MOJ
eISSN: 2573-2919


Research Article Volume 7 Issue 3
1Regional University Center Atlantic Zone, National University of Comahue,Viedma, Argentina
2Agricultural Experimental Station Valle Inferior. Agreement INTA-Province of Rio Negro,Viedma, Argentina
Correspondence: Kröpfl A, Regional University Center Atlantic Zone, National University of Comahue.Viedma, Argentina, Tel +54 9 2920600921
Received: July 14, 2022 | Published: July 28, 2022
Citation: Kröpfl A, Cecchi G. Key sites for seedling emergence in degraded areas of the monte ecoregion of the Rio Negro Province, Argentina. MOJ Eco Environ Sci. 2022;7(3):89-94. DOI: 10.15406/mojes.2022.07.00252
In disturbed shrublands of the Monte ecoregion in the Rio Negro province, herbaceous vegetation occurs in patches of high plant cover beneath shrubs, interspersed with patches of low cover between them, which leads to the existence of differential microsites in the environment. The protected understory in the shrub patches is occupied by other functional groups that conform this system: perennial grasses, annuals and biological crusts. The areas between the shrubs may be more or less degraded by grazing, and may exhibit physical impediments to the germination and establishment of seeds and to seedling emergence and survival, in the form of vesicular crusts that develop on the soil surface and contribute to desertification in those systems where they appear. The objective of our study was to relate the presence of seedlings in three microsites of the environment: a) beneath shrubs, and in the open spaces between them discriminated into b) flat and c) rough places, with different covers of the soil surface (biological crust, litter, perennial grasses and gravel), and with the presence and thickness of vesicular crusts, in a semi-arid shrubland of the south-eastern Monte ecoregion. We found that the presence of seedlings differed between the microsites and was related to the different soil cover in each microsite. In flat microsites, that presence was positively related with litter and biological crust covers and negatively related to vesicular crust thickness and gravel cover; in rough places we only found a positive influence of litter, and beneath shrubs, there were beneficial effects due to perennial grass cover and scarce negative effects due to vesicular crusts.
Keywords: bare ground areas, microsites, vesicular crusts, biological crusts, litter, perennial grasses
In arid ecosystems, vegetation is often spatially distributed as a mosaic with dispersed "islands" in a matrix of bare ground,1,2 and this spatial patterning can be induced by both abiotic and biotic factors.3 The soil surrounding the plants can be modified through the accumulation around their canopies of water, nutrients, sediments, litter and seeds, derived from processes that take place in the bare ground between plants.4 Herbivore activity can also directly (through consumption) or indirectly (i.e., through trampling) affect bulk density and soil infiltration patterns.5 It can also affect the quantity and quality of litter produced,2 reinforcing the ‘fertile island’ phenomenon around individual plants.
The distribution of vegetation in islands dominated by shrubs (with “leopard” pattern1 in our region) would mainly be determined by a shortage of rainfall, since runoff from bare inter-shrub areas can increase the availability of water in the islands with higher infiltration. But that distribution in islands can also be attributed to the facilitation effect of shrubs, due to both higher concentration of soil nutrients and lower soil temperature and evaporation under their canopy.6,7 Other authors8,9 argue that these bare areas are due not to climatic factors but to the action of grazing, so once this situation is reached, they will hardly be revegetated but, on the other hand, the desertification process observed in many of these systems would advance from these areas,10,3 which thus tend to be dominated by shrubs. The area of land globally affected by encroachment of woody plants has increased dramatically over the past half century,11 and, consequently, desertification processes.
On the other hand, shrub cover would have favored biological crust development due to shading and protection from herbivore trampling which is known to provoke fragmentation of the crusts.12,13 Some authors14,15 argue that biological surface crusts are "safe places" for seed capture and germination and for seedling establishment, although it is not clear whether all those processes are equally favored. It is not clear, either, if the biological crust is a prerequisite for the development of vascular plants cover or vice versa, or if both processes occur simultaneously. In the eastern Monte ecoregion, biological crusts are mainly composed of mosses, having the highest cover in undisturbed places and beneath the shade of shrubs, and they tend to disappear in sites where the soil surface is altered by grazing, burning or mechanical removal with soil surface alteration.16 It was in those places with moss biocrusts that we found both the largest number of perennial grass seeds and seedlings in previous works.17
After excluding grazing, shrubs also have a beneficial effect on the recovery of degraded areas since shading increases the survival of seedlings.18
In addition to the presence of biological crusts, soils in arid and semi-arid environments exhibit a variety of distinctive morphological features, among which may be mentioned the presence of animal tracks, the development of abiotic soil crusts, and the occurrence of vesicular horizons.19 The appearance of these vesicular crusts is common in areas where vegetation is sparse, and their morphological characteristics may prevent, or at least reduce, the emergence and survival of seedlings in those places,20–22 jeopardizing the stability of the system. The natural factor that determines the surface crusting of soils with fine superficial textures and low organic matter is the impact of raindrops on the bare soil surface.23,15 The kinetic energy of raindrops provokes disintegration of the surface structure of individual particles or small aggregates. These small particles can be drawn into micro depressions and clog the soil pores. If the temperature causes an increase in the pressure of the gas trapped in the soil, vesicles are formed and a thin crust of topsoil is generated.23 The crust acts by sealing the soil surface, increasing its mechanical strength and the surface runoff, and reducing infiltration rates up to 90%;24,15 thus, the emergence of seedlings is affected because of the impedance they provide and the dry pedoclimate they produce.25 Moreover, wind and surface water flow invariably remove seeds that may be deposited on the unprotected soil surface. All those management practices that reduce vegetation cover, such as overgrazing and mechanical tillage, increase soil exposure to the above factors and favor the formation of vesicular crusts in these ecosystems.
Some authors14,15 have shown that continuous and repetitive animal trampling compacts soil aggregates, developing a relatively impermeable surface layer. However, in a trial simulating cattle trampling, Wood et al.26 found that in the treatments without trampling, seedling emergence was very poor (<3%) for several years and locations, the emerged seedlings died early in the summer, and seedlings only appeared where there was some microrelief, moreover this behavior was also observed when soil was disturbed by tillage. These data are consistent with the results obtained by Warren et al.27 who observed that the undulations and depressions in the soil surface favored slower surface water flow and allowed greater infiltration. A biological cover such as litter can help to moderate evaporative soil moisture loss and reduce the formation of physical soil crusts by reducing the impact of raindrops on the surface.5 These studies suggest that grazing management that maintains plant vigor and the contribution of litter may reduce the possibility of soil crusting. On the other hand, cattle footprints can create a network of microsites favorable for the establishment of new seedlings of desirable species.
Due to the small size of the seeds, their physical environment is a differential microenvironment that may have large-scale features and, in that sense, any roughness or alteration of the surface structure which disrupts soil surface becomes the determinant factor for their establishment. In this respect, Boudell et al.28 found a large number of seeds in crevices bordering cryptogamic crusts, suggesting that crevices play a role in determining the small-scale distribution pattern of shrub-steppe plants.
The processes of formation of vegetation islands and the vesicular crusting of the soil between them affect most of the plains and gently sloping areas with silty loam that occupy over 70% of the surface of the Monte ecoregion in Rio Negro province.29 As in other semiarid ecosystems, vegetation in the eastern Monte is grouped in "islands" on phytogenic mounds surrounded by sparsely vegetated areas or bare ground.30,31,16 Many soils have physical characteristics which, together with denudation caused by overgrazing, favor the development of vesicular crusting on the soil surface. Surface crusts are rarely found on phytogenic mounds, whereas the soil in the areas between those mounds usually has some sort of crust formation, and the presence of microsites there with a different micro-relief is due to the existence of some kind of cover.
The objective of our study was to relate the presence of seedlings in different kind of microsites with their type of surface cover in a semi-arid shrub steppe of the eastern Monte ecoregion in Rio Negro province. Our hypothesis is that the emergence and survival of plants is higher in protected sites and lower in uncovered sites with vesicular surface crusts, although ground cover that causes roughness on the surface may also contribute to the retention of seeds and facilitate seedling establishment even outside the shrub-dominated “fertile islands”.
The study was conducted southeast of the Monte phytogeographical province32 in a field located in the northeast of Rio Negro province, Argentina (40º 40 ' S, 64º 45' W). The climate of the region is temperate and semiarid, with an annual mean temperature of 14°C; it rains more frequently in spring and fall and the mean annual rainfall is 260 mm, with high inter-annual variation.29 The dominant soils of these plains are classified as Haplargids and Calciortids, and vesicular surface crusts occur in areas without any vegetation cover.29 The vegetation is a low shrubland dominated by the evergreen shrubs Condalia microphylla Cav. and Chuquiraga erinacea D. Don. ssp. erinacea, accompanied by Monttea aphylla (Miers) B.et H. var. aphylla, with the herbaceous layer dominated by the perennial grass Nassella tenuis (Phil.) Barkworth (syn. Stipa tenuis) and to a lesser extent by Piptochaetium napostaense (Speg.) Hack and annual grasses such as Bromus spp. and Hordeum spp,29 with seed sizes that vary between 1 and 7mm. The biological crust is dominated by the mosses Syntrichia princeps and Ceratodon purpureus. The vegetation is grouped in patches dominated by shrubs, with areas of bare ground or low cover between them. Shrub patches present a convex relief due to the accumulation of sediments trapped by the vegetation and also litter produced by the same shrubs, developing phytogenic mounds.
Our study was carried in spring in a field subject to continuous grazing of cattle and sheep (~0.08 cows or 1.5 sheep ha-1). We placed sample plots (of 7.5cm sides) systematically at every meter along a 250m transection, and recorded the following variables: number of annual seedlings (grasses and forbs), basal cover of perennial grasses, presence and cover of litter, biological crust and gravel (measured as the percentage of the soil surface covered by each of them within the plot), and the presence of vesicular crust and its thickness (by extraction) in the sample plot. By recording the observations, each plot was assigned to one of the following microsites: phytogenic mounds beneath shrubs (B) or between shrubs, classified in flat (F) or rough (R) places, according to their microtopography.
The data were analyzed by non-parametric tests (Kruskal-Wallis) to characterize the microsites using the statistical software INFOSTAT/P 2011,33 and Correlation Coefficients φ were calculated to examine potential relationships between different soil surface covers and presence of seedlings. Finally, we performed a Principal Component Analysis to display all the observations as points on a map, with the purpose of showing associations between microsites, between soil covers, and between soil covers and microsites.
The F microsites were the most abundant in the sampled area although they only represented 61% of the plots, whereas R represented 17% and B, 22%.
We detected significant differences in number of seedlings (Kruskal-Wallis; H2=29.93, P 0.0001, n=256), vesicular crust thickness (H2=94.65, P<0.0001), cover of litter (H2=80.54, P<0.0001), biological crust (H2=46.50, P<0.0001), perennial grasses (H2=38.94, P<0.0001) and gravel (H2=74.52, P<0.0001) between the three microsites (Figure 1).
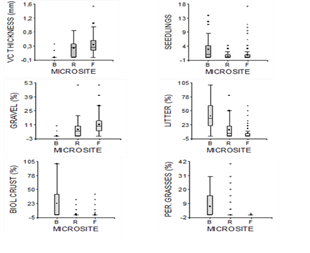
Figure 1 The presence of different covers and seedlings demonstrates diverse values for three microsites (Beneath shrubs, in Flat or Rough spots between shrubs). P<0.05, Kruskal-Wallis test. Diagrams show little symmetry in all of them, and generally more scattered points in the inter-shrub sites.
B microsites had greater cover of litter and biological crust, and also a larger number of seedlings per plot than either of the microsites between shrubs, although they did not differ from the R microsites in the basal cover of perennial grasses (Table 1) (Figure 1). Both gravel cover and vesicular crust thickness were larger in the F microsites than in the others, and larger in R than in B, where CV was also greater for both variables. 62.5% of plots in B microsites produced seedlings, whereas 33.3% and 27.8% of plots in R and F microsites produced seedlings.
Microsites |
|||
Variables |
B |
R |
F |
Seedlings per plot (Number) |
|||
Mean ± SD |
2.73±3.54 |
0.55±0.97 |
0.82±2.24 |
Median |
1a |
0b |
0b |
Vesicular crust thickness (mm) |
|||
Mean ± SD |
0.02±0.08 |
0.27±0.25 |
0.38±0.26 |
Median |
0a |
0.3b |
0.3b |
Basal cover of perennial grasses (%) |
|||
Mean ± SD |
6.61±9.68 |
4.52±9.74 |
0.49±2.51 |
Median |
0a |
0ab |
0b |
Biological crust cover (%) |
|||
Mean ± SD |
22.59±31.9 |
1.90±6.34 |
1.57± 5.37 |
Median |
0a |
0b |
0b |
Litter cover (%) |
|||
Mean ± SD |
39.64±27.9 |
12.74±17.6 |
6.29± 11.63 |
Median |
35a |
5b |
0 b |
Gravel cover (%) |
|||
Mean ± SD |
0.36±1.61 |
6.43±8.64 |
11.45±10.3 |
Median |
12:00 AM |
5b |
10c |
Table 1 Mean (±1SE) and Median of seedlings, vesicular crust thickness and cover percentage of perennial grasses, biological crust, litter and gravel in samples of 56.25cm2 (P<0.05 Kruskal-Wallis test) in three microsites (B= Beneath shrubs, F=Flat or R=Rough spots between shrubs). For each variable, values followed by the same letter are statistically comparable (P>0.05)
The greatest cover found in the F microsites was that of gravel (12%), and in the other two sites, of litter (13% in R and 39% for B).
In Table 2 we present φ Correlation Coefficients between seedlings presence and the different ground covers for the three microsites. This coefficient is positive when the relationship between the two variables goes in the same direction (the presence of one is associated with the presence of the other), and negative when they go in different directions. In our study, the highest negative associations were those which related the presence of seedlings to vesicular crust and gravel, especially in the F microsites (P<0.001), and conversely, the highest positive correlations were those with cover of litter and of biological crust (P<0.001); for the R microsites the clearest positive relationship was the presence of litter, as for the B microsites, and the highest positive correlation was with basal cover of perennial grasses.
Microsites |
|||
Soil cover |
B |
R |
F |
Vesicular crust |
-0.15* |
-0.08ns |
-0.34** |
Biological crust |
0.02ns |
0 ns |
0.17** |
Litter |
0.02ns |
0.11* |
0.22** |
Perennial grasses |
0.34** |
0.06ns |
-0.001ns |
Gravel |
-0.14ns |
0.06ns |
-0.19** |
Table 2 φ Phi) correlation coefficients between frequency of samples with seedlings and different superficial characteristics (vesicular crust thickness, cover of litter and biological crusts, gravel and perennial grasses) in three microsites. Asterisks denote significant differences between microsites (P<0.001)
The frequency of plots with seedlings decreased as the vesicular crust thickness increased (Figure 2), and it increased with the increase of the diverse biological soil covers (Figure 3).
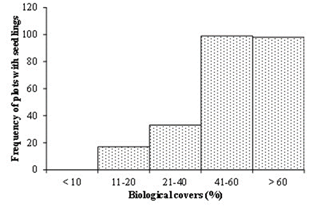
Figure 3 Distribution of plots with seedlings as diverse biological covers (biological crust, litter and perennial grasses together) increase.
The seedling type (forb or annual grass) varied little between microsites (Figure 4), and the latter contributed greatly in all of them.
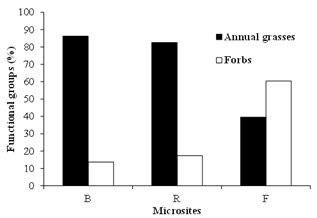
Figure 4 Proportion of functional types of seedlings for the three microsites (Beneath shrubs, in Flat or Rough spots between shrubs).
In Figure 5 it can be seen that the first component separates the physical covers (stones and vesicular crust) from the biological ones (perennial grasses, biological crust and litter), explaining the greatest variability between the sites (94.2%). Observations for the microsite F have relatively high values for the physical soil covers (gravel and vesicular crust) and low values for the biological ones (biological crust, perennial grasses and litter) and the B microsites show the completely opposite situation, whereas the R microsites are in an intermediate situation. Furthermore, both microsites (B and R) show greater dispersion than F, which is very concentrated around the horizontal. Angles between the variables confirm the previously described associations, as all the biological covers are strongly correlated between them (PG with BC and L), as well as the physical types between them (G and VC), and correlations are highly negative between the two types of covers. In addition, the length of all vectors suggests that all of them provide similar contributions.
The loss of vegetation cover in semiarid grasslands is one of the first signals of desertification processes,34,35 and the possibilities of restoring such cover in degraded areas are associated with the existence of an adequate supply of seeds and favorable environmental conditions in which they can establish and prosper.
We found a greater presence of seedling in phytogenic mounds dominated by shrubs than in the soils of the inter-shrub spaces. This is because shrubs create protected microenvironments for greater seed production of herbaceous species so as initial conditions more favorable for germination.36 The facilitation effect may be due to reduced evaporation and herbivores activity7 and the modulation of soil properties.37 However, the initial facilitation effect provided by shrubs would not be necessary for the survival and growth of adult plants that are already established, as suggested here by the lack of differences in cover of perennial grasses between those sites and the rough inter-shrub microsites. In addition, under harsh conditions, shrubs can play a competitive roll with the herbaceous understory in obtaining water,38,39 although the interception of precipitation by woody species and the underlying leaf litter has been shown to reduce soil moisture underneath the canopy of woody plants in semiarid regions.6,40
The homogeneity of the soil surface can cause difficulties for seedling establishment, especially in hard horizons, and the presence of vesicular crusts further reduces radicle penetration and seedling emergence.41 The seedling presence we found beneath the shrubs in the Monte region is consistent with that found in protected sites of other arid ecosystems with vesicular crusts.41,42,26 Nuttall43 showed that in agricultural soil, the hardness of the crust could be used as a general index of the physical condition of soil that prevents seedling emergence. Phytogenic mounds have the advantage of not having strong vesicle surface crusting because the shrubs prevent exposure of the soil surface to both the violent impact of raindrops and alternate cycles of wetting and drying. Erosion of surface sediments from open sites to shrub mounds explains the accumulation of coarse textured soil beneath the shrubs and the lower frequency and thickness of vesicular crusts in those microsites, as well as the increased presence of gravel in the inter-shrub spaces normally covered by those sediments.44 In another study45 where sediments trapped in the mounds were redistributed after a wildfire, we found that seedling frequency, cover and density increased with sediment thickness.
Nash et al.46 showed that microtopography loss due to the impact of livestock on the piospheres exacerbates erosion and contributes to desertification. Both biological crusts and leaf litter were greater in the phytogenic mounds than the inter-shrub spaces; the former as it is very sensitive to disturbances and tends to decrease in cover due to trampling,13 and litter because it is produced in greater amounts under the same shrubs. The main factor associated with the appearance of seedlings in both kinds of microsites in the open patches of this grassland was the cover of litter on the soil surface, and it was more significant in the flat sites; this is to be expected, as litter constitutes a layer that retains more seeds.
In the intershrub areas, the thickness of the crust did not differ between flat and rough sites but it influenced negatively the appearance of seedlings in the flat spots, where seeds can be swept away by either wind or water.
All these characteristics point the importance of some kind of roughness on the soil surface for it to become suitable to capture seeds and favor seedling emergence, thus introducing the possibility to refute the conclusion of Busso et al.47 that “seedling establishment will not be possible for any species of the plant community in the patch interspaces under the conditions of the (Monte) region”. This dependence on microsites for seedlings to become established was also observed by Leder et al.,36 in the Monte.
Domestic herbivores would play a decisive role in the evolution of this system through two opposing effects:
A small disturbance (<2m in diameter) reduces the chances of soil stripping, showing how the inverse relationship between the size of a disturbance and its frequency of occurrence has significant effects on the plant community over time.
Although it is true that the appearance of hard horizons with superficial vesicular crusts can only take place in soils with certain textures, soil denudation is also an essential requirement for this to occur, and high stocking rates and/or continuous grazing can contribute to this process. It would be necessary to carry out grassland management practices that ensure the protection of soil surface through maintenance of the plants and biological crusts that cover it. That cover and its contribution to the input of litter and organic matter in the soil offer the only possible solution to reduce physical crusts and the negative effect that they entail for the retention of seeds and the establishment of seedlings. This would be the key to making the revegetation of these systems possible by creating the appropriate microenvironments.
When the study was carried out, CONICET (National Council for Scientific Research) gave the financial support. As Gustavo Cecchi passed away when we began to write this article, I am especially grateful for the statistical assistance that my friend and colleague Susana Polo M.Sc. gave to me, so that I could finish the paper.
When the study was carried out, CONICET (National Council for Scientific Research) gave the financial support.
The authors declare that they have no conflicts of interest.

©2022 Kröpfl, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.