Journal of
eISSN: 2374-6947


Review Article Volume 4 Issue 2
1CSIHM Centro de Salud Integral del Hombre y la mujer, Mexico
2Anahuac University Mexico North Campus, Mexico
3Universidad Nacional Autónoma de México, Mexico
Correspondence: Zoraida Axtle Serrano, Department of Nutrition and Metabolism, CSIHM Centro de Salud Integral del Hombre y la mujer, Tehuantepec 251 cons 506, México City, México, Tel 011 554 165 2834
Received: March 08, 2017 | Published: April 5, 2017
Citation: Sánchez JML, Berebichez-Fridman R, Rodríguez SR, et al. Pharmacoeconomic meta-analysis of oral dual therapy options in overweight patients with uncontrolled diabetes. J Diabetes Metab Disord Control. 2017;4(2):62-68. DOI: 10.15406/jdmdc.2017.04.00107
Introduction: The American Diabetes Association (ADA) recommends metformin as the first line treatment of diabetes. If appropriate control is not achieved with metformin, oral dual therapy with four different options should be considered.
Objectives: To compare the effects of using various oral dual therapies on cost-utility by quality adjusted life-years (QALYs) in overweight patients with uncontrolled diabetes, to obtain diabetic control by homeostatic model assessment (HOMA) and HgA1c evaluation in time.
Methods: Literature search was performed in PubMed from June 2000 to September 2016 using the search terms metformin plus the next options: canagliflozin, pioglitazone, sitagliptin, and glimepiride. QALY reports were adjusted at each option by the report of failure percentage. Data from preclinical and Phase 1 and/or 2 trials were reported when appropriate. After that model-based cost-utility analysis was performed using a lifetime horizon, and a Markov model was constructed and populated with probabilities, outcomes, and cost data from published sources, including 1-way and probabilistic sensitivity analysis.
Results: The dual therapy options iSGLT, TZD, IDPP4, and SU were used to reduce the relative risks of high HgA1c levels and represent a control for each 3month evaluation. Following glycemic control, we improved QALYs in terms of years without insulin (0.046), which resulted in lower costs per patient ($956). The impact of iSGLT on weight was high with estimates of relative risk reductions (p<0.001) and QALY gains. iSGLT and IDPP4 were more expensive than others, but QALYs were better for these drugs. Among these two drugs, iSGLT2 achieved better QALY with long-term use.
Conclusion: This analysis suggests that canagliflozin and metformin combination is the best option for body weight reduction. Diabetic education helps to gain more years of control, increase the QALYs, decrease glucose, weight, pressure, and lipids by adequate exercise resulting in improved outcomes and reduced costs.
Keywords: hga1c, metformin, canagliflozin, qalys
BMI, body mass index; HbA1c, glycated hemoglobin; HOMA, homeostasis model assessment; QALYs, quality adjusted life-years
Diabetes causes 1.5 million deaths worldwide and is currently the seventh leading cause of deaths in the United States.1 In addition, higher-than-optimal blood glucose causes an additional 2.2 million deaths, increasing the risks of cardiovascular and other diseases.2 In 2016, the American Diabetes Association (ADA) reported that 1.4 million Americans are diagnosed with diabetes every year1 with an economic impact of $263.2 billion dollars invested directly for its prevention and management. The International Diabetes Federation suggests that the worst is yet to come, estimating a 9.9% growth in diabetic population by 2035.3
The diabetes attention and awareness it’s difficult to diagnosis because most of the times the cases can be asymptomatic and remain undiagnosed for several years, with complications often manifest at the time of diagnosis. Undiagnosed diabetes accounts for up to 50% of all cases, these been associated with poorer cardiovascular outcomes.4
Pre-diabetes, defined as having a blood glucose concentration higher than normal but lower than diabetes thresholds, is also becoming increasingly common. It is estimated that pre-diabetes affects 35% of all adults in the United States, rising to > 50% in those aged 65 and over. Up To 70% of individuals with pre-diabetes will go on to develop diabetes at a rate of 5-10 Per year, but the estimate of the total outcomes measures impact in world economic is nowadays a real public health epidemic, because increasing the screening and the appropriate medical care do not change the fact that the most important measure is prevention with education and a healthy life style.5 however, most of the people do not modify their lifestyle, Needing treatment for their glycemic control.
Patient education, life-style modifications, and exercise helps in the reduction of glycaemia,6-8 the additional support of medical therapy is usually necessary, and most of the times a β-cell function evaluation of the newly diagnosed patients is required to direct the medical therapy to insulin resistance.9 Metformin is the ideal first-line therapy of diabetes owing to its safety and important effects on glycated hemoglobin (HbA1c) and body weight.10,11 The second-line therapy involves adding multiple oral hypoglycemic agents to metformin, leaving incorporation of insulin as the last step of choice when oral treatments are no longer effective.
HbA1c is used as the gold standard test to evaluate a patient’s response to treatment, usually reevaluating every 3 months to achieve control.12 If a patient fails to achieve HbA1c values below 7.5%, progression therapy or insulin should be considered, with an estimated duration of at least 3 years after failure to achieve the control values.13 Therapeutic intervention should be adjusted over time14 owing to the continuous decline in β-cell function that leads to deteriorating glycemic control in type 2 diabetes patients,15 which is usually detected by HbA1c value greater than 9%.16
The association of diabetes to obesity is correlated with chronic low-grade inflammation and immune system activation accompanied by insulin resistance and it has been referred to as “diabesity”17 a condition that accelerates the progression of medication to triple therapy or insulin.18 The implications of this association rely on the multiple pathologies presented in these patients, failure to achieve lasting lifestyle modifications,19 and continuous disobedience of the patients to medical indications despite deteriorating health.20 All these problems affect blood glucose levels and are associated with metabolic syndrome, another contributing factor that helps to accelerate the natural history of the diabetic patient.21 Add risk factors for T2DM, such as BMI values over 25 kg/m2, have been investigated but have not consider the undiagnosed T2DM is correlated with other specifically identified risk factors and whether undiagnosed T2DM patients might be distinguished from diagnosed T2DM patients by these risk factors.22
The initiation of insulin therapy in the afore mentioned patients has been associated with worse prognosis in terms of the required insulin units, mainly because higher baseline HbA1c and homeostasis model assessment (HOMA-IR) elevation involves higher insulin dose requirements.9 Diabetes by itself has a personal and financial impact; the health care costs of patients with diabetes are high. In patients who do not adhere to lifestyle modifications, the health care cost has been reported to be higher than that reported in previous economic papers.23 Moreover, the health care costs in obese decontrol diabetic have more economic impact, and clinical prognosis.24
If the uncontrol diabetic patient finally response to a medication that provides normal glucose levels, that effect is directly associated with reduction in morbidity and mortality, that also delay the progression of the physiopathology. That effect produce great savings, as well as increase the quality adjusted life-years (QALYs) for patients and the respective National Health Services, resulting in the significant reduction of the overall economic burden.25 The aim of the current analysis is to track the impact of body mass index (BMI) at overweight and obese unfollow diabetic education patients to investigate the QALYs express at decrease of weight, directly indicating insulin resistance decrease and glycemic control.
Literature search was performed in PubMed from June 2000 to September 2016 using the search terms metformin plus the next options: canagliflozin, pioglitazone, sitagliptin, and glimepiride. QALY reports were adjusted at each option by the report of failure percentage. English-language articles focusing on Phase 3 clinical trials for diabetes patients with non-control were analyzed. Data from preclinical and Phase 1 and/or 2 trials were reported when appropriate. Then, a meta-analysis of data was performed to conduct a model based cost-utility analysis using a lifetime horizon, with outcomes measured in terms of QALYs for economic evaluation. Studies had to meet the following criteria to be included in the analysis: Phase 3 clinical trials for diabetes patients with non-control; include DMT2 patients, basal treatment with metformin, uncontrol glycemic values measure by basal HgA1c measure, body weight with BMI over 29; also, basal measurements and after dual therapy. Studies that have a BMI beyond 29, DM1 diagnosis, or treated with insulin were excluded from our analysis. The percentage of dropout result in metabolic control and impact in weight was adjusted by the results reported in each group as an additional outcome measure.
Model structure
A long-run Markov state-transition model was used to estimate the expected costs and outcomes over an estimated life-time horizon of 10 years, using cycles of 3 months because of HgA1c control report (Figure 1). The data were compiled from the clinical trials of each prescription. The costs were calculated using the price presented in US$ on the official FDA cost page in October 2016. In the long-run model, all costs, and outcomes after the first year were adjusted by moving from the failure rate reported for each drug to the need of insulin, represent by QALYs.
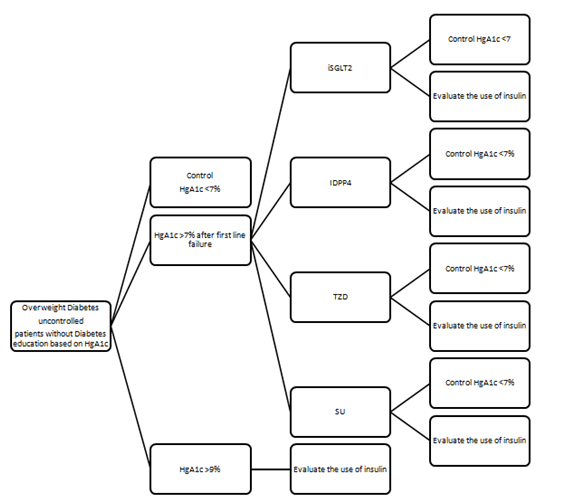
Figure 1 Markov-model of uncontrolled Diabetes education based on levels of HgA1c base on treatment.
The layout in this Figure is the natural evolution of Overweight Diabetes Uncontrolled Diabetes patients without diabetic education based on HgA1c measures. Each box corresponds to one or more states in the HgA1c levels. Parts of the model is that baseline the patient have metformin, if we have less than 7% of HgA1c means control and need evaluation after 3 months, if their parameters are higher 7% we add in the second line the dual therapy with the four options study if in the study the HgA1c>9 we directly evaluate the use of insulin.
Management strategies
Four oral medical treatment options (canagliflozin, pioglitazone, sitagliptin, and glimepiride) were considered for controlled or uncontrolled therapy results per the HgA1c value. Only data from recently published randomized controlled trials using metformin and one of the four medical treatment options were used. The clinical outcomes were based on the baseline change of HgA1c measured 90 days after treatment, which were assumed to be affected by BMI, lipids, and baseline blood pressure (BP) change. Finally, β-cell function was assessed using the HOMA report.
Costs
The treatment cost of an obese diabetic patient was estimated depending on the controlled and uncontrolled blood glucose levels as well as the associated risks. For cost patients, the price per patient/year was $56,458 and for not high-cost patients, was $4,674 per patient/year.26,27 This included the cost of medication (assuming that all patients had BMI over 25) associated with previous metformin treatment (Table 1). Oral diabetes medication costs were estimated using the published data for each treatment option (see Table 1 in cost Data Supplement).
|
iSGLT2 Canagliflozin |
TZD Pioglitazone |
IDPP4 Sitagliptin |
SU Glimepiride Amaryl 6 mg |
Price/Month |
448 |
401 |
415 |
36 |
Price/3 Months |
1,344 |
1,203 |
1,245 |
108 |
Price/Year |
5,376 |
4,812 |
4,984 |
432 |
Price/10 Years |
53,760 |
48,120 |
49,800 |
4,320 |
Table 1 Estimate of Cost of Antidiabetic Oral Agents in Obese Diabetic Patients in US dollars30
The cost of acute management of patients in the first ninety-day treatment was attributed to glycemic control or the need for other therapy if they did not achieve their goal representing the ongoing annual costs taken from a published report. We evaluated the glycemic control from the HgA1c values. The cost of other medicines, such as antihypertensive and lipid-lowering agents, is not the focus of this paper; however, the impact of these medications might be worth considering in future studies to optimize economic and clinical results.
Outcomes
A Markov model was used to distribute a theoretical cohort of 1000 patients into multiple oral medications of dual therapy administered over time. The additional result of control and need of insulin therapy or triple management were included. The cohort of patients receiving the intervention was estimated using the United Kingdom Prospective Diabetes Study (UKPDS) model. QALY combined the length of life and quality of life, latter being measured by utility scores. A utility score of 1 represented control and a score of 0 indicated the need for insulin.
Probabilities
The probability of being independent, dependent, or dead in each treatment arm was calculated using the data obtained from eight randomized controlled trials. We applied the transition probabilities transformed for 3-month cycles. Based on HgA1c control, the probability was adjusted by moving from a state of control to a state of insulin requirement.
Cost-utility evaluation
The total QALYs and costs were calculated by multiplying the number of patients in each state by the calculated utilities and costs for that state. Cost-utility was measured in terms of the incremental cost per QALY gained and the Net Monetary Benefits (NMB) of weight loss, decreased insulin resistance, glucose stabilization, and gaining insulin-free years were compared among the four dual therapy options. The NMB was calculated as the mean QALY per patient accruing from the treatment multiplied by the decision maker’s maximum willingness to pay for a QALY advantage for the treatment based on weight loss and glycemic control.
Using the pharmacoeconomic base-case values, medical insulin therapy was used to treat diabetic overweight patients after dual oral medication. The cost report demonstrated that the cheapest option was SU (glimepiride) compared to the other three oral agents, representing 10% of the spending. The other 3 groups were similar in cost with a variability of 5-13%, the iSGLT2 (canagliflozin) group being the group with the highest cost (Table 1). The baseline parameters are represented in Table 2. With regards to the effect of various treatment combinations, the major impact on BMI was observed in the iSGLT2 (p<0.001) group. TZD (pioglitazone) and SU treatment induced weight gain (Table 3).
|
iSGLT2 Canagliflozin Invokana 300 mg |
TZD Pioglitazon Actos 45 mg |
IDPP4 Sitagliptin Januvia 100 mg |
SU Glimepiride Amaryl 6 mg |
HgA1c (%) |
8 (1)* |
8.8** |
7.9±0.9 (63±9.8)+ |
7.8±2--- |
FPG (mmol/L) |
9.6(2.4)* |
9.4±2** |
9.4±2.3+ |
>55ml/min--- |
Body weight (Kg) |
86.9(20.5)* |
81** |
87.7±21.6+ |
85±16--- |
BMI (kg/m2) |
31.7(6.0)* |
30.4** |
32.0±6.1+ |
32±1--- |
Duration of Diabetes (years) |
4.3(4.7)* |
4.6** |
6.8±5.2+ |
5.4±2--- |
Systolic BP (mmHg) |
128.5(12.7)* |
125.3** |
128.0±13.5+ |
128.0±4--- |
Diastolic BP (mmHg) |
79.1(8.3)* |
80±4** |
77.5±8.0+ |
79±2--- |
Triglycerides (mmol/L) |
2.0 (1.1)* |
2±1** |
2.0±1.1+ |
1.9 (1.2) |
HDL-C (mmol/L) |
1.2 (0.3)* |
2±0.1** |
1.2±0.3+ |
1.2 (0.3) |
LDL-C (mmol/L) |
2.6 (0.9)* 2.8±0.9+ |
2.6±0.7** |
2.6±1.0+ |
2.7 (0.9) |
HOMA (%) |
53.5(29.5)* |
|
|
|
|
iSGLT2 |
TZD |
IDPP4 |
SU |
HgA1c (%) |
-1.03* |
-1.7±0.1** |
-0.73±0.05 |
-0.74- |
FPG (mmol/L) |
-2.4* |
|
-1.0±0.1 + |
-0.96-- |
Body weight (Kg) |
-2.9* |
+1,8 |
-1.2±0.2 + |
+0.01- |
BMI (kg/m2) |
-3.3* |
|
|
1.0% (0.2)*** |
Systolic BP (mmHg) |
-5.4* |
-0.4** |
-0.7±0.6 + |
-1.03- |
Diastolic BP (mmHg) |
-2.0* |
-0.2** |
-0.3±0.4 + |
0.9- |
Triglycerides (mmol/L) |
-0.18 *(0.007) |
-1.4** |
-0.15±0.05 + |
-0.01 (0.05)*** |
HDL-C (mmol/L) |
0.11 *(0.002) |
0.8** |
0.06±0.01 + |
-0.01 (0.01)*** |
LDL-C (mmol/L) |
0.12 *(0.005) |
1.2** |
-0.04±0.04 + |
0.05 (0.04)*** |
HOMA (%) |
20.3 *(2) |
|
|
31 |
HgA1c (%) |
-2.5 * |
0.9** |
-0.73±0.05 |
0.4±0.2-- |
QALYs |
2.9 |
1.2 |
0.6 |
0.2-- |
Cost per QALY |
-13,112.19 |
-8,296,55 |
-7,778,25 |
-654-- |
The incremental cost-utility of overweight diabetic patients was compared among the four dual oral therapies, and iSGLT2 showed the highest QALY followed by IDPP4 (Figure 2) (Table 4). The dual therapy combination with iSGLT2 was projected to reduce the relative risks of having higher HgA1c levels compared to TZD, IDPP4, and SU, thereby representing a control at each third-month evaluation. After attaining glycemic control, we estimated the improved QALY in terms of control years without insulin (0.046), decrease in weight and BP (p=0.003), and impact on insulin resistance. The model showed that the four treatment options were generally similar, but the impact on weight was higher for iSGLT2, with estimates on relative risk reductions (p <0.001) and QALY gains. In addition, the associated cost differences were smaller. Both iSGLT and IDPP4 were more expensive than the others, but QALY was better for patients treated with these drugs. Among these two drugs, iSGLT2 resulted in better QALY upon long-term use, achieving savings of $13,112 dollars in ten years, with controlled weight and glucose.
Medication |
SGLT2 |
TZD |
iDPP4 |
SU |
QALYs |
2.9 |
1.2 |
0.6 |
0.2 |
Table 4 The Effect of dual therapy on QALYs
The results of this analysis suggest that among the four oral dual therapies administered to diabetic patients, SU is the cheapest option; however, it is not associated with any change in weight. Furthermore, iSGLT2 and IDPP4 groups are associated with decreased BMI, and iSGLT2 300 mg leads to gain in QALYs and reduction in HOMA-related cost, clearly indicating that it helps in the control of glucose levels. Greater reductions in HbA1c, systolic BP (SBP), and weight were observed in this study compared to those in other studies. The analysis of insulin metabolism affected at Markov model resulted in similar cost-utility evaluation.
The analysis of dual therapy in obese patients in all clinical trials showed additional benefits such as decrease in BP and weight in all cases which is independent to the insulin mechanism.28 When the blood glucose level increases linearly and exceeds the threshold of 180 mg/dL, it can result in glycosuria. However, taking a diuretic can lower the BP as a compensatory mechanism.
Another effect that was observed in our population was the change in weight, which was consistent with previous studies.29 In overweight diabetic patients, weight loss is associated with easier glucose control. In fact, an education program is recommended as a part of diabetes management to achieve weight loss. A lower BMI improves metabolic profile and reduces drug doses. In the case of treatment with canagliflozin (300 mg), patients’ weight reduced by 4.2%.30
In the economic evaluation, iSGLT2 had the most positive impact on QALYs, secondary to the regulation of weight, glucose, and BP, involving multiple high-cost sodium-glucose cotransporter-2 (SGLT) inhibitors that improve QALYs. The cost is compared by the price of the drug per ten years by simulation of QALY gains. This is probably explained secondary to the control of HgA1c owing to the decrease in microvascular lesions during glucose control in obese patients. Additionally, the evidence of from clinical analysis of these patients indicates that insulin mechanism may not be the first line of treatment for HbA1c decrease. Weight loss and BP reduction is the key to these results because the association of metabolic syndrome and diabetes is high in long-term models.31-35
This analysis opens the possibility to apply larger economic values in long-term clinical trials involving overweight diabetic patients by combining clinical evidence and optimizing the economic burden of patients not only in short-term trials, but also in long-term trials. This can help in optimizing therapeutic decisions in obese patients.
The results of this study illustrate that weight-related changes are important in the cost-utility analysis of overweight or obese patients with uncontrolled diabetes managed with dual therapy. The combination of canagliflozin and metformin is the best option with respect to weight reduction, HOMA, and QALYs gain in terms of more years of control. Diabetes education can improve outcomes and reduce the costs for patients. These pharmacoeconomic evaluations not only benefit the economic view, but also have clinical impact on overweight patients by enabling QALY gain.
J.M.L.S, Z.A.S. and E.B.C were responsible for defining the research question. Designed the strategy for the literature search. M.P.O., and S.A.S., participated in study selection. Data analysis was performed by S.R. and R.B.; L.F.M, A.G.L.V and E.P.R. were the major contributors in manuscript writing.
Author declares that there is no conflict of interest.

©2017 Sánchez, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.