Journal of
eISSN: 2574-9943


Research Article Volume 2 Issue 6
Professor, Chaudhary Charan Singh University, India
Correspondence: Rameshwari Thakur, Professor, Chaudhary Charan Singh University, Meerut, U.P. U-18/75, F.F. DLF, Phase- 3, Gurugram- 122002, Harayana, India, Tel +9196 5477 5082
Received: October 11, 2018 | Published: November 14, 2018
Citation: Thakur R, Kalsi AS. Clinico-mycological study of onychomycosis in Botswana. J Dermat Cosmetol. 2018;2(6):95-100. DOI: 10.15406/jdc.2018.02.00095
Background: Onychomycosis is a fungal disease of the toenails or fingernails that may include any part of the nail unit, including the matrix, bed, or plate. The causative organisms of onychomycosis are dermatophytes, Candida (yeasts), and non-dermatophytic molds, associated with different clinical presentations.
Methods: A retrospective study was carried out including the patients diagnosed with onychomycosis at the National Health Laboratory, Gaborone (Botswana), from January 2009 to December 2009. The objective of the study was to find out the predominant species and most common clinical form of onychomycosis in Botswana.
Results: 59 patients were diagnosed with onychomycosis (37 females and 22 males). Trichophyton violaceum (T. violaceum) was found to be the predominant species (44 patients, 74.58%) with male to female ratio of 10:34 and the commonest clinical type was endonyx onychomycosis (45 patients, 76.27%), followed by distal subungual onychomycosis (6 patients, 10.17%), white superficial onychomycosis (4 patients, 6.78%) and 4 (6.78%) cases of candida paronychia. Fingernail involvement was more common (48 patients, 81.35%) compared to toenail involvement (11 patients, 18.64%, all males).
Conclusion: Efforts ought to be made for the precise diagnosis and timely treatment of onychomycosis as it does not ordinarily cure by itself and can trigger more infectious lesions in other parts of the body. The treatment of onychomycosis is aimed at the eradication of the causative organism (both clinical and mycological cure). HIV status should be investigated where multiple fingernails or toenails are involved.
Keywords: Trichophyton violaceum, Trichophyton interdigitale, Trichophyton tonsurans
The term ‘Onychomycosis’ includes tinea unguium and also infection by non-dermatophyte fungi including yeasts. In 80-90% cases, the fundamental causative pathogens are Trichophyton rubrum and Trichophyton interdigitale.1‒4 About 2-11% cases of onychomycosis are due to non-dermatophyte fungi such as Acremonium spp., Alternaria spp., Aspergillus spp., Fusarium spp., Scytalidium spp. and Scopulariopsis spp. About 2-10% fungal nail infections are due to yeasts, including Candida spp.5‒9 These infections are frequently under diagnosed because of poor healthcare services in underdeveloped countries. A nail has the function of protecting the distal phalanx and the surrounding tissues from the trauma.10 The prevalence of onychomycosis increases with immunosuppression (HIV/AIDS, diabetes, use of immunosuppressive therapies, cancer chemotherapy or antibiotics), increasing age, repeated injuries on nail, poor hygiene of nails and foot including occlusive foot wears, poor peripheral circulation, repeated exposure to pathogenic fungi and increasing use of health clubs for sports, swimming pools.11
Also, the prevalence of onychomycosis is increasing in Western countries, probably due to lifestyle changes and ageing of the population. About 10% of the general population, 20% of the population aged >60years, up to 50% of people aged >70years and up to one-third of diabetic individuals have onychomycosis.12 Onychomycosis may have a genetic predisposition, but only in a very less percentage of persons.13 The condition of nails represents the social status as observed by Gonzalez-Serva, 1990.14 Onychomycosis constitutes 50% of the nail problems.15
There are five major clinical presentations of onychomycosis
A retrospective study was carried out including the patients diagnosed with onychomycosis at the National Health Laboratory, Gaborone (Botswana), from January 2009 to December 2009. A total of 165 patients either clinically diagnosed or suspicious of onychomycosis were sent to the mycology laboratory for the collection of the samples. To avoid misdiagnosis as nail psoriasis, lichen planus, contact dermatitis, nail bed tumors such as melanoma, trauma, or yellow nail syndrome, laboratory confirmation was performed. The nails were cleaned with 70% isopropyl alcohol and samples were taken. Sterile nail clippers and No. 15 surgical blades were used to collect nail clippings and subungual debris in sterile petri dishes and labeled. Potassium hydroxide (20% KOH) mounts were prepared to look for the fungal elements.
Samples were inoculated on two plates of Sabouraud’s dextrose agar with chloramphenicol and Derm agar. One plate was incubated at 37°C and one at 25°C along with the Derm agar plate. Candida species were differentiated on Chromogenic candida agar. For dermatophytes, KOH and/or culture positive samples were considered positive. For non-dermatophyte molds, both KOH and culture positive samples were considered positive. Also, a second sample was taken and if both KOH and culture were positive, it was considered positive. Patients with only positive direct microscopy and negative culture were excluded. The criteria for the diagnosis of onychomycosis caused by non-dermatophyte molds were based on
When the filaments were seen on the light microscopy but showed a non-dermatophyte growth on culture, then another nail specimen was examined by light microscopy and culture to confirm non-dermatophyte mold. Lacto phenol Cotton blue (LPCB) mounts were prepared in order to study the microscopic structures in details Figure 4. The standard basic tests such as urease and in vitro hair perforation test were performed for the differentiation of T. interdigitale and T. rubrum.
A total of 59 patients were found positive following the criteria and diagnosed with onychomycosis. The most common clinical pattern was endonyx onychomycosis (EO) i.e., 45 patients (76.27%), followed by distal subungual onychomycosis (DSO) 6 patients (10.17%), total dystrophic onychomycosis (TDO) 5 patients, white superficial onychomycosis (WSO) 4 patients (6.78%) and 4 patients (6.78%) cases of candida paronychia (Table 1). We did not found proximal subungual onychomycosis in any of our patients. All the six cases of total dystrophic onychomycosis (TDO) were KOH and culture negative and were not included. These patients were females in the age group of 20-40years.
S. No. |
Clinical types |
Number & percentage |
1 |
Distal Subungual Onychomycosis (DSO) |
6 (10.17%) |
2 |
White Superficial Onychomycosis (WSO) |
4 (6.78%) |
3 |
Proximal Subungual Onychomycosis (PSO) |
0 (0) |
4 |
Endonyx Onychomycosis (EO) |
45 (76.27%) |
5 |
Total Dystrophic Onychomycosis (TDO) |
5 - Not included |
6 |
Candida paronychia |
4 (6.78%) |
|
Total |
59 (100%) |
Table 1 Clinical pattern of onychomycosis
Age group (in years) |
T. violaceum |
T. interdigitale |
T. tonsurans |
Non-dermatophytic molds |
Candida albicans |
|||||
Fingernails |
Toenails |
Fingernails |
Toenails |
Fingernails |
Toenails |
Fingernails |
Toenails |
Fingernails |
Toenails |
|
0-15 |
04 (6.77%) |
- |
- |
01 (1.69%) |
- |
- |
- |
- |
01 (1.69%) |
- |
16-30 |
11 (18.64%) |
- |
- |
01 (1.69%) |
- |
01 (1.69%) |
- |
01 (1.69%) |
02 (3.38%) |
- |
31-45 |
10 (16.94%) |
- |
- |
01 (1.69%) |
- |
- |
- |
02 (3.38%) |
- |
- |
46-60 |
07 (11.86%) |
- |
- |
- |
- |
- |
- |
01 (1.69%) |
- |
- |
>61 |
12 (20.33%) |
- |
- |
- |
- |
- |
- |
02 (3.38%) |
01 (1.69%) |
01 (1.69%) |
Table 1 Study animal information: ID, gender, age, and dates of data collection for each study animal
Species |
Number of isolates |
Male: Female |
Percentage |
T. violaceum |
44 |
10:34 |
74.58 |
T. interdigitale |
3 |
3:00 |
5.09 |
T. tonsurans |
1 |
1:00 |
1.69 |
Aspergillus sp. |
3 |
3:00 |
5.09 |
Fusarium sp. |
2 |
2:00 |
3.39 |
Alternaria sp. |
1 |
1:00 |
1.69 |
Candida albicans |
4 |
0:04 |
6.78 |
Candida parpsilosis |
1 |
1:00 |
1.69 |
Total |
59 |
21:38 |
100 |
Table 3 Onychomycosis pathogens
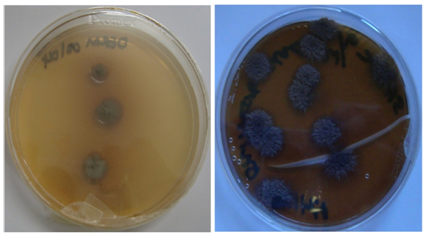
Figure 2 (a) Colonies of T. violaceum with waxy surface. (b) Verrucose colonies of T. violaceum with deep violet pigment.
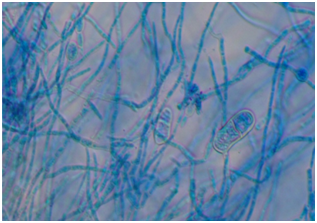
Figure 4 Trichophyton violaceum (Lacto Phenol Cotton Blue mount). Irregularly branched hyphae with intercalary chlamydoconidia and clavate two-celled macroconidia. Magnification X400.
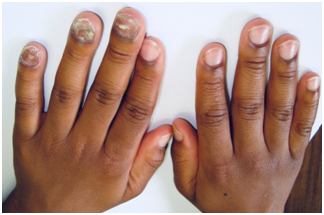
Figure 7 A 10-years old boy with tinea unguium (endonyx type) of left hand affecting nails of middle, ring and little fingers.
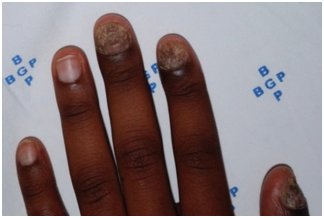
Figure 8 Tinea unguium of the left hand in a 14-years old male, affecting nails of thumb, index and middle fingers.

Figure 10 Tinea unguium of all the fingernails with melanonychia in a HIV positive 51years old female. She was on azidothymidine.
Out of total 59 patients with onychomycosis, 37 patients (62.7%) were females and 22 patients (37.2%) were males (Table 2). Fingernail involvement was more common (48 patients, 81.3%) compared to toenail involvement (11 patients, 18.64%, all males). In the age group of 0-15years, we found four cases of tinea unguium of fingernails due to T. violaceum (Figures 2,4,7,8) and one case of toenail involvement due to T. interdigitale (Figure 1). In three HIV positive females, fingernails were bilaterally involved with culture positive for T. violaceum (Figure 9). 34 (57.62%) females and 10 (16.94%) males had onychomycosis of the fingernails due to T. violaceum (Table 3). Out of four male patients, onychomycosis of toenails due to T. interdigitale was found in 3 patients (5%) (Table 3) and T. tonsurans (Figure 3) was insolated in 1 patient (1.69%). Non-dermatophyte molds were isolated from the toenails of 6 (10%) male patients and candida from 1 (1.69%) male patient. Onychomycosis of the fingernails due to candida was isolated in 3 (5.08%) females and of toenail in 2 (3.38%) male patients (Table 2). HIV status was known in 15 patients only. One HIV positive female patient was on azidothymidine and had melanonychia along with tinea unguium of fingernails due to T. violaceum (Figure 10). Distribution of tinea unguium of the fingernails due to T. violaceum as shown in Figure 11, shows the highest prevalence among the age group 16-30 years and second highest among 31-45years age group. These possibly correspond to the highest prevalence of HIV/AIDS among these age groups.
Onychomycosis is caused by dermatophytes, non-dermatophyte molds (NDMs), and yeasts. Dermatophytes are the main causative agent in temperate climates, whereas Candida and non-dermatophyte molds in tropical regions.16 It is traditionally a disease of the elderly.17 It has a significant impact on patient’s life and may cause psychological, social or employment related problems.18
Overall, dermatophytes are most commonly implicated, accounting for 90% of toenail infections and 50% of fingernail infections, and T. rubrum is responsible for most cases worldwide.19 But scenario here in Botswana differs to a great extent from other geographical locations. In our study, we found T. violaceum as the predominant species accounting for 100% fingernail infections.
Onychomycosis is one of the early manifestations of HIV infection with a prevalence of 15-40%.20 It is four times more common in HIV infected individuals than in the general population, which is reported to be up to 23% when their T-lymphocytes count is as low as 400/mm3 (normal range: 1200-1400/mm3), usually affecting all finger-nails and toe-nails.21‒23 Multiple nail involvement, isolation of both common and rare species, and resistance to treatment are the characteristics of onychomycosis in HIV.20 Proximal subungual onychomycosis is usually an indication of HIV infection.24‒26 Botswana has the second-highest HIV infection rate in the world after Swaziland, with one in three adults infected. Females were infected more than males (21 males, 38 females), this can be related to the high prevalence of HIV in females (28.9%) as compared to males (18.9%) during the period 2007-2009.27 Population migration during the world wars led to the distribution of T. rubrum from its source regions to the new places like Europe and America.28,29
The first documented case of onychomycosis in US has been reported in 1928. 28,30 The prevalence of tinea pedis and onychomycosis increased in 20th century during the World War II and the Korean and Vietnam wars and due to the occlusive footwear and population migration. Vietnam war resulted in increased prevalence of T. rubrum worldwide.31 But in Botswana, 74.58% cases of onychomycosis are due to T. violaceum. The authors did not report a single case due to T. rubrum. The involvement of the fingernails is the most common. The Asian Achilles Survey conducted in the late 1990s confirmed that the prevalence of onychomycosis is relatively low in Asian countries.32
Onychomycosis in children was thought to be more frequent in fingernails due to Candida spp.33 But we found T. violaceum 4 (6.77%) to be the predominant species for fingernail infection in children (up to 15 years of age) as well. The reason for low frequency of onychomycosis in children is due to faster nail growth, lower incidence of tinea pedis and less exposure to spores, less chances of nail injuries and smaller surface area. Patients with Down’s syndrome have more chances of onychomycosis as studied in Mexico.34 High prevalence of onychomycosis in children could be either due to high prevalence of tinea capitis 27 and/or HIV. Other reasons include fungal infections in the other family members, pre-existing tinea pedis, physical activities, trisomy 21, obesity and diabetes mellitus.35 In a study held at Colorado, 46.8% children suffered from onychomycosis, with high prevalence at the age group between 6-10.36 High prevalence of HIV among the population predisposes to increased fungal infections due to low immunity. One study has announced an evidently higher percentage of onychomycosis in HIV-positive patients with respect to general patients visiting dermatologic centers.37 Goodman et al observed that the pervasiveness of dermatophytosis was four times higher among HIV infected people.38
Endonyx type was the most common clinical form of onychomycosis in our study followed by distal subungual onychomycosis (DSO) and total dystrophic onychomycosis (TDO). In this, patients presents with milky-white discoloration of the nail plate without hyperkeratosis or onycholysis. The dermatophyte responsible for endothrix type of hair invasion usually invades the nail.39,40 We found very less cases of onychomycosis due to NDMs (10.16%) and Candida albicans (8.47%), this is possibly due to the fact that NDMs and Candida are the causative agents in tropical regions, whereas Botswana has a semi-arid climate with short rainy season. NDMs accounted for 100% toenail infections, whereas Candida albicans accounted for 80% fingernail infection and 20% toenail infection. The treatment of onychomycosis is aimed at eradication of the causative organism and return to a normal appearance of a nail. Treatment of onychomycosis relies on factors like the patient’s age and inclination of regimen (daily, weekly or monthly pulse therapy), the contaminating fungus and number of nails involved, level of nail association, whether toenails or fingernails are influenced and whether different medications are being taken.41
The first line systemic treatment for onychomycosis is Griseofulvin, Itraconazole, fluconazole and terbinafine.42 An alternative topical treatment with 8% ciclopirox, 5% amorolfine and 40% urea plus 1% bifonazole can also be given but these are generally ineffective, with a failure rate of more than 60%, due to significant nail plate disease or nail matrix involvement. Concomitant nail debridement further increases the cure rates. Lasers and photodynamic therapies are also available. The long term cure is difficult to achieve in patients with onychomycosis.42 Relapses and/or recurrences are common in these patients. The relapses are less common in patients treated with terbinafine compared to itraconazole.42 Long term and continued treatment with efinaconazole 10% topical solution was found to be very effective and appeared to prevent relapses.43 Efforts should be made for the accurate diagnosis and timely treatment of toenail onychomycosis as it does not typically cure itself and can trigger more infectious lesions in other parts of the body.44 According to Mehregan and Gee treatment of onychomycosis should never be started without proper diagnosis of fungal infection.45
Efforts ought to be made for the precise diagnosis and timely treatment of onychomycosis as it does not ordinarily cure by itself and can trigger more infectious lesions in other parts of the body. The treatment of onychomycosis is aimed at the eradication of the causative organism (both clinical and mycological cure). HIV status should be investigated where multiple fingernails or toenails are involved.
The study was conducted in accordance with research ethics.
Authors thank all the individuals who contributed to this study. No funding was received for the study.
The authors declare there is no conflict of interest.

©2018 Thakur, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.
 World Vitiligo Day is a global observance dedicated to raising
awareness about Vitiligo, a chronic skin condition affecting pigmentation. Each year on June 25th,
communities, researchers, and advocates unite to dispel myths, fight stigma, and promote acceptance
of diverse skin tones. This year’s theme, “From Stigma to Strength,” calls for transforming
misunderstanding into empowerment and celebrating resilience and diversity. The Journal of
Dermatology & Cosmetology invites researchers to contribute to advancing Vitiligo treatment and awareness.
Submissions received by June 25th will receive a 40% discount on the Article Processing Charge (APC),
encouraging broader participation and valuable contributions.
World Vitiligo Day is a global observance dedicated to raising
awareness about Vitiligo, a chronic skin condition affecting pigmentation. Each year on June 25th,
communities, researchers, and advocates unite to dispel myths, fight stigma, and promote acceptance
of diverse skin tones. This year’s theme, “From Stigma to Strength,” calls for transforming
misunderstanding into empowerment and celebrating resilience and diversity. The Journal of
Dermatology & Cosmetology invites researchers to contribute to advancing Vitiligo treatment and awareness.
Submissions received by June 25th will receive a 40% discount on the Article Processing Charge (APC),
encouraging broader participation and valuable contributions.