eISSN: 2377-4304


Case Report Volume 12 Issue 6
1Academia Mexicana de Cirugia y Academia Nacional de Medicina de Mexico
2Servicio de Ginecología Hospital Juárez de México
Correspondence: Víctor Manuel Vargas Hernández, Academia Mexicana de Cirugia y Academia Nacional de Medicina de Mexico, Insurgentes Sur 605-1403, Nápoles, 03810 CDMX, Mexico, Tel 5552179782, 55746647
Received: December 05, 2021 | Published: December 30, 2021
Citation: Manuel VHV, Fabiola MHV, Vargas-Aguilar VM. Non-gestational primary choriocarcinoma of the ovary. Presentation of a clinical case. Obstet Gynecol Int J,/i>. 2021;12(6):423-427. DOI: 10.15406/ogij.2021.12.00616
Background: Ovarian germ cell tumors are derived from the primordial germ cells of the ovary, they can be benign or malignant. Non-gestational ovarian choriocarcinoma is extremely rare and aggressive that are of gestational or non-gestational origin, its prevalence is less than 0.6% of all ovarian germ cell tumors. Due to the rarity of the tumor, there is a lack of information on the clinical-pathological characteristics, diagnosis and treatment.
Objective: present a case of non-gestational ovarian choriocarcioma and review of the literature
Clinical case: We present the case of a 20-year-old woman who presented with an acute abdomen, due to abdominal pain and distention, with scant vaginal bleeding and pain on cervical mobilization; An ultrasound was performed with a right annex with a lesion measuring 114 x83x79mm and a total volume of 394cc, heterogeneous with linear images inside punctiform and human chorionic beta-gonadotropin levels, elevated 112.337 mUI/mL, the patient underwent an exploratory laparotomy with the finding of an ovarian tumor; performing salpingo-oophorectomy and the histopathological report of the definitive surgical specimen and immunohistochemical study, the diagnosis of non-gestational ovarian choriocarcinoma was made.
Conclusion: Non-gestational choriocarcinoma is an extremely rare malignant neoplasm that can present clinically in different ways, even as an acute abdomen, which requires differential diagnoses and management is the combination of surgery and adjuvant chemotherapy.
Keywords: non-gestational ovarian choriocarcinoma, ectopic pregnancy, diagnosis and treatment
Primary ovarian choriocarcinoma may arise from gestational tissue or from pure germ cells of the ovary; it is known as gestational choriocarcinoma or non-gestational choriocarcinoma.1 The estimated incidence of ovarian gestational choriocarcinoma is 1: 369,000,000 pregnancies, while non-gestational choriocarcinomas correspond to less than 0.6% of ovarian germ cell tumors, they comprise 2.1% of all germ cell malignancies ovarian, which makes this neoplasm extremely rare;2–4 in pubmed from 1976 to 2021 there are 54 reports.
Furthermore, both gestational and non-gestational diseases exhibit identical clinical manifestations and histology. but difficult cases often require DNA analysis, clinical diagnostic criteria are unreliable, except in patients who are unable to conceive or who have never had sexual intercourse.5–9 To differentiate them, it is necessary to determine whether there is a paternal contribution to the tumor genome. The tumor genetic polymorphism and comparison with those found in the patient and partner should define the presence or absence of paternal DNA and establish whether a tumor is gestational or not; only five cases have been reported of non-gestational pure ovarian choriocarcinoma diagnosed with polymorphic DNA analysis; Gestational choriocarcinoma shows broader variations in karyotype, the majority being aneuploid, and some in the hyperdiploid and hypotetraploid range. Many forms of chromosomal gains, losses, and rearrangements are seen, but no specific chromosomal abnormality has yet been identified.10
A medical history of pregnancy, amenorrhea, or gestational trophoblastic disease can help determine the diagnosis. These types of tumors should be considered distinct entities with different therapeutic approaches, chemotherapy regimens, and prognosis associated with each disease; diagnostic criteria for non-gestational ovarian choriocarcinoma were performed in 1963; including absence of disease in the uterine cavity, pathological confirmation of the disease, and exclusion of molar pregnancy and intrauterine pregnancy;11,12 in this case all criteria were met. It is highly aggressive; It can develop as a metastatic gestational choriocarcinoma from a choriocarcinoma of the uterus or uterine tubes, or as a primary gestational choriocarcinoma associated with ovarian pregnancy, or as a non-germ cell tumor of germ cells that differentiates towards trophoblastic structures; Gestational choriocarcinoma is associated with the spectrum of gestational trophoblastic disease. Non-gestational ovarian choriocarcinoma is associated with germ cell transformation. Both types of choriocarcinoma tend to develop early hematogenous metastases at different sites including the lung, liver, brain, bone, vagina, and other viscera.4,13,14
Compared with gestational choriocarcinomas, non-gestational choriocarcinomas are relatively chemoresistant and are associated with a worse prognosis, especially in advanced stage, early diagnosis and timely initiation of therapy is important, early diagnosis presents unique challenges in women in reproductive age.15
A 20-year-old married woman, with no history of pregnancy, who was admitted to the hospital for colicky abdominal pain in the right iliac fossa with radiation to the right iliac fossa from moderate to severe with Visual Analogue Scale with nausea and vomiting treated with analgesics presenting slight improvement; It is requested, quantification of the beta fraction of gonadotropin with figures of 112,337mIU/mL and pelvic ultrasound with a report of a complex cyst of the right ovary without evidence of free fluid in the cul-de-sac; in relation to probable dysgerminoma or tumor of etiology to be determined; the hematic biometry with hemoglobin of 15.8 and hematocrit of 45, platelets of 313,000, leukocytes of 11,300 and 2 bands; reason for which she is referred to Hospital Juárez de México and once hospitalized; on August 18, 2017; she presents low-cost symptoms; and the studies are repeated quantification of beta fraction of human chorionic gonadotropin (hGC) greater than 5,000, hemoglobin of 18 grams, hematocrit of 47%, leukocytes of 12,000, platelets of 367,000, lactic dehydrogenase of 473, direct bilirubin of 0.8 and the 2.3 total; normal liver transaminases; computed tomography and Ultrasound reports uterus in anteversion of 79x26x31mm, homogeneous myometrium, endometrial thickness of 7.9mm and right annex with lesion of 114 x83x79mm and total volume of 394cc, heterogeneous with linear images in its interior punctiform, the ultrasound diagnosis was, probable right annex teratoma tumor cystic (Photos 1–6).
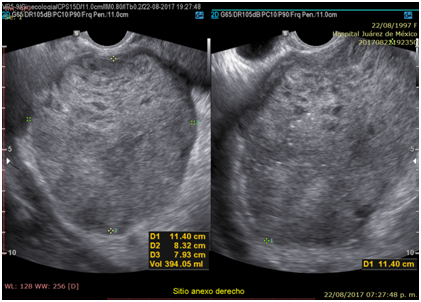
Photo 1 Transvaginal ultrasound. Right Annex. Heterogeneous echogenic tumor due to the presence of scattered irregular small hypoechoic images surrounded by echogenic tissue, as well as echogenic punctate images Dimensions of 114 x 73 x 89 mm.
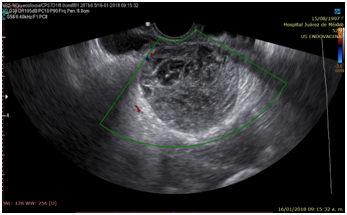
Photo 2 Transvaginal ultrasound. Right adnexal tumor, lesion without vascular flow in the Doppler modality.
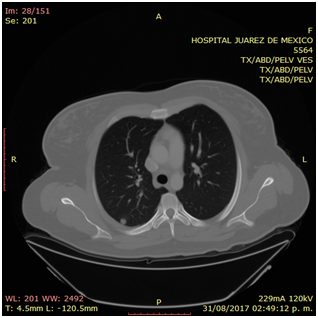
Photo 3 Thoracoabdominal computed tomography. Posterior basal pulmonary nodule on the right side of 11 mm in diameter with a pleural base.
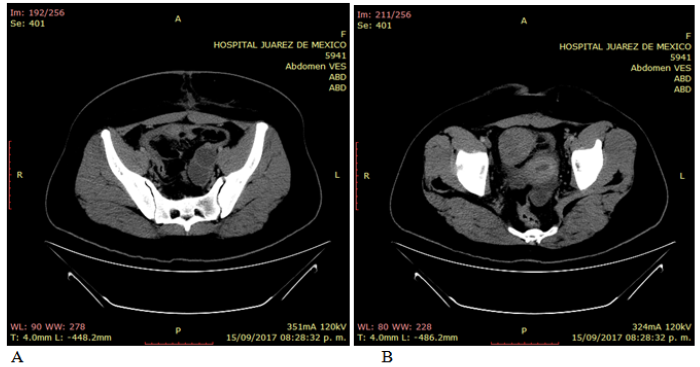
Photo 4 Abdominal computed tomography. Free intra-abdominal fluid (A). Left annex with the presence of two well-defined hypodense lesions with cystic behavior. Solid tumor with well-defined and delimited edges, located forward and to the right of the uterus, measuring 57 x 49 mm, which presents moderate enhancement (B).
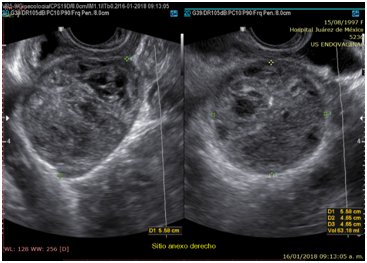
Photo 5 Transvaginal ultrasound. Mixed solid tumor with almost anechoic, small scattered areas surrounded by echogenic tissue. Dimensions 55 x 46 x 46 mm.
On physical examination, the patient with generalized pallor of the skin and integuments, hypotensive, tachycardia, diaphoretic, without cardiorespiratory compromise, globose abdomen due to abundant adipose panniculus, perstalsis present, pain on palpation in the right iliac fossa on bimanual touch, the uterus is delimited 8 x 5 cm, right annex, 12 x 9 cm adnexal tumor, irregular, firm, pain on cervical mobilization, with spotting-shaped transvaginal hemorrhage, extremities without edema, normal tendon reflexes. According to the clinical presentation, imaging studies, on suspicion of ruling out ectopic pregnancy, an exploratory laparotomy was performed, with findings of the right annex of 20 x 15 cm adhered to the uterus and sigmoid rectum without evidence of hemoperitoneum and a right salpingo-oophorectomy was performed for probable right ectopic pregnancy with hematic loss due to the surgical procedure of 40cc and sent to pathology for definitive histopathological study, during follow-up a control gonadotropin quantification was requested with the last report of 1 103 918.7mUI/mL. The histopathological results of the macroscopic surgical specimen were right salpingo-oophorectomy, with choriocarcinoma with extensive necrosis, 1 cm tumor extension to the ovarian surface not identified, lymphovascular invasion present and free surgical margins of the residual ovarian tumor with cystic follicles, Figure 1 and the report microscopic, Figures 2–4. A new hCG quantification is performed, negative head tomography and chest tomography with probable metastasis due to pulmonary nodule, in abdomen and pelvis, presence of peritoneal carcinomatosis, with free fluid and lumbar puncture reports negative for malignant cells and chemotherapy based on cisplatin and etoposide 5 sessions and the patient is disease free after two years of follow-up.
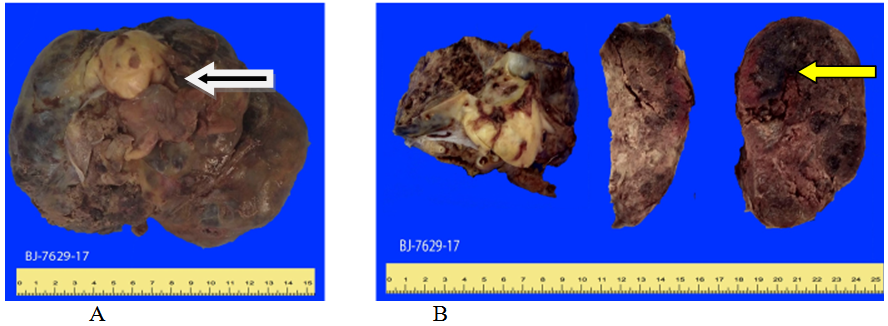
Figure 1 Histopathological report (8BJ-7629-19) Choriocarcinoma; A) tumor that forms a large solid gray hemorrhagic mass (white arrow) involving the ovary and uterine tube; B) When the ovarian tumor is sectioned, a solid mass with extensive areas of necrosis and hemorrhage (yellow arrow) is observed, the ovarian surface when sectioned with hemorrhagic cysts of different sizes.
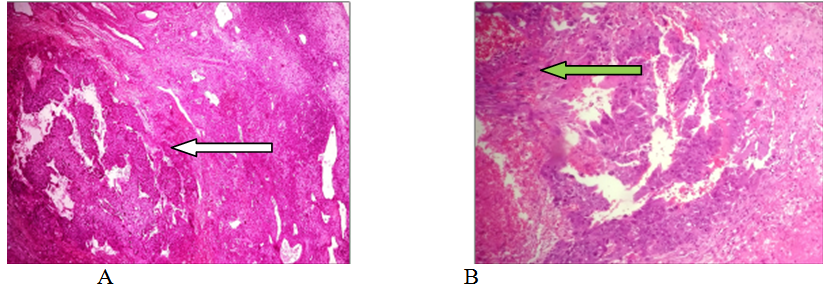
Figure 2 (BJ-7629-17). Choriocarcinoma: A) Panoramic view of ovary with extensive areas of hemorrhage, necrosis and nests of neoplastic germ cells (E&E, original x40) (white arrow); B) A germinal neoplasm formed by mononulcerated trophoblast and syncytiotrophoblast cells is observed in the periphery of the blood channels (H&E, original x100) (green arrow).
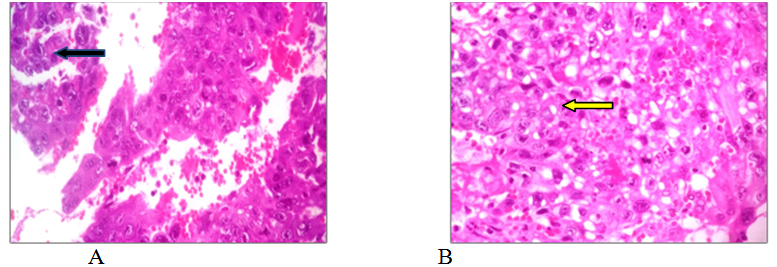
Figure 3 (BJ-7629-17). Choriocarcinoma: A) Panoramic view of ovary with extensive areas of hemorrhage, necrosis, and nests of neoplastic germ cells (E&E, original x40) (black arrow); B) A germinal neoplasm formed by mononulcerated trophoblast and syncytiotrophoblast cells is observed in the periphery of the blood channels (H&E, original x100) (yellow arrow).
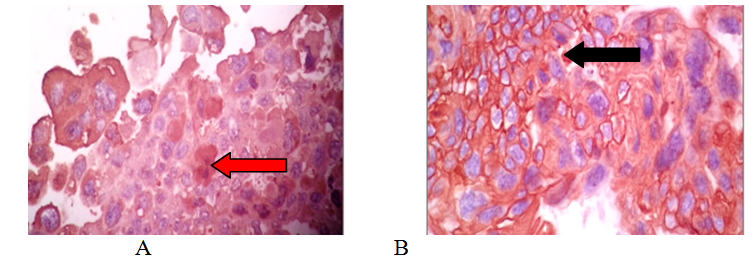
Figure 4 BJ-7629-17. Choriocarcinoma; A) syncytiotrophoblast, composed of large basophilic vascuolate cells with irregular borders; These cells contain multiple hyperchromatic nuclei, which vary in shape and size (H&E, original x400) (red arrow); B) mononucleated trophoblast, composed of cytoplasm and intermediate trophoblast, cells are medium-sized, polygonal, round or oval with clear cytoplasm and sharp edges. Some cells have centralized, small, round and hyperchromatic nuclei, others have larger vesicular nuclei with nucleolus and mitotic activity (H&E, original x400) (black arrow).
Non-gestational ovarian choriocarcinoma, which is not associated with pregnancy, is a rare germ cell tumor of high-grade malignancy that occurs more frequently in pre-adolescent women;11,16 Its clinical manifestations of NGCO include vaginal bleeding, abdominal pain, and pelvic masses, most of which are misdiagnosed and initially treated for tumor torsion, ovarian tumor, or ovarian cancer,4,13,14 as the case presented. The rarity of primary ovarian choriocarcinoma makes it a diagnostic challenge and generally an incidental finding in the surgical specimen of an ovarian tumor, the low incidence of these cases, a differential diagnosis should be made in women of reproductive age who present with an acute abdomen and suspected complications of an ectopic pregnancy;15 for the symptoms of pelvic pain, positive pregnancy test, adnexal tumor and anemia; as the case presented; the majority of non-ovarian choriocarcinomas are histopathologically diagnosed after surgery (4); the best treatment for non-gestational ovarian choriocarcinoma is surgery with adjuvant chemotherapy; however, they often have a poor prognosis;10,13,14 a histopathological study is essential. The serum quantification of the beta-hCG fraction is decisive because it is not only the basis for diagnosis but also for prognosis;1–4 The clinical case that we present was initially seen and referred to our hospital; with imaging studies and quantification of hGC; Due to the acute abdomen symptoms that he presented and the ultrasound report, emergency surgery was indicated; It is reported that the complications of tumors, causing an acute abdomen, which requires emergency surgery in 40 to 50% of cases, such as this case.
Choriocarcinomas, due to their great angioinvasion capacity, which facilitates their dissemination and encourages metastases to the lung, brain, liver and other organs, have early metastasis to the lung even in 80% of cases, in our case, pulmonary metastasis was diagnosed by imaging, confirming the great tumor vascular invasion; the majority die, even when diagnosed early to initiate appropriate chemotherapy, with a mortality rate of 10% to 15%.17,18 This report describes a case that simulated an ectopic pregnancy, which underwent surgery of the administration of chemotherapy; We report that the reproductive prognosis is favorable and treatment with conservative surgery does not affect fertility. Ovarian germ cell malignancies are sometimes associated with pregnancy or term pregnancies are achieved after uncomplicated treatment with healthy term infants.19
Gestational choriocarcinoma shows wider variations in karyotype, the majority being aneuploid, and some in the hyperdiploid and hypotetraploid range. Many forms of chromosomal gains, losses, and rearrangements are seen, but no specific chromosomal abnormality has yet been identified. Pure non-gestational choriocarcinoma of the ovary is so rare that it is not known whether the prognosis is worse than gestational choriocarcinoma or not. Some articles reported that non-gestational choriocarcinoma of the ovary has a worse prognosis and is less sensitive to methotrexate-based chemotherapy regimens than gestational neoplasia. But they were not definitively diagnosed by DNA polymorphism analysis. It is important to clarify whether the tumor arose from a gestational or non-gestational origin to accurately understand the prognosis of this disease; nongestational ovarian choriocarcinoma has a less favorable prognosis than gestational type.20–24
There are few case reports in our country and no case had been reported in our hospital; For this reason, we consider it important to report the form of presentation for its timely diagnosis, to improve the prognosis and survival of the patient. According to previous reports, it seems that pure ovarian choriocarcinoma responds well to the combination of surgical ablation and postoperative chemotherapy. To date, a definitive treatment modality for pure ovarian choriocarcinomas has not been established due to the low incidence. Therefore, pure ovarian choriocarcinomas are generally treated with the same protocols that are used for ovarian germ cell tumors and gestational trophoblastic disease. In the current case, treatment included debulking surgery followed by adjuvant chemotherapy.
Non-gestational choriocarcinoma is an extremely rare malignant neoplasm that can present clinically as an acute abdomen and requires differential diagnoses. Management is the combination of surgery and adjuvant chemotherapy, our patient responded well; for women with a desire to preserve fertility it is a viable option.
None.
There was no funding for the preparation of this work.
The authors have no conflicts of interest.

©2021 Manuel, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.