MOJ
eISSN: 2573-2919


Research Article Volume 6 Issue 2
1Environmental Management Unit, College of Agriculture, Engineering and Science, Bowen University, Nigeria
2Department of Biological Sciences, Fourah Bay College, University of Sierra Leone, Nigeria
3Department of Environmental Health Sciences, Faculty of Public Health, College of Medicine, University of Ibadan, Nigeria
Correspondence: Ogungbile PO, Environmental Management Unit, College of Agriculture, Engineering and Science, Bowen University, P.M.B 284, Iwo-Osun State, Nigeria, Tel 234-8037708479
Received: February 23, 2021 | Published: March 25, 2021
Citation: Ogungbile PO, Akande JA, Adepoju AO, et al. Bioaccumulation of heavy metals in Oreochromis niloticus (Linn.1758) of Agodi Reservoir, Ibadan, Nigeria. MOJ Eco Environ Sci. 2021;6(2):35-41. DOI: 10.15406/mojes.2021.06.00211
The concentration of Cd, Co, Cu, Fe, Mn, Ni, Pb, and Zn in Oreochromis niloticus(generally called Tilapia fish) was assessed at three different locations along Agodi Reservoir in Ibadan. Their relative concentrations were determined using Atomic Absorption Spectrophotometer (AAS). Test location differences and seasonal variations of the values were examined using statistical analysis of variance at P<0.05 to establish pollution dynamics. The results showed declining order of heavy metal concentrations in the order of Mn>Fe>Zn>Cu>Co>Cd>Pb>Ni. Gradient effect was indicated. While Cd was found in decreasing order of concentration from inlet to the outlet, other metals like Co, Cu, Fe, Mn, Ni, Pb, and Zn had concentrations that gradually increased from inlet to the outlet.The concentration of Cd, Co, Cu, Pb, and Zn did not change considerably with season unlike Fe, Mn, and Ni that exhibited substantial seasonal variation. It was concluded that heavy metal accumulation caused marked pollution dynamics inside the Agodi Reservoir. Also, bio-accumulation of the metals in Oreochromis niloticus could give birth to location and seasonal dependency.
Keyword: Agodi Reservoir, Oreochromis niloticus, heavy metal, bioaccumulation, gradient effect
Fishes are vertebrate animals with gills that live inside water. Freshwater fishes are those that spend their lives in freshwater like rivers, lakes, and reservoirs with low salinity of less than 1.05%.1 Fish is a salient asset for humans, especially as food, commercial, or livelihood, and has had a role in the way of life through the ages serving as the supreme being and the subject of art, books, and movies.2 According to Picker and Griffiths,3 fish are consumed as food virtually in all parts of the world. They are a significant source of protein and other nutrients for humans. Fishes are important in the aquatic food chain, sensitive to pollutants that can either be organic or inorganic including heavy metals (such as Hg, Pb, As, Cd, Cr, and Cu). Heavy metals are so characterized as having molecular weights heavier than water at 18g/mol. Heavy metals find their way into water bodies from concentrated wastewater, sewage, industrial activities, and mining operations.4 They contaminate soil systems, water sources, and humans exposed to heavy metals through food consumption. Some metals are poisonous to humans even in small concentrations.
In every ecosystem, organisms are intricately interconnected through food chains and food webs. When heavy metals find their way into an organism, they can build up and linger on a phenomenon called bioaccumulation. Michael et al.,5 defined bioaccumulation as a process in which a chemical pollutant enters into an organism and is not excreted, but accumulated in the tissues. The two main causes of bioaccumulation are the amount of the chemicals coming into the living organism faster than the living organism can assimilate and the build-up of contaminants such as heavy metals or pesticides in living organisms.6 Aquatic organisms are often subject to bioaccumulation because they absorb contaminants from the water faster than their bodies can excrete them. Humans are also subject to bioaccumulation, either from consuming contaminated aquatic organisms or from exposure to contaminants either in food, air, or water. Heavy metals do not biodegrade, which means they can last for a long time in the system.7 Heavy metals could bind to the surface of microorganisms including phytoplankton in oceans and sometimes enter the cells. Once they enter into the cells, heavy metals can react with chemicals released by microorganisms to digest food and undergo chemical transformations Elley et al.8 In this process, heavy metals bio-accumulate in the bodies of living organisms, and by the time they are assimilated, they can constitute health effects in the human body.
There are many reported studies on the bioaccumulation of heavy metals in fish. Elleyet al.8 assessed heavy metals in water, sediment, and fish tissues from Sharkia province in Egypt. Their study revealed that the muscles, gills, kidney, and liver tissues of fish samples collected from industrial drainage and sewage canals had the highest level of heavy metal residues followed by agriculture drainage canals. Similarly, Elleyet al.8 carried out studies of bioaccumulation of some heavy metals in fish samples from River Benue in Vinikilang, Adamawa State, Nigeria. They observed differences in trace metal concentrations between different tissues within the fishes. The level of metals bioaccumulated in tissues of Tilapia zilli amd Clariasangullaris differ. The highest level of all the metals was observed in the gills and liver of the four fish samples while their flesh showed the lowest values. Fabio et al.9 investigated the concentrations of mercury (Hg), cadmium (Cd), zinc (Zn), chromium (Cr), and lead (Pb) in muscle, liver, and spleen tissues of Pesdoplatystoma corruscans specimen collected from two sites on the Paraopeba River, Brazil. The results of their findings indicate that the level of heavy metal concentrations in the tissues was often higher in viscera (i.e. liver and spleen) than in muscle, and thus the viscera should not be considered for human consumption.
The differences that occur among the various heavy metals in the fishes are compounding. Primarily it could be due to variation in the concentration of contaminant in the water bodies.10 Olgunoglu et al.11 reported that heavy metal accumulation is influenced by external factors including dissolved metals physicochemistry, dissolved oxygen, interactions between metals, sediment, food, seasonal effects, geographical differences, and internal (individual variability, body size and development stage, sex, breeding condition, brooding, molding and growth, behavior), physicochemical properties of the contaminants, its distribution pattern in the aquatic ecosystem, the feeding mode, lipid content in the tissue and the metabolism of the aquatic organism.12 Also according to the literature bioaccumulation by fish and subsequent distribution in organs is inter-specific by the type of tissue/organ, age, size of fish, exposure period, mechanisms of uptake, intrinsic factors, and environmental conditions of the habitat of fisheries.12,13
Heavy metals have biological functions. Heavy metals are dangerous to life once the level exceeds the tolerable limit over a long period of exposure.10 Also, extremely low concentration could harm the organisms. Izah et al.,10 evaluated the function of some essential heavy metals that are needed by human bodies. For example, iron has an important role as a constituent of enzymes, such as cytochromes and catalase, and oxygen-transporting proteins such as hemoglobin.10 The deficiency of iron could cause various types of diseases.14 Unfortunately, heavy metals do have serious health effects on humans. Many play a role in cancer development or cause internal organ damage even at low concentrations of Cd, Co, Pb, Ni, and Hg which are also known to affect the formation of blood cells and make the metals react to the surface of the cells, making them less elastic and therefore, less able to circulate throughout the body.15
The objective of this work is to study heavy metal pollution inside Agodi Reservoir, a man made water body that existed in Ibadanfor several decades by evaluating the concentration of heavy metals in Oreochromis niloticus caught from three different locations in the reservoir.
Study area
The study was carried on Agodi Reservoir located in Ibadan, Oyo State, Southern Nigeria. Ibadan is located on longitude 3˚5’ East of Greenwich meridian and latitude 70˚23’N west of the equator. Agodi reservoir was formed along Ogunpa River that flows into the reservoir. The reservoir has an area of 13 hectares stretching about 1km with a maximum depth of about 5m. The river upstream is often polluted by deposited solid and liquid waste products resulting from anthropogenic activities. The sources of pollution are predominantly sewage, urban run-off, and agricultural effluents consisting of chemical fertilizers, pesticides, industrial sources associated with different types of chemical pollutants which are both organic and inorganic, containing heavy metals and microbial organisms. Agodi Reservoir is therefore subject to enormous anthropogenic stress that deteriorates the water quality and contaminates the aquatic system including the fishes (ref).
Sampling sites
The samples were collected for 12 months comprising of dry and wet seasons. They were cropped with the help of a cast net from three sampling locations identified as inlet (F), center (G), and outlet (H) of the reservoir (Figure 1).
Material handling
The fish samples were washed with distilled water and kept in the icebox and transferred to the laboratory for further treatment. In the laboratory, the fish were dried individually to constant weights kept in polythene bags after capturing and dried individually to constant weights in an oven set at 105˚C according to AOAC.16 Thereafter, the dried samples were grounded into a fine powder with mortar and pestle, ready for digestion.
Analytical procedures
The digested fish samples were analyzed for Cd, Co, Cu, Fe, Mn, Ni, Pb, and Zn using Atomic Absorption Spectrophotometer. The instrument setting and operational conditions were done following the manufacturers' (BUCK 200) specifications.
Statistical Analyses
Data collected were subjected to one-way analysis of variance to determine significant differences in the heavy metal concentration found in the fishes at the sampling locations.
Concentrations of heavy metals in test fishes
Table 1 presents heavy metal concentrations in the test fishes between the dry and wet seasons. The overall mean of the heavy metals in a calendar year of 12 months showed declining concentrations in this order Mn>Fe>Zn>Cu>Co>Cd>Pb>Ni.
|
Heavy metals |
Agodi Reservoir |
|||||||||
|
Dry season |
Mean |
Wet season |
Mean |
Overall mean |
FAO std |
|||||
|
F |
G |
H |
F |
G |
H |
|||||
|
Cd (mg/kg) |
8.77 |
0.14 |
0.06 |
2.97 |
0 |
0.14 |
0.12 |
0.09 |
1.53 |
0.2 |
|
Co (mg/kg) |
0 |
0 |
11.06 |
3.69 |
0.22 |
1.22 |
1.68 |
1.04 |
2.37 |
5 |
|
Cu (mg/kg) |
5.23 |
2.85 |
5.98 |
4.69 |
6.51 |
15.76 |
16.88 |
13.05 |
8.87 |
3 |
|
Fe (mg/kg) |
0.63 |
0 |
0 |
0.21 |
102.44 |
511.38 |
1012,19 |
542 |
271.1 |
0.5 |
|
Mn(mg/kg) |
75.19 |
64.13 |
48.69 |
62.67 |
53.88 |
609.79 |
966,66 |
543.44 |
303.06 |
0.5 |
|
Ni (mg/kg) |
0 |
0 |
1.44 |
0.48 |
0.6 |
1.08 |
1.27 |
0.98 |
0.73 |
67.9 |
|
Pb(mg/kg) |
0 |
0 |
0 |
0 |
0 |
0.29 |
6.04 |
2.11 |
1.06 |
2 |
|
Zn (mg/kg) |
187.05 |
82.07 |
95.07 |
121.4 |
174.15 |
223.37 |
312,24 |
236.59 |
179 |
30 |
Table 1 Dry and wet seasons variations of heavy metals in Oreochromis niloticus along Agodi Reservoir
T-test statistic at 95% C.I. was employed to determine seasonal variations while regression curves were plotted to show indicative slopes, gradients effect, and correlation coefficients (Figures 2–9).

Figure 2 Concentration of Cd in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.

Figure 4 Concentration of Cu in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.
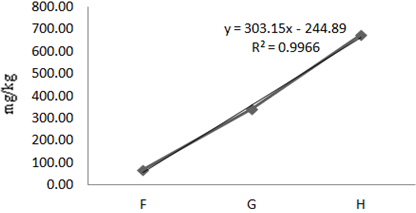
Figure 5 Concentration of Fe in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.
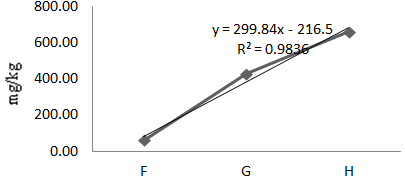
Figure 6 Concentration of Mn in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.

Figure 7 Concentration of Ni in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.
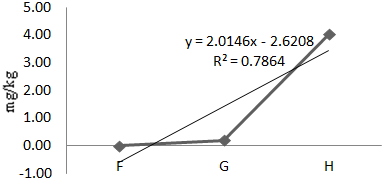
Figure 8 Concentration of Pb in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.
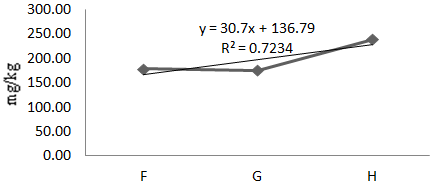
Figure 9 Concentration of Zn in Oreochromis niloticus along test locations (F,G,H) in Agodi Reservoirs.
Interpretations of the curves are presented in Tables 2–4 where the gradient, seasonal variations and two-tailed correlation coefficients between the heavy metal concentrations are respectively presented. The following explanations summarize the bio-accumulation patterns of each of the analyzed heavy metals in the captured fishes relative to their locations in the reservoir and the season of the year they were cropped.
|
Locations |
F |
G |
H |
r2 |
|
|
Metals |
|||||
|
Cd |
77% |
||||
|
Co |
85% |
||||
|
Cu |
92% |
||||
|
Fe |
99.60% |
||||
|
Ni |
97% |
||||
|
Mn |
98% |
||||
|
Pb |
77% |
||||
|
Zn |
|
|
|
72% |
Table 2 Gradient of heavy metal concentrations in Oreochromis niloticus with sampling locations in Agodi Reservoir
r2 is the correlation coefficient of the data
|
Reservoir |
Gradient along test locations on Agodi Reservoir ANOVA at P<0.05 |
Seasonal variation T-test Statistic at P<0.05 |
||
|
Heavy metals |
Fish F – H |
r2 |
dy/dx |
|
|
#Slope |
Fish F – H |
|||
|
Cd |
-ve |
0.77 |
1 |
0 |
|
Co |
+ve |
0.85 |
1 |
0 |
|
Cu |
+ve |
0.92 |
1 |
0 |
|
Fe |
+ve |
1 |
1 |
1 |
|
Mn |
+ve |
0.98 |
1 |
1 |
|
Ni |
+ve |
0.97 |
1 |
1 |
|
Pb |
+ve |
0.79 |
1 |
0 |
|
Zn |
+ve |
0.72 |
1 |
0 |
Table 3 Heavy metals analyses for gradient and seasonal variation for Oreochromis niloticus in Agodi Reservoir
Note: For gradient (dy/dx) and seasonal variation (1) significant established (0) = Not significantly established #slope is slanting course from horizontal
|
|
Cd |
Co |
Cr |
Cu |
Fe |
Mn |
Ni |
Pb |
Zn |
|
(mg/kg) |
(mg/kg) |
(mg/kg) |
(mg/kg) |
(mg/kg) |
(mg/kg) |
(mg/kg) |
(mg/kg) |
(mg/kg) |
|
|
Cd |
1 |
||||||||
|
(mg/kg) |
|||||||||
|
Co |
0.046 |
1 |
|||||||
|
(mg/kg) |
|||||||||
|
Cr |
0.031 |
0.011 |
1 |
||||||
|
(mg/kg) |
|||||||||
|
Cu |
0.085 |
0.09 |
0.258 |
1 |
|||||
|
(mg/kg) |
|||||||||
|
Fe |
0.099 |
0.039 |
0.192 |
.351* |
1 |
||||
|
(mg/kg) |
|||||||||
|
Mn |
0.067 |
0.087 |
.461** |
1 |
|||||
|
(mg/kg) |
|||||||||
|
Ni |
0.067 |
.426** |
-0.065 |
-0.103 |
.495** |
0.132 |
1 |
||
|
(mg/kg) |
|||||||||
|
Pb |
0.012 |
0.045 |
0.006 |
.382* |
.480** |
0.015 |
1 |
||
|
(mg/kg) |
|||||||||
|
Zn(mg/kg) |
0.055 |
-0.052 |
-0.081 |
0.098 |
|
|
0.322 |
0.122 |
1 |
Table 4 Two-tailed correlation coefficient between heavy metals in Oreochromis niloticus
**Correlation is significant at 0.01 level (2-tailed)
*Correlation is significant at 0.05 level (2-tailed)
Cd
The mean concentration of Cadmium in Oreochromis niloticus samples captured during the experiment was 1.53mg/kg (Table 1). Analysis of variance at P<0.05 showed that the values were significantly different across the locations. A linear regression of Cd in Oreochromis niloticus along the sample locations showed a strong negative correlation of 77% (Eqn. 1, Figure 2). The mean value during the dry season was 2.97mg/kg and the wet season was 0.09mg/kg (Table 1). T-test statistic at 95% C.I. showed no seasonal variation (Table 3).
(1)
Note: that in all equations 1 to 8 presented
Y=specific heavy metal measured in mg/kg (dependent variable)
X= distance from source in meters (independent variable)
Co
The mean concentration of Co during the experimental year was 2.37mg/kg. Analysis of variance at P<0.05 showed that the mean values did not differ across the locations, hence, no gradient effect. A linear regression of cobalt in Oreochromis niloticus along the sample locations revealed a strong positive correlation of 85% (Eqn. 2, Figure 3). The mean value of Cobalt in the dry season was 3.69mg/kg and the mean value for the wet season was 1.04mg/kg.T-test statistic showed no significant seasonal variation (Table 3).
(2)
Cu
The mean value at Cu in Oreochromis niloticus during the experimental year was 8.87mg/kg (Table 1). The mean values were significantly different (P<0.05) from the reservoir inlet through the center to the outlet locations which signify the gradient effect. Linear regression of Cu in Oreochromis niloticus along the sample locations showed a strong positive correlation of 92% (Eqn.3, Figure 4). The mean values of the dry season and wet season were 4.69mg/kg and 13.05 mg/kg respectively. T-test statistic at 95% C.I. showed that the values were not significantly different and thereby, suggested no seasonal variation (Table 3).
(3)
Fe
The mean concentration of Fe during the experimental year was 271.10mg/kg (Table 1). At P<0.05, the analysis of variance of the means showed significant differences of iron in fishes at various locations. A linear regression line of iron in Oreochromis niloticus along the sample locations showed a strong positive gradient at a correlation of 99.9% (Eqn. 4, Figure 5). That means, as one move from location F to G to H, there is a strong positive increment in Fe concentration. The concentration of Fe in the dry season was 0.21mg/kg and the concentration of Fe in the wet season was 542mg/kg. Iron varied with the seasons and the variations were statistically significant at 95% C.I. (Table 3).
(4)
Mn
The mean concentration of Mn in Oreochromis niloticus for the experimental year was 303.06mg/kg (Table 1). The values differed significantly (P<0.05) across the sample locations which imply a gradient effect. A linear regression of Mn in Oreochromis niloticus along the sample locations revealed a strong correlation of 98% (Eqn. 5, Figure 6). In other words, as one moves from location F to G to H, there is a strong positive correlation that Mn will increase in the fish. The mean values of Mn in the fish for dry and wet seasons were 62.67mg/kg and 543.44mg/kg respectively (Table 3). Manganese varied with season and the variations were statistically significant.
(5)
Ni
The mean concentration of Nickel in Oreochromis niloticus during the experimental year was 0.73mg/kg (Table 1). At P<0.05, the analysis of variance of the means showed significant differences in Nickel in fishes at various locations which imply a gradient effect. A linear regression line of Ni in Oreochromis niloticus along the sample locations revealed a positive correlation of 97% (Eqn. 6, Figure 7). The mean value of Nickel in the dry season and wet season were 0.48mg/kg and 0.98mg/kg respectively (Table 3). T-test statistic at 95% C.I. also showed significant seasonal variation as more solid wastes containing Ni were dropped in the river during the rainy season.
(6)
Pb
The mean concentration of Pb in Oreochromis niloticus during the test period was 1.06mg/kg (Table 1). Analysis of variance of the means at P<0.05 showed a significant difference in the locations. A linear regression of Pb in Oreochromis niloticus along the sample locations gave a positive correlation of 79% (Eqn. 7, Figure 8). Notably, Pb was not detected in Oreochromis niloticus during the dry season but 2.11mg/kg was obtained in the wet season. T-test statistic at 95% C.I. showed no significant difference between the seasons which implied no seasonal variation (Table 3).
(7)
Zn
The mean concentration of Zn in Oreochromis niloticus during the test period was 179.00mg/kg (Table 1). Analysis of variance of means along sample locations showed no significant difference which implies no gradient effect. A linear regression of Zn in Oreochromis niloticus is positive with a 72% correlation coefficient (Eqn.8, Figure 9). The mean value of Zn in Oreochromis niloticus during the dry season was 121.40mg/kg while the wet season showed a value of 236.59mg/kg. T-test statistic at 95% C.I. showed no seasonal variation (Table 3).
(8)
The interrelationship between heavy metals in Oreochromis niloticus
Two-tailed correlation coefficient (r) values for heavy metals in fish are presented in Table 4. Co concentration in Oreochromis niloticus is positively correlated with Ni at P<0.01. A significant positive correlation also occurred between Fe and Ni at P<0.01 but with Pb at P<0.05. Mn concentration is positively correlated with Pb at P<0.01.
Biological essential heavy metals include Cu, Ni, Fe, and Zn. They are required by a wide variety of enzymes and other cell components also having a crucial role in all living organisms, but high concentrations can have unfavorable health effects.17,18 Non-biologically essential heavy metals include Pb and Cd. They have no biological function and hence are dangerous to live organisms even at low levels, but become toxic as well at higher concentrations.19 In this study, the heavy metals concentrations in the fish Oreochromis niloticus were recorded in the following order Mn>Fe>Zn>Cu>Co>Cd>Pb>Ni with concentrations of essential elements higher than non-essential except for Ni. These consequences may confirm the essential role of the former to fish species.17
In the present study, the concentrations (mg/kg) of heavy metals in for Cd (1.53), Cu (8.87), Fe (271.10), Mn (303.06), and Zn (179.00) were higher than the permissible limits of Cd (0.20), Cu (3.00), Fe (0.500), Mn (0.500) and Zn (30.00) for fish (FAO, 2007), while concentrations of Co (2.37), Ni (0.73), Pb (1.06) were below FAO (2007) permissible limits of Co (5.00), Ni (67.90) and Pb (2.00).
In O. niloticus, Cd, Co, Ni and Pb were within 0-100mg/kg range, Cu between 0-20mg/kg while Fe, Zn, and Mn could be 700mg/kg maximum (Table 1). These results demonstrate that apart from Fe, Mn, and sometimes Zn which have their concentrations greater than 100mg/kg, Co, Cu, Cd, N, and Pb were present in smaller quantities. T-test statistics for the heavy metals in O. niloticus variation of Fe, Mn, and Ni were established from wet to dry season through the reservoir, Cd, Co, Cu, Pb, and Zn showed no significant difference in the two seasons (Table 2). Generally, the concentrations of heavy metals in fish were higher during the wet season (Table 1). The concentration of Cu, Fe, Mn, Ni, Pb, and Zn were higher in the wet season than in the dry season.These results were following those obtained by Saddiq20 who found that heavy metals concentration showed seasonal variations, being greater in winter and lower in summer. This could be due to run-offs. This may further be attributed to the phytoplankton growth which was higher in summer and can absorb large quantities of heavy metals from water.
The results of heavy metals analyses in Oreochromis niloticus in the reservoir from the inlet to the outlet as shown in Figures 1–8 and Table 2 revealed that gradient existed in fishes obtained from Agodi Reservoir. Cadmium was found in decreasing order of concentration from the inlet to the outlet sources (Figure 1) (Table 2). The concentrations of Co, Cu, Fe, Mn, Ni, Pb, and Zn were found to be significant concentrations that increased from the reservoir inlet to the outlet (Figures 2–8) (Table 2).
Relatively high r2 values above 70% were obtained (Table 2). The correlation powers of Co (r2=8.5%), Cu (r2=92%), Fe (r2=99%), Ni (r2=97%), Mn (r2=98%), Pb (r2=79%) and Zn (r2=72%) were strong enough to establish the positive gradient effect from the inlet to the outlet sources of the reservoir. The visible exception was Cd that attenuated from the inlet F to the center G and H outlet part of the reservoir. The deductible gradient effect in the reservoir may be attributed to the compelling gravitational or centrifugal accumulation of heavy metals as the water flow rate slowed down in the reservoir. Heavy metals discharged into the water body may accumulate in the body of the fish and not be excreted.21–23
Heavy metal concentrations in Oreochromis niloticus in Agodi Reservoir showed that all the heavy metals bioaccumulated in the fish. There were significant variations in Fe, Mn, and Ni loadings between the wet and dry seasons. In general higher concentrations of heavy metals were recorded in the wet season for Cu, Fe, Mn, Ni, Pb, and Zn than in the dry season. This may be due to increased metal pollutants into the aquatic ecosystem during the wet season. However, the heavy metal concentrations in fish Cd and Co in the dry season were higher than in the wet season.
Heavy metal concentrations in sampled fish increased from the inlet to the outlet except for Cd. The results of the linear regression of heavy metal concentration revealed that a gradient effect existed in the fishes obtained from Agodi Reservoir.. Heavy metals are important pollutants to the fish because they cannot be eliminated from the aquatic ecosystem by natural methods. The presence of Cd and Pb in the fish is a serious matter of concern which may expose human beings to the heavy metals from consuming fishes obtained from the reservoir.
None.
None.
The authors declare that there is no conflict of interest.

©2021 Ogungbile, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.
 International Day for Biological Diversity, 2026 is observed every year on May 22nd
to emphasize the importance of protecting nature. The theme for 2026 is “Acting locally for global impact.” This day reminds
us of the need to protect nature, preserve species, and promote harmony with our planet. Together, we can safeguard biodiversity
for a sustainable future. On this occasion, MOJ Ecology & Environmental Sciences invites researchers to contribute articles in the
field of Ecology and Environ.
International Day for Biological Diversity, 2026 is observed every year on May 22nd
to emphasize the importance of protecting nature. The theme for 2026 is “Acting locally for global impact.” This day reminds
us of the need to protect nature, preserve species, and promote harmony with our planet. Together, we can safeguard biodiversity
for a sustainable future. On this occasion, MOJ Ecology & Environmental Sciences invites researchers to contribute articles in the
field of Ecology and Environ.