Journal of
eISSN: 2373-6453


Editorial Volume 10 Issue 2
1Faculty of Medicine, Western University, Thailand
210th Zonal Tuberculosis and Chest Disease Center, Thailand
3Department of Pathology, Faculty of Medicine, Chiang Mai University, Thailand
Correspondence: Attapon Cheepsattayakorn, 10th Zonal Tuberculosis and Chest Disease Center, 143 Sridornchai Road Changklan Muang Chiang Mai 50100, Thailand, Tel 6653140767, 6653276364, Fax 6653140773, 6653273590
Received: June 05, 2023 | Published: June 5, 2023
Citation: Cheepsattayakorn A, Cheepsattayakorn R, Siriwanarangsun P. Designing of pan-coronavirus fusion inhibitors and vaccines with targetable elements in SARS-CoV-2 S2 subunit. J Hum Virol Retrovirol. 2023;10(2):43-45. DOI: 10.15406/jhvrv.2023.10.00264
Receptor-binding domain (RBD), a major antigenic determinant is the development-of-fusion-inhibitor and -of-COVID-19-vaccine critical target as the sole antigen.1–5 COVID-19-vaccine efficacy has greatly decreased due to poorly conservation of the multiple-mutated RBD with the new emergence of the Omicron subvariants that contributes to existing-neutralizing-antibody (nAb)-immune evasion.6–10 Several developed COVID-19 vaccines and nAbs demonstrates ability limitation of new-highly-pathogenic-human-coronaviruses (HCoVs)-infection prevention and treatment due to their weak cross-neutralization activity.11,12 SARS-CoV-2 S2 fusion subunit has three monomers that each composes of 1,273 amino acids and establish an S1 and S2 subunit heterodimer, forming head and stalk region, respectively (Figure 1).13 Characteristic coronavirus, including various zoonotic coronaviruses, HCoVs, and SARS-CoV-2 variants are demonstrated in Figure 2.13 Any antibody interfering with the process of pre-fusion conformation by S1 and membrane-fusion induction accompanying the S2-post-fusion change (Figure 3).13 For antibody recognition and neutralization, R815 within the SARS-CoV-2 “ fusion activation ” proteolytic site is critical (Figure 4).13
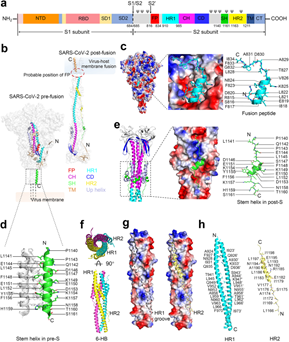
Figure 1 Demonstrating the structural features of targetable elements (fusion peptide, stem helix, and heptad repeats 1 and 2) in SARS-CoV-2 S2 subunit.
a Schematic structure of SARS-CoV-2 spike protein. The conserved glycosylation sites within the SARS-CoV-2 S2 subunit are shown and marked as ψ.
b Conformational transition of SARS-CoV-2 S2 subunit from pre- (PDB: 6XR8) to post-fusion (PDB: 7E9T) state.
c The electrostatic surface representation of the FP binding area and the structural illustration of the FP in pre-fusion spike (PDB: 6XR8). The segment corresponding to FP is shown in sticks and colored in cyan.
d The stem 3-helix bundle is shown in pre-S trimer (PDB: 6XR8). The detailed residues are shown in sticks and colored in green.
e The electrostatic surface representation of the SH binding area and structural illustration of SH in post-fusion spike (PDB: 6XRA). The segment corresponding to SH is shown in sticks and colored in green.
f–h Structure of the SARS-CoV-2 six-helix bundle (6-HB) in the post-fusion conformation (PDB: 6LXT). The symmetry-related structure of the 6-HB bundle is shown. Structures from a top view (up) and a side view (down) are presented in (f). The electrostatic surface of the HR2 binding site is shown in (g). The detailed hydrophobic and hydrogen-bond interactions between HR1 and HR2 are shown and labeled in (h). The distance cut-off is 3.1 Å for hydrogen-bond contact.13

Figure 2 Demonstrating the sequence features of targetable elements (fusion peptide, stem helix, and heptad repeats 1 and 2) in coronavirus S2 subunit.
a Amino acid mutations in the S2 subunit of SARS-CoV-2 variants (https://covariants.org). The number of mutations in S1 and RBD are demonstrated.
b Phylogenetic tree of typical coronaviruses, including SARS-CoV-2 (MN908947.3), RaTG13 (QHR63300.2), PANG/GD (QLR06867.1), SARS-CoV (AY278554.2), WIV1 (AGZ48828.1), HKU3 (QND76034.1), HCoV-HKU1 (YP_173238.1), HCoV-OC43 (NP_937950.1), MERS-CoV (QJX19948.1), HCoV-229E (NP_073551.1), and HCoV-NL63 (YP_003767.1), built via MEGA and maximum likelihood analysis of spike amino acid sequences.
c Sequence identity of S and its targetable elements among SARS-CoV-2 and other coronaviruses.
d–f Alignment of the fusion peptide (d) and stem helix (e), and HR1 and HR2 (f) among typical CoVs. The helical bundles of FP in the prefusion state and SH in the post-fusion state are outlined by a rectangle box. Residues composed of HR1-HR2 six-helical core are outlined. The dots represent similar residues, and the short lines represent missing residues.13
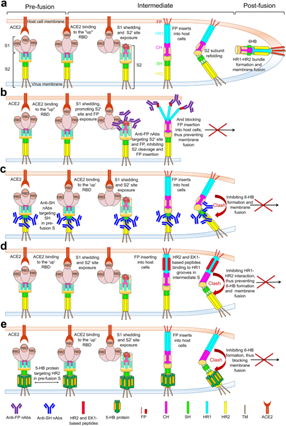
Figure 3 Demonstrating the proposed anti-viral mechanism for antibodies and fusion inhibitors targeting S2.
a An overview of SARS-CoV-2 spike conformation change and its mediated membrane fusion. After the standing RBD engages ACE2 and S1 subunit dissociates, S2ʹ site will expose and be cleaved by the host proteases, followed by the insertion of FP into host cell membrane and the formation of HR1-HR2 six-helix bundle (6-HB). Subsequently, membrane fusion occurs between viral particles and host cells.
b–e Proposed mechanism of virus neutralization by anti-FP nAbs (b), anti-SH nAbs (c), HR2 or EK1-based peptides/lipopeptides (d), and 5-HB proteins (e). Anti-FP nAbs bind to the uncovered S2ʹ site and FP after S1 dissociation, inhibiting S2ʹ cleavage and/or preventing FP insertion into the host cell membrane. Anti-SH nAbs bind to SH in pre-S, disrupting 6-HB formation to prevent membrane fusion. 5-HB proteins bind to HR2 in pre-S trimer and HR2/EK1-based peptides bind to the HR1 groove in the intermediate state of S, both of which would block 6-HB formation. FP fusion peptide, HR1 heptad repeats 1, CH central helix, SH stem helix, HR2 heptad repeats 2, TM transmembrane domain, CT cytoplasmic tail, nAbs neutralizing antibodies, HB helix bundle, ACE2 angiotensin-converting enzyme 2.13
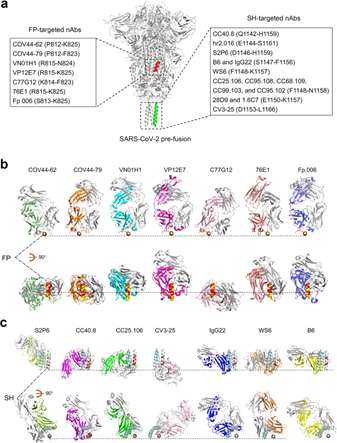
Figure 4 Demonstrating the broad-spectrum therapeutic nAbs targeting fusion peptide and stem helix.
a S2 epitopes recognized by FP-targeted nAbs and SH-targeted nAbs are listed along with the SARS-CoV-2 pre-fusion S protein (PDB: 6XR8). Helical fusion peptide region (R815-V826) is colored red, and stem helix (P1140-S1161) is in green.
b, c The binding mode of each nAb to fusion peptide (b) or stem helix (c). Each nAb-epitope peptide complex is aligned with the SARS-CoV-2 pre-fusion S protein (PDB: 6XR8) over fusion peptide (P812-V826, red) or stem helix (P1140-S1161, palecygan). The nAbs-bound peptides in the complex are colored yellow (b) and red (c), respectively. The light chain of each nAb is colored in gray, and the heavy chain is in a different color to distinguish.13
In conclusion, other targetable S2 elements in antibody recognition and neutralization and anti-COVID-19-therapy development include HR1-HR2 bundle, stem helix, and fusion peptide.
None.
None.
Author declares that there is no conflict of interest.

©2023 Cheepsattayakorn, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.