International Journal of
eISSN: 2574-8084


Case Report Volume 8 Issue 4
1St Vincent’s Hospital, 390 Victoria St, Darlinghurst, 2010, NSW, Australia
2GenesisCare, Department of Radiation Oncology, St Vincent’s Clinic, 438 Victoria St, Darlinghurst, 2010, NSW, Australia
3Department of Haematology, St. Vincent’s Health Network, Kinghorn Cancer Centre, 370 Victoria Rd, Darlinghurst, 2010, NSW, Australia
Correspondence: Prof Gerald Fogarty, Radiation Oncology, Mater Sydney, Crows Nest, NSW Australia, Tel +61 2 9458 8050, Fax +61 2 9929 2687
Received: September 13, 2021 | Published: October 6, 2021
Citation: Zhang RLL, Dunkerley N, Wright K, et al. Superior outcomes of Volumetric Modulated Arc Therapy (VMAT) in the palliation of skull metastases of multiple myeloma: a case report. Int J Radiol Radiat Ther. 2021;8(4):139-142. DOI: 10.15406/ijrrt.2021.08.00306
Palliative radiotherapy (RT) is often given to symptomatic sites of multiple myeloma (MM). Three-dimensional conformal radiotherapy (3DCRT) is often the main modality due its efficiency but it may involve irradiation volumes which include organs at risk (OARs). A case of end-stage MM with innumerable skull lesions was treated with volumetric modulated arc therapy (VMAT) which demonstrated superior conformity compared to a 3DCRT plan. VMAT planning yielded an 88% dose reduction to the hippocampi, 60% dose reduction to the brain yet equivalent dose homogeneity to the skull target. Treatment with 15Gy in five fractions gave significant almost complete symptom resolution with no adverse OAR related complications at 4 weeks.As the life expectancy of MM patients and other cancers increases with better systemic therapy, there will be a greater chance of experiencing late effects of RT in OARs. This case supports the notion that perhaps VMAT could be considered in some cases of palliative RT.
Keywords: multiple myeloma, radiotherapy, skull, lytic lesions, bone pain, volumetric modulated arc therapy, homogeneity, conformity, case study
MM, multiple myeloma; RT, radiotherapy; Gy, gray; VMAT, volumetric modulated arc therapy; 3DCRT, three dimensional conformal radiotherapy; OARs, organs at risk; QA, quality assurance; RIBI, radiation induced brain injury
MM is a monoclonal proliferation of plasma cells accounting for approximately 1% of all malignancies.1 It is characterised by anaemia, hypercalcaemia, renal failure and bone pain secondary to lytic lesions.2 Bone pain is the presenting complaint in 70% of MM patients and patients require palliative RT at least once during their treatment course.3 RT has been used for MM related symptoms since 1931.4 In the beginning, techniques such as hemibody RT irradiated large parts of the body to ensure doses of radiation to all the tumour.5 The current MM treatment paradigm recognises RT as an effective palliative treatment for bone pain.6
In many departments, RT treatments with palliative intent are delivered using simpler techniques like three-dimensional conformal radiotherapy (3DCRT). This is done to reduce time to treatment7 and save resources, especially those involved with quality assurance (QA). QA is necessary for more complex techniques such as intensity modulated radiotherapy (IMRT), and the evolution of IMRT to volumetric modulated arc therapy (VMAT).
With appropriate planning all these techniques deliver a homogeneous dose to the planning target volume (PTV).8 However, 3DCRT has a higher integral dose,9 and Organs at Risk (OARs) that may be in the normal tissue volume may inadvertently receive the treatment dose.10 IMRT and VMAT have superior conformity to PTVs. IMRT and VMAT are especially advantageous when there is an OAR in the concavity of the PTV, for example brainstem in the concavity of nasopharynx or brain in the concavity of the scalp.11
With advancements in systemic therapy, overall survival of MM patients now approaches 50% at 10 years after diagnosis.12 Minimizing adverse late effects of RT to OARs should be a priority even in palliation to maximise the quality of survivorship.13 We present a case of an MM patient with osseous and soft tissue lesions in the skull requiring palliative RT for symptom control.
A 54-year-old female was urgently transferred for further management from her local regional hospital with innumerable osseous and soft tissue lesions. Symptoms on arrival included headaches, diplopia worst on upward gaze and left elbow pain. Examination revealed proptosis in the left eye, tender masses in the skull of varying sizes and several others in the limbs. Computed tomography (CT) and magnetic resonance imaging (MRI) of skull base confirmed diffuse skull marrow disease, extensive dural involvement and infiltration of the left lacrimal gland causing proptosis. Previous treatment included cyclophosphamide, bortezomib and dexamethasone. Compliance was poor. She had declined stem cell transplantation. A radiation oncology opinion was sought for palliative RT to thoracic, elbow and cranial lesions. The patient was consented for RT and to be involved in this study, focusing on the skull plan.
Radiation planning and treatment
Our usual protocol was followed. Briefly, to ensure immobilisation, a personalised thermoplastic mask was made. CT slices were taken from skull vertex to clavicle at 2mm slice width. The skull treatment volumes were contoured and these encompassed all the painful and tender areas. OARs were contoured with particular attention to the hippocampi and brain. A radiation script was written, prescribing 15 Gray (Gy) in 5 fractions over 5 days to the skull which is similarly effective as doses > 20 Gy.13,14 Dose restraints were set on brain with a point maximum of 15Gy and mean of 9Gy; and hippocampi with maximum of 9Gy and mean of 5Gy.
The plan was produced on the Elekta Pinnacle Treatment planning system and was accepted by the prescribing Radiation Oncologist (RO). The plan consisted of 2 VMAT Arcs with a 6 Mega Voltage (MV) photon beam. There was excellent conformity of dose to PTV in two perpendicular planes: inferior to superior and left to right. A 3DCRT plan was also completed for comparison. QA was performed on the VMAT plan with Sun Nuclear’s Per FRACTION software and was satisfactory. As seen in Table 1, 3DCRT is quicker to implement however, given the prescription dose constraints to the OARs, the 3DCRT plan was not acceptable.
|
|
VMAT |
3DCRT |
|
Planning time |
120 minutes |
15 minutes |
|
QA time |
12 minutes |
2 minutes |
|
Beam-on time |
85 and 91 seconds for the two arcs |
20 seconds per lateral field |
|
Total time on bed per fraction |
10 minutes |
6 minutes |
Table 1 Comparison of planning, QA and beam-on time between VMAT and D-CRT
Our patient tolerated RT well, only reporting mild nausea. Follow up immediately post treatment revealed a decrease in pain and a corresponding decrease in analgesia. Her regular long-acting opiate requirement had halved from 20mg of oxycodone to 10mg and she no longer required breakthrough oxycodone when discharged a week later. Examination immediately post treatment revealed already that her diplopia had lessened significantly and there was reduction in size of the skull lesions. At four weeks post RT, our patient had continuing alleviation from her symptoms with no pain and no diplopia. She denied any new lesions or new cranial neuropathies. She did not report any mental fog, confusion or memory issues, symptoms typical of brain irradiation.
The dosimetry of 3DCRT compared to VMAT is displayed in Figure 1. Both gave a homogenous dose to the PTV but VMAT had superior conformity of dose to PTV. This is made evident when comparing the dosimetry in two perpendicular planes: inferior to superior and left to right. As seen in Figure 2, 3D-CRT uses large fields to ensure homogeneity to PTV but will inadvertently capture OARs in the concavity of the PTV within such fields. VMAT, on the other hand, achieves the same dose to PTV by irradiating through an arc. As there is dose from almost all directions, this allows for selective dose modulation to avoid OARs.
Figure 1 Comparative dosimetry of the 3DCRT and VMAT plans.
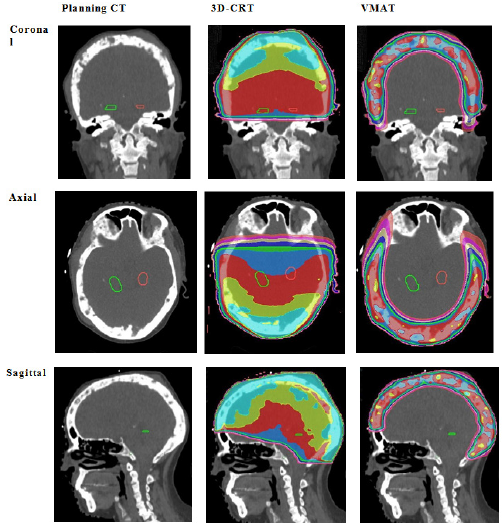
Figure 1A Comparison of baseline Planning CT, 3D-CRT planning volumes and VMAT planning volumes. Hippocampi contoured in green (left) and red (right). The first column shows the planning scan. Note the obvious metastases within the skull. The second column shows the 3D-CRT plan, the third column shows the VMAT plan. The top row shows coronal slices, second row shows the axial slices, and the third row shows the sagittal slices. Note the exposure of healthy brain tissue to significant doses of radiation with 3D-CRT. Descriptive interpretation: this figure compares the dosimetry graphically between VMAT and 3D-CRT in three planes.
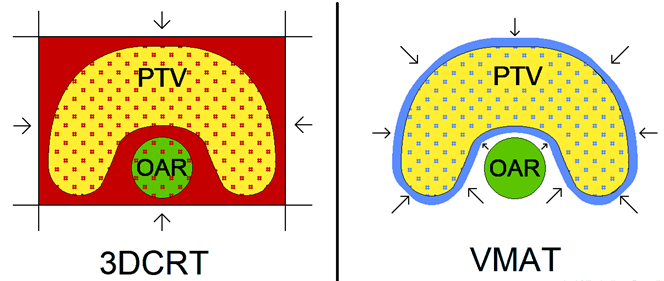
Figure 2A A simplified graphic that compares the general schematic of radiation administration in 3DCRT and VMAT. The arrows represent angles from which the radiation is directed. Note that with 3DCRT, the OAR in the concavity of the PTV receives the full treatment dose, but the OAR is spared with the VMAT plan. Descriptive interpretation: this figure demonstrates that 3D-CRT, unlike VMAT, inadvertently irradiates an OAR in the concavity of the PTV.
|
Area |
|
|
Area of dose in 3DCRT |
|
|
Area of dose in VMAT |
|
|
Planned Target Volume |
|
|
Organ at Risk |
|
|
Radiation exposure in specified area |
Dots |
Figure 2B Dosimetry Legend for Figure 2A.
Our case study demonstrates this. The brain lies within the concavity of the skull. 3DCRT with its wide, fixed fields is not able to treat around the curvature of the skull unlike VMAT. VMAT gave prescription dose to the PTV but reduced dose to the hippocampi and brain OARs, expressed as a dose-volume histogram in Figure 3. The VMAT plan satisfied the prescription and was accepted.
|
Colour of curve |
Modality |
Region of Interest |
Mean Exposure (Gy, % of intended 15 Gy) |
Dose difference (%) |
|
3D-CRT |
Brain |
14.92 (100%) |
60% reduction with VMAT |
|
|
VMAT |
5.59 (40%) |
|||
|
3D-CRT |
Left Hippocampus |
14.13 (93%) |
88% reduction with VMAT |
|
|
VMAT |
0.82 (5%) |
|||
|
3D-CRT |
Right Hippocampus |
14.10 (93%) |
88% reduction with VMAT |
|
|
VMAT |
0.78 (5%) |
|||
|
3D-CRT |
PTV |
15.97 (107%) |
No difference |
|
|
VMAT |
15.12 (100%) |
Figure 3 Dose-Volume Histogram for our patient’s RT. When comparing homogeneity within planned treatment volumes, VMAT is highly comparable to 3D-CRT. However, when comparing exposure to OARs, specifically hippocampi in the green and red, there is significant exposure with 3D-CRT whilst minimal with VMAT. Likewise, there is reduced mean dose delivered to the brain with VMAT. Descriptive interpretation: this figure shows in graphic form that VMAT spares OARs in the concavity of the PTV.
We present a case of a 54-year-old female with MM involving the skull, manifesting with refractory headaches, cranial neuropathy and tender skull masses. Despite the palliative nature of the treatment intent, her potential longevity was kept in mind and radiation induced brain injury (RIBI) was avoided by minimising the amount of healthy brain tissue in the treated volume, especially the hippocampi, as seen in Figure 3. The case shows excellent conformity of dose to PTV with VMAT in two perpendicular planes of convexity: inferior to superior and left to right a seen in Figure 1, clearly demonstrating that VMAT dosimetry was superior to 3DCRT for OAR avoidance.
MM survivorship has significantly improved for newly diagnosed patients with a ten-year survival rate of up to 41%.15 Many other cancers requiring palliative RT are similarly living longer. Many departments continue to prioritise minimising delays13 and maximising cost effectiveness when providing palliative RT, using 3DCRT rather than VMAT.
RIBI is a well-recognised complication of healthy brain tissue irradiation with manifestations including hippocampal associated learning and memory dysfunction,16 dementia17,18 and cognitive impairment.19 In fact, amongst patients with brain tumours survivors, commonly treated with partial or whole brain RT (P-;WBRT), RIBI is considered the second most important outcome measures for quality of life, after survival.20 Jalali et al.21 compared Stereotactic RT (SRT) to conventional RT for brain tumours and found that the population that underwent SRT had superior outcomes in several neurocognitive domains, especially memory. This is supported by Gondi et al.16 who found, when comparing IMRT with hippocampal avoidance and WBRT in patients with brain tumours, there is significantly longer retention of memory when the hippocampi are exposed to less than a mean dose of 9 Gy.
Current common RT techniques for palliative intent include 3D-CRT and even 2D-CRT as they are much quicker to plan and deliver. However, as seen from Figures 1-3, 3D-CRT falls short of VMAT in conformity and, consequently, has been mostly superseded in recent years for radical treatments.22-25 The main barrier to using VMAT in palliative cases is the multiple steps involved in preparation of the RT plan, notably QA. However, comparing newer techniques of RT delivery, VMAT is much quicker than IMRT requiring only 3-5 minutes per fraction as opposed to 20-25 minutes; as IMRT requires repositioning of the gantry in between beams.26-28 With life expectancy, due to better systemic treatments, approaching the time at which late radiation toxicities to OARs develop, our case supports considering VMAT even in palliative treatments.
We present a case of a 54-year-old female with skull bony pain secondary to MM referred for palliative RT. When comparing 3DCRT and VMAT plans, 3DCRT was not acceptable given the dose to OARs. She was treated with VMAT and had excellent palliation of her symptoms at 4 weeks with no sequalae in OARs. With life expectancy, due to better systemic treatments, approaching the time at which late radiation toxicities to OARs develop, our case supports considering VMAT even in palliative treatments.
The authors wish to thank the patient involved for consenting to have her clinical details and photos used. Thanks should also be extended to the GenesisCare radiotherapy department at St Vincent Hospital, Sydney, Australia. Finally, the authors would like to thank Jack Fogarty (jackf.illustrations@gmail.com) for his work on Figure 2.
None.

©2021 Zhang, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.