MOJ
eISSN: 2374-6920


The word plastic is adopted from the Greek terminology referring to “capable of being shaped or molded”. These are largely synthetic petrochemicals, but are sometimes found naturally. Today, plastics have become an inevitable part of the human life. Plastics are one of the major causes of environmental pollution. Not only that, it is noted that the chemical Bisphenol A (BPA) present in the plastics trigger the formation of breast cancer. The present article elucidates the environmental factors causing breast cancer and the steps taken to prevent the growth of such incidences.
Keywords: plastics, breast cancer, bisphenol a, BPA, environmental pollution
Plastic pollution is one of the major threats of the world these days. Plastic pollution refers to the accumulation of plastic and its products causing severe adverse environmental issues not only to humans but also to the animals and plants.1 These are one of the major compositions of land and water pollutions.2 Though; known for these adverse effects plastic has become an integral part of human life. It was noted earlier that the industrial effluents hamper the normal physiological responses3,4 mimicking steroids, retinoid, thyroid hormones and lipophilic hormones.5–9
Bisphenol A (BPA) is used in the preparation of polycarbonate plastics seen widely in the consumer products,10 which was firstly synthesized in 189111 and is “BPA is usually known as Estrogenic agent”,12 both in vivo and in vitro10 and binds to the same receptor as the female hormone does. Vanden Berg et al.,13 in 2009 stated that humans come in contact with BPA primly when they consume food and water from the materials used for containers and packages. This endocrine disruptor thus, enters the body by ‘Leaching’ results when the polymer breaks to release the BPA monomers.14,15
BPA binds and activates estrogen receptors ER α and ER β, but with a much lesser affinity when compared to estradiols16 Figure 1.
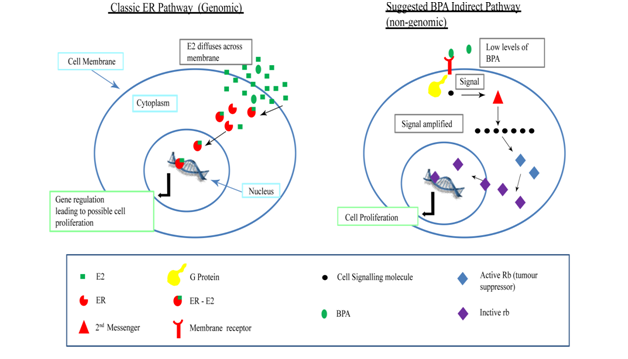
Physiologically BPA can alter the ovarian cycle and interferes with the embryonic development and exerts its role in many physiological and biological changes in women,10,17–20 besides causing a variety of cancers21,22 including breast cancer.
Chemically BPA (CH3)2C(C6H4OH)2, Figure 2, is a colorless compound belongs to diphenyl methane derivatives and bisphenol with two hydroxyl phenyl groups. It is soluble in organic solvents.
The objective of the present article is to assess and understand the ADMET / toxicological properties of Bisphenol A and its metabolite, 4-methyl-2, 4- bis (4-hydroxyphenyl) pent-1-ene [MBP] after they enter the human body.
Software used
Discovery studio Accerly 2.5, ADMET predictors.
Selection of the molecules
The molecules for the present study were imported from Pubchem with the IDs 6623 and 83494 for Bisphenol A and MBP respectively.
Upon studying the ADMET descriptors which gives an idea of the absorption, distribution, metabolism, and excretion-toxicity in pharmacokinetics of a given compound. following are studied and understood for the compound, taking the standard parameters as constant; ADMET Absorption: which Predicts Human Intestinal Absorption (HIA) after it is orally administered, ADMET Aqueous Solubility: which predicts the solubility of each of the compounds in water at 25°C, ADMET Blood Brain Barrier: that helps in predicting the ratio of concentrations of compound on both sides of the blood brain membrane after oral administration, ADMET Plasma Protein Binding: this predicts whether or not a compound is likely to be highly bound to carrier proteins in the blood, ADMET CYP2D6 Binding: Predicts cytochrome P450 2D6 enzyme inhibition, ADMET Hepatotoxicity:23 Predicts dose-dependent human hepato toxicity of compounds. Table 1 shows the ADMET results of MBP and BPA and Figure 3 represents the ADMET plot and Figure 4 demonstrates the graphical, comparative results of ADMET.
|
Name |
ADMET_BBB |
ADMET_BBB_Level |
ADMET_Absorption |
ADMET_Solubility |
ADMET_Solubility_Level |
ADMET_Hepatotoxic |
1 |
83,494 |
0.654 |
1 |
0 |
-4.39 |
2 |
1 |
2 |
Bisphenol A |
0.355 |
1 |
0 |
-3.594 |
3 |
1 |
ADMET_Hepatotoxic |
ADMET_CYP2D6 |
ADMET_CYP2D6_Level |
ADMET_PPB_Level |
ADMET_AlogP98 |
ADMET_Unknown |
ADMET_PSA_2D |
1 |
0 |
0.346 |
2 |
4.746 |
0 |
41.631 |
1 |
0 |
0.247 |
1 |
3.778 |
0 |
41.631 |
Table 1 ADMET results
Blood brain barrier
The results states that the MBP has high penetration level when compared to that of Bisphenol A.
ADMET BBB penetration level
The obtained results states that the penetration levels of the compounds fall in the category of high penetration value with the level of 1.
ADMET absorption
The level obtained for both the compounds is 0 stating that they can be absorbed easily by the human intestinal system.
ADMET solubility/level
The compounds show a moderate to good level of solubility.
ADMET hepatotoxicity
The hepatotoxicity model predicts potential organ toxicity for a wide range of structurally diverse compounds.
The generated values states that the compounds are likely to cause toxicity to the liver with the score of 0.7.
ADMET CYP2D6
The generated values show that the compounds, non inhibitor and unlikely to inhibit CYP2D6 enzyme with the score of 0.346 and 0.247 respectively.
The present article elucidates on the toxic effects of plastic and its byproduct. In order to reduce its hazardous effects on humans, it is utmost essential to reduce its encounter. BPA and MBP have already proved to alter and induce certain notable changes in breast cancer and in the mammary glands. Their binding affinities with ER α and ER β were also studied and analyzed by Barker and Chandsawangbhuwana. In conclusion, I strongly feel that there should be more awareness among the people on this aspect. Further, it should be made known to the people to minimize eating canned food and polycarbonate plastic food containers and to choose fresh foods.
None.
The author declares no conflict of interest.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.