eISSN: 2377-4304


Mini Review Volume 13 Issue 1
1FMH specialist in radiology, EHC Hospital Morges, Switzerland
2Resident in radiology, EHC Hospital Morges, Switzerland
Correspondence: Dr Quoc Duy Vo, FMH specialist in radiology, EHC Hospital Morges, Chemin du Cret 2, Switzerland
Received: January 04, 2022 | Published: January 14, 2022
Citation: Molteni T, Patrique POS, Niasme E, et al. Incidental breast lesion on chest CT scan: a review. Obstet Gynecol Int J. 2022;13(1):15‒17. DOI: 10.15406/ogij.2022.13.00619
Most breast cancers are diagnosed by conventional breast imaging which includes mammography, ultrasound and MRI. However, CT is an often overlooked modality in the detection and diagnosis of breast cancer. Incidental findings of breast cancer by CT are not so rare. This article aims to aware radiologists and gynaecologists to the potential of breast cancer detection with CT. In this brief review of the literature, we will discuss the morphological characteristics of breast tumours on CT and the imaging mangement of suspicious lesions discovered on CT.
Breast cancer is the most frequent cancer amongst women and it represents a global public health problem, with 1.7 million new cases diagnosed every year1 and a worldwide annual prediction of 3,2 million new cases per year by 2050.2 It is also the leading cause of cancer death in women aged between 20 and 59 years, with approximatively 0.5 million deaths per year in 2013.1 Today, breast cancer is a well-known disease with readily recognized risk factors, which include family history, genetic abnormalities, alcohol consumption, nutrition, female’s hormonal context, obesity and thoracic radiotherapy.3
Breast cancer diagnosis relies on clinical examination and radiological investigations, which include a plethora of medical imaging techniques, most commonly mammography. Ultrasound and magnetic resonance imaging (MRI) have been frequently employed as complementary tools to mammography, and have repeatedly proved their efficiency.4 Currently, CT scan has become an exceedingly common imaging modality, used in nearly all areas of medicine. The steady increase in utilization of this technique in the past few decades has led to an increased detection rate of “incidental” breast lesions, which may be of major health concern.5–8 Therefore, radiologists must be familiar with breast findings on CT scans and must be able to propose an adequate workup.
Mammography is the most common imaging modality used to assess breast disease and it is capable of detecting subtle abnormalities such as microcalcifications, architectural distortion and opacities with spiculated margins with a specificity of 55% and sensibility of 70%.9 Recently, computer–aided detection (CAD) has been developed to help radiologists in identifying breast anomalies. It employs a specific computer algorithm capable of individualizing and marking suspicious mammogram findings, assisting the radiologist during the interpretation of the exam.10,11 However, there are some limitations to mammograms, especially breast density that can provoke superimpositions, which are able to hide pathological lesions and avoid detection, even with the use of CAD.4,10 For these reasons, digital breast tomosynthesis has been extensively used in conjunction with mammography. It acquires multiple low dose mammographic projections of the breasts which are organized in stacks of slices after tomographic reconstruction, similar to CT, reducing tissue superimposition and improving detection of masses or architectural distortion (Figure 1).4,12
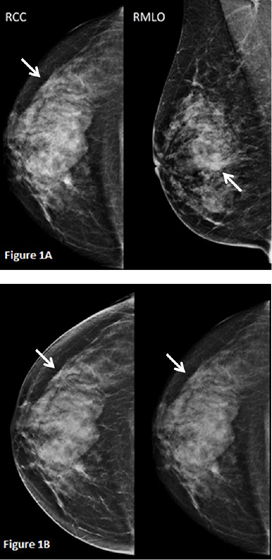
Figure 1 A. Mammography of the right breast revealed an irregular and spiculated opacity in the outer upper quadrant (white arrow). B. Tomosynthesis performed on the cranio-caudal view (left image) improves characterization of the lesion compared to standard mammography (right image), with better visualization of the spiculated aspect of the lesion.
Breast ultrasound is usually used conjointly with mammography, particularly in patients with dense breasts. It was first used to differentiate solid from cystic lesions during the 70’s.4,10 Nowadays, with technological advancement, modern ultrasound devices are able to characterize breast lesions (Figure 2) and to improve diagnosis of breast cancer with a negative predictive value of 99.5% and a sensitivity of 98.4%,13 thus avoiding unnecessary biopsies. In spite of its technical improvements, ultrasound still bears some limitations: first of all, it is an operator-dependent modality, therefore physicians’s experience may play a crucial role in disease detection; secondly, it is unable to detect microcalcifications, one of the early hallmarks of ductal carcinoma in situ (DCIS), which is believed to herald infiltrating ductal carcinoma.4,10,13
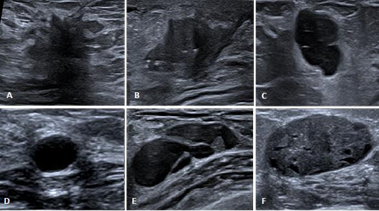
Figure 2 A. Hypoechoic lesion with spiculated borders associated with a hyperechoic halo and an acoustic shadow. B. Hypoechoic lesion with irregular and angulated borders. C. Well delimited hypoechoic lesion presenting a “taller than wide” shape. D. Simple cyst presenting as a well delimited anechoic lesion with posterior enhancement. E. Complex cyst with septations and mural nodule. F. Well delimited hypoechoic lesion containing small cysts with ovoid shape.
During the past two decades, breast MRI has emerged as one of the most valuable imaging techniques in the diagnosis of breast cancer. It is considered the most sensitive breast imaging modality, providing a sensitivity of 90% for breast cancer detection with variable specificity ranging from 37% to 100%.14–16 Its use allows detection of multifocal cancer, recognition of invasive components in DCIS, detection of cancer in excessively dense breasts, evaluation of tumor response in neoadjuvant chemotherapy17 and screening for high risk women including those with BRCA mutations.4 Its main disadvantages lie on its availability and on its lengthy scan times which can be difficult to manage, especially for claustrophobic patients.
Studies and frequency
As mentioned earlier, a dramatic rise in the use of CT scan, normally performed for indications other than breast disease, has led to an increased detection of incidental breast abnormalities. Consequently, a considerable number of studies have been conceived to evaluate the incidence and possible outcomes of incidental breast lesions detected on unenhanced or contrast-enhanced CT, with variable results depending on the use of contrast media. In a large study, Shojaku et al. performed unenhanced chest CT scans on 1’008 female patients to investigate the frequency of breast tumors. Breast anomalies were detected in 6 patients, in which 3 were diagnosed with breast cancer (0,3%).8 In another large-scale study, Swensen et al. assessed the performance of low-dose chest CT to evaluate the presence of incidental pulmonary nodules in 1’520 individuals aged 50 years or older, who had smoked 20 pack-years or more. In 3 female patients, a diagnosis of breast cancer was found (0,1%).18 Lin et al. reported 23 female patients with incidental breast findings in a series of 2’250 contrast-enhanced chest CT scan. Among these 23 patients, 16 had breast cancer which was confirmed by further investigations (0,7%),5 suggesting that the use of contrast media may improve breast cancer detection in CT Scan.19
Some authors advocate that contrast-enhanced CT improves detection and characterization of breast lesions. Perrone et al. propose its use as an alternative to MRI in patients with usual contraindications such as incompatible pacemakers or surgical clips, severe dyspnea and claustrophobia. They recommend to acquire CT images at 1 minute after contrast administration and to apply a cutoff attenuation value of 90 Hounsfield units that would allow to differentiate between benign and malignant lesions (Figure 3).20 Inoue an al. demonstrate that the use of dynamic contrast-enhanced CT (DCE-CT) like DCE-MRI, can be useful to identify and to characterize breasts lesions. In their protocol, acquisitions were obtained at 1, 3, and 8 minutes after intravenous contrast injection. On time-density curve analysis, the washout pattern had a positive predictive value for breast malignancy of 93%, with a sensitivity of 91%.21 Morphological criteria can also be implemented in order to distinguish malignant from benign lesions. Spiculated or hazy margins, irregular shape and rim enhancement can all be looked upon as reliable features of malignancy.6,21 Moreover, axillary lymphadenopathies, pectoralis muscle infiltration and pleural effusion strongly suggest tumoral invasion.6 In summary, contrast-enhanced CT scan seems to be a reliable alternative to MRI, with the advantages of being faster, particularly for dynamic studies, as well as less susceptible to movement artifacts.20 However, due to radiation exposure concerns, chest CT scan must be reserved for elderly patients.22
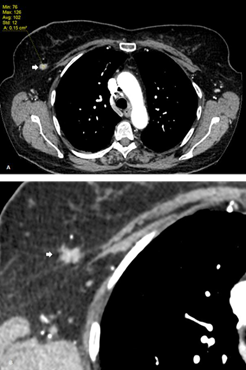
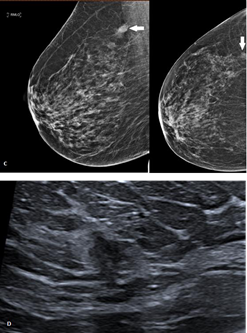
Figure 3 A. Follow-up contrast-enhanced chest CT in a patient with lung carcinoma shows an incidental right breast lesion with contrast enhancement up to 102 HU. B. Same image with smaller FOV (field-of-view) evidencing the spiculated and irregular borders. C. Digital mammography of the same patient showing an irregular lesion with blurred borders. D. Ultrasound imaging reveals a hypoechoic lesion with angulated borders associated with a hyperechoic halo.
Nowadays, chest CT scan is widely performed for a variety of indications other than breast disease, though its relentless increasing use over the years has resulted in an ever-growing detection of incidental breast findings. Therefore, radiologists must be able to recognize and to assess these findings. Breast tumors morphological criterias on CT are the same as in MRI and can therefore be reliably used. However, any incidental breast lesion on CT requires conventional breast imaging assessement including mammography and echography, breast MRI can be performed for dense breasts or in case of multicentric lesions.
None.
None.
The author declares that they have no competing interests.

©2022 Molteni, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.