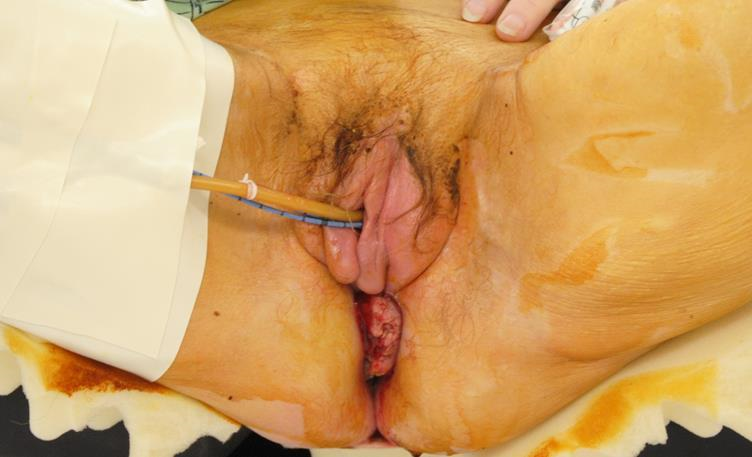
Figure 8a Anorectal cancers that encroach on the vaginal wall are resected with an abdominoperineal resection. The vertical rectus myocutaneous flap can be used to reconstruct the anorectal defect in conjunction with a vaginal wall defect.
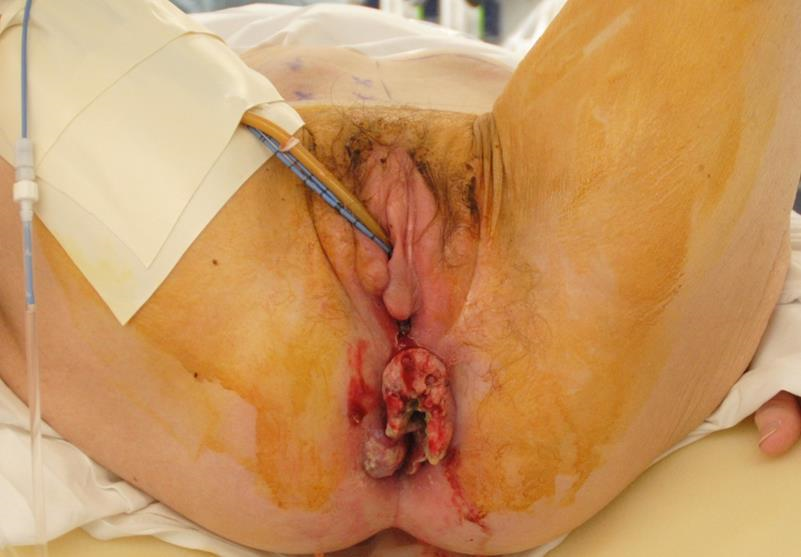
Figure 8b Further inspection of the anorectal tumor shows the fungating nature of the lesion. The dimensions of the lesion, plus an additional skin margin, is recorded. A skin paddle with these minimum dimensions is then harvested from the abdominal wall.

Figure 8c The donor site must be contralateral to the location of an existing or planned colostomy. In situations in which only one side can be used, it is particularly important to verify the integrity of the blood supply.
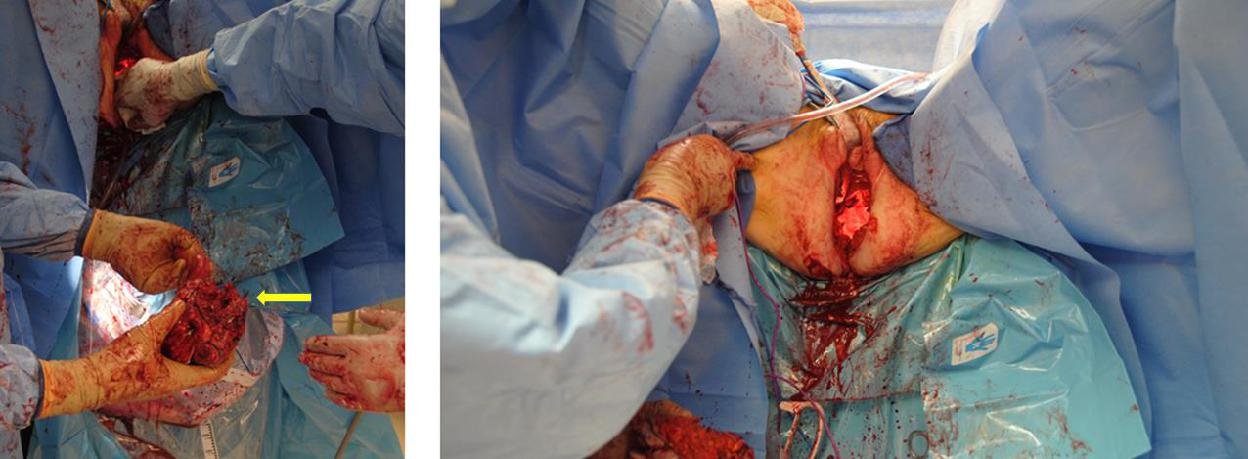
Figure 8d The anorectal specimen is resected (yellow arrow). The defect to be closed and the dead space in the pelvis are identified.
Step 4: Immediate and long term postoperative care measures
Close follow-up is recommended for flap success. Patients are instructed to shower two days after surgery and clean the wound with soap and water to decrease bacterial colonization, as hygiene of the intertriginous area is difficult to maintain. Patients are cautioned to avoid using caustic cleaning solutions, such as Hibiclens or hydrogen peroxide.
While ambulation is encouraged to decrease DVT occurrence, the dependent position of the flap when the patient is upright can increase the venous pressure and cause venous congestion of the flap. Therefore, when not ambulating, patients should remain supine to facilitate flap drainage.
For local flaps, it is necessary to keep the groin and perineum dry. Topical agents, including powders, may cause irritation that is difficult to differentiate from cellulitis. In the event of local fungal infection, intravenous fluconazole is superior to oral or topical antifungals. Consultation with infectious disease specialists may be helpful.
Successful reconstruction following surgery for gynecologic and colorectal malignancies with desirable aesthetic outcomes and minimal rates of recurrence can be gratifying for both physicians and patients. Optimal incision placement allows adequate exposure and lowers the chance of wound dehiscence. Additionally, skin breakdown and seroma formation can be minimized by maintaining blood supply and decreasing dead space. Results in this operative field are variable and often patient-dependent, but with appropriate systematic planning and experience, gynecologic oncology and plastic and reconstructive teams can yield consistent results.
None.
Availability of data and material: The chart and photographic review material are available from chart review from the office of Brian P. Dickinson, M.D., Inc.
Author’s contributions: The manuscript work presented has not been published before and it is not under consideration for publication anywhere else. The publication has been approved by all co-authors, as well as by the institute where the work has been carried out.
Consent to participate: The data collection for the publication was completed via chart review. Photographic consent was obtained from the patients in the chart review for presentation and publication.
Consent for publication. Photographic consent was obtained from the patients in the chart review for presentation and publication.
Author’s contributions: All authors have contributed to the publication of the manuscript.
There was no funding for the completion of the chart review or preparation of the manuscript.
The authors have no conflicts of interest and no competing interests.

©2021 Prajapati, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.