Group theoretical method
Tin oxide (SnO2) has space group number 136. First of all, atomic structure and its topview of SnO2 are presented in Figure 1(a) and (b), respectively.
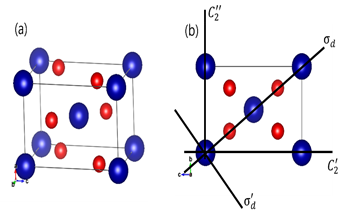
Figure 1 (a) atomic structure and (b) topview of SnO2. Blue and red spheres are represent Sn and O atoms, respectively. (These figures are drawn by visualization program, VESTA).
If we choose one Sn atom which is placed at the corner as the original position, (0,0,0), the symmetry operations with τ =(0.5,0.5,0.5) are given as:
(1)
In the international tables for X-ray crystallography, the sites of Sn and O location are 2a and 4
, respectively.
We find the equivalence transformation for SnO2 at the center of the Brillouin zone (Table 1).
|
|
|
|
,
|
,
|
,
|
|
|
,
|
|
|
|
|
|
|
|
|
|
|
|
χ (Sn)
|
2
|
2
|
0
|
0
|
2
|
2
|
2
|
0
|
|
χ (O)
|
4
|
0
|
0
|
0
|
2
|
0
|
4
|
0
|
|
χ (total)
|
6
|
2
|
0
|
0
|
4
|
2
|
6
|
0
|
Table 1
for SnO2
Also we find the lattice modes at the zone center
, including their symmetries, degeneracies and the normal mode patterns.
At
, the wave vector group contains the full symmetry operations of the space group. The character table of the group of the wave vector would be the same as that of
because of the phase factor
. By using the charater table of
, we have χ(Sn)= A1g+B 2g, χ(O)=
, and =
.
Therefore, the lattice vibration normal modes are given as equation (2).
(2)
Since all the representations of D 4h are one dimensional except for E μ and E g, they have single modes without degeneracy while E μ and E g modes are doubly degenerated. A 2μ moves along z direction. Both Sn and O atoms are out-of-phase.
B 1μ also moves along z direction. Two Sn atoms which is placed different sites point opposite directions. Similar with Sn, two SnO atoms which is placed different sites point opposite directions. There are two kinds of E g. And both move along z-direction. Movements of Eμ is similar with Eg . But they translate into x and y directions.
For a next step we indicate the IR-activity and Raman activity of these modes. IR-active modes includes A2μ and 3Eμ while Raman acitvity modes represent A 1g, B1g , B 2g and Eg .
#1 The A2μ mode is active to z-polarized light while 3Eμ modes are active to x or y polarized light.
#2 A1g and B1g have diagonal matrix elements but B2g and Eg are off-diagonal.
When we move away from k = 0 the mode splitting along the (100) and (001) directions, the group of the wave vector contains
, and
. Two dimensional k-space is presented in (Figure 2).
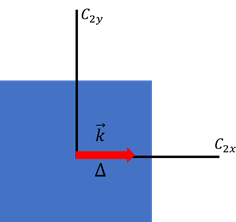
Figure 2 2-dimensional K-space. Direction of the mode splitting is denoted as an red arrow. (I remake this figure which is in dresselhaus lecture note).
The individual characters are given in Table 2, where the phase factor
is taken out.
|
|
|
|
|
|
|
Δ1
|
1
|
1
|
1
|
1
|
|
Δ2
|
1
|
1
|
-1
|
-1
|
|
Δ3
|
1
|
-1
|
1
|
-1
|
|
Δ4
|
1
|
-1
|
-1
|
1
|
Table 2 Individual character for SnO2
Now, we use the decomposition rule to see how the representations at Γ split in to Δ1, Δ2, Δ3, and Δ4 (Table 3).
|
|
ϵ
|
iC2z
|
iC2x
|
iC2y
|
|
|
A1g
|
1
|
1
|
1
|
1
|
Δ1
|
|
A1μ
|
1
|
-1
|
1
|
-1
|
Δ3
|
|
A2g
|
1
|
1
|
-1
|
-1
|
Δ2
|
|
A2μ
|
1
|
-1
|
-1
|
1
|
Δ4
|
|
B1g
|
1
|
1
|
1
|
1
|
Δ1
|
|
B1μ
|
1
|
-1
|
1
|
-1
|
Δ3
|
|
B2g
|
1
|
1
|
-1
|
-1
|
Δ2
|
|
B2μ
|
1
|
-1
|
-1
|
1
|
Δ4
|
|
Eg
|
2
|
-2
|
0
|
0
|
Δ3+ Δ4
|
|
Eμ
|
2
|
2
|
0
|
0
|
Δ1+ Δ2
|
Table 3 Splitting behaviors in Δ1, Δ2, Δ3, and Δ4 at Γ.
Now, along (001) direction, the group of the wave vector contains
, and
.
Since the point symmetry operations form C point group, the character can be given as Table 4, where the phase factor
is taken out (as same as (100) case).
|
|
ϵ
|
|
|
|
|
|
Σ1
|
1
|
1
|
1
|
1
|
1
|
|
Σ2
|
1
|
1
|
1
|
-1
|
-1
|
|
Σ3
|
1
|
1
|
-1
|
1
|
-1
|
|
Σ4
|
1
|
1
|
-1
|
-1
|
1
|
|
Σ5
|
2
|
-2
|
0
|
0
|
0
|
Table 4 Characters in prespective of Σn
Table 5 shows the decomposition of each mode.
|
|
ϵ
|
|
|
|
|
|
|
A1g
|
1
|
1
|
1
|
1
|
1
|
Σ1
|
|
A1μ
|
1
|
1
|
1
|
-1
|
-1
|
Σ2
|
|
A2g
|
1
|
1
|
1
|
-1
|
-1
|
Σ2
|
|
A2μ
|
1
|
1
|
1
|
1
|
1
|
Σ1
|
|
B1g
|
1
|
1
|
-1
|
1
|
-1
|
Σ3
|
|
B1μ
|
1
|
1
|
-1
|
-1
|
1
|
Σ4
|
|
B2g
|
1
|
1
|
-1
|
-1
|
1
|
Σ4
|
|
B2μ
|
1
|
1
|
-1
|
1
|
-1
|
Σ3
|
|
Eg
|
2
|
-2
|
0
|
0
|
0
|
Σ5
|
|
Eμ
|
2
|
-2
|
0
|
0
|
0
|
Σ5
|
Table 5 Decomposition of each mode of Σn
The splitting mode is presented in Figure 3.

Figure 3 Sketchy of the mode splitting behaviors.
Energy dispersion
Using the empty lattice, we find the energy eigenvalues, degeneracies and symmetry types for the two electronic levels of lowest energy for the fcc lattice at the Γ point (
= 0). Note that the lowest energy state is a non-degenerate state with
symmetry.
Reciprocal lattice of fcc → bcc lattice
The nearest neighbor point in reciprocal lattice:
(3)
At Γ point, the energy eigenvalues are given by
, where
is the reciprocal lattice vector. Therefore, the lowest energy eigenvalue = 0. Also, second lowest energy eigenvalue can be obtained as followed equation.
(4)
Since there are 8 equivalent {1 1 1} points, the degeneracy of the second lowest level is 8. The group of the wave vector at Γ point is
. The characters for the equivalent transform are shown in Table 6.
|
|
E
|
|
|
|
|
|
|
|
|
|
|
|
1
|
1
|
1
|
1
|
1
|
1
|
1
|
1
|
1
|
1
|
|
|
8
|
0
|
0
|
2
|
0
|
0
|
0
|
4
|
0
|
0
|
Table 6 The characters for the equivalent transform of Σn
Therefore, lowest energy and second lowest levels are
(5)
(6)
We also find the appropriate linear combination of plane waves which provide basis functions for the two lowest L-point electronic states for the fcc lattice.
At L(
) point, the energy levels are given by E=
(k1 + k). The lowest energy level corresponds to
= (0,0,0) and
= (-1,-1,-1) with E0 =
The plane waves are
for (111) and
for (-1-1-1).
The second energy level consists of k=
(-1-11),
(1-1-1),
(-11-1),
(00-2),
(0-20),
(-200) with E1 =
.
The corresponding plane waves are:
(7)
We denote these plane wave states by (-1-13), (11-3), (1-31), (3-1-1), (-311).
At L point, the group of the wave vector is D3d , the character is shown in Table 7.
|
|
E
|
2C3
|
3C2
|
i
|
2iC3
|
3iC2
|
|
L1
|
1
|
1
|
1
|
1
|
1
|
1
|
|
L2
|
1
|
1
|
-1
|
1
|
1
|
-1
|
|
L3
|
2
|
-1
|
0
|
2
|
-1
|
0
|
|
L 1 '
|
1
|
1
|
1
|
-1
|
-1
|
-1
|
|
L2 '
|
1
|
1
|
-1
|
-1
|
-1
|
1
|
|
L3 '
|
2
|
-1
|
0
|
-2
|
1
|
0
|
Table 7 The group of the wave vector is D3d at L point
The equivalence transform of the plane waves are shown in Table 8.
|
|
E
|
2C3
|
3C’2
|
i
|
2iC3
|
3iC’2
|
|
|
(111), (-1-1-1)
|
2
|
2
|
0
|
0
|
0
|
2
|
|
|
(-311), etc
|
6
|
0
|
0
|
0
|
0
|
2
|
|
Table 8 The equivalence transform of the plane waves
The symmetry of the lowest state at L point is L
+ L
. The basis functions of the two symmetry states are:
(8)
The symmetry of the second lowest state is
. The basis functions are obtained as follows:
(9)
We had have question that "which states of the lower and upper energy levels in (a) and (b) are coupled by optical dipole transitions?".
At Γ points,
=
. The lowest energy state has symmetry
.
(10)
Therefore, only the
state in the second energy level will couple with
state in the lowest energy level. At L point,
. The lowest level has symmetry L1 +L’2 . For L1 state, L1
(L’2 +L’3 )= L’2 +L’3. Therefore, the L1 state in the lower level will couple with L’2 and L’3 states in the upper level via optical transition.
For L2 state, L’2
(L’2 +L’3 )= L1 +L3 . Therefore, the L’2 state in the lower level is coupled with L1 and L3 states (Figure 4).
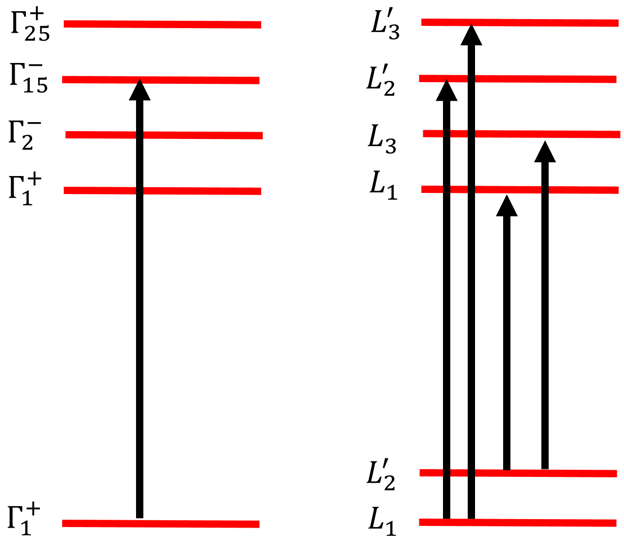
Figure 4 Sketchy of the transition behaviors.
Using compatibility relations, we find the symmetries of the energy levels that connect the two -point and two L-point energy levels. For = (
), the group of wavevector is C3v {E,2C3, 3
}.
Therefore equations are presented as followed.
(11)
Perturbation theory
Using Ek perturbation theory and we find the form of the kp relations near the L-point in the Brillouin zone for a face centered cubic lattice arising from the lowest levels with L1 and L’2 symmetry that are doubly degenerate in the free electron model.
Let’s assume |i> has L1 symmetry and |j> has L2 has L’ symmetry. Since
transform as a vector,
=L’2 +L’3 at L point.
L1
L’2 = L’2 . So it contains L’2 .
L2
L’32 = L’3 . So it does not contains L’2 .
Now, let’s take the new
,
, and
coordinates where
is parallel to (111) in the original coordinate. In this new coordinate,
transforms like
whereas
, and
transform like
.
Therefore, the only non-vanishing matrix element is
(12)
Then, equation would becomes
(13)
Therefore,
(14)
Using the Slater Koster technique, we find the form for E(k) for the lowest two levels for a face centered cubic lattice.
For fcc lattice, d=0 is the zeroth neighbor at a(0,0,0), d=1 is the nearest neighbor at a(1/2,1/2,0) and d=2 are the second nearest neighbor at a(1,0,0).
(15)
We expand your our results for the L-point in a Taylor expansion. At L point,
Let
then
(16)
Therefore
(17)
Now, let’s use
,
,
coordinate where
is parallel to (111) direction.
Then we obtain followed equation.
(18)
Now, let’s Taylor expand the result.
(19)
This result suggests that carriers at L point have anisotropic effective mass tensor.
Application of perturbation theory
Using perturbation theory, we find the form of the secular equation for the valence band of Si with
symmetry. For the valence band of Si with
symmetry, we use the degenerate
perturbation theory. Due to the parity requirement (
=0 since H’ has the odd parity), we have to to the second-order perturbation.
Since H’ transforms like
and
=
+
+
+
,
is coupled with only the following intermediated states;
(20)
For
where
, the secular equation becomes followed equation.
(21)
If we suppose that our silicon sample is a thin film with 10 nm thick grown pseudomorphically on a germanium substrate, what happens to E(k) for the silicon valence band in the thin film if the germanium substrate is oriented along a (100) direction or if it is oriented along a (110) direction? Since there is a lattice mismatch between Si and Ge, thin silicon film has tensile stress when it grows on Ge substrate. Therefore, the symmetry of the crystal is reduced as follows:
(22)




