International Journal of
eISSN: 2574-8084


Research Article Volume 8 Issue 3
1GenesisCare, Mater Hospital, North Sydney, NSW 2060, Australia
2GenesisCare, St Vincent’s Hospital, Darlinghurst, NSW 2010, Australia
3School of Surgery, University of New South Wales, Randwick, NSW 2031, Australia
4Douglass Hanly Moir Pathology, Macquarie Park, NSW 2113, Australia
Correspondence: Gerald B. Fogarty, GenesisCare, Mater Hospital, North Sydney, NSW 2060, Australia , Tel +61(2)9458 8050
Received: July 27, 2021 | Published: August 10, 2021
Citation: Fogarty GB, Moutrie Z, Cusi M, et al. Does psoriasis need more radiation dose to clear recalcitrant plaques? A case series of lesions from a single subject. Int J Radiol Radiat Ther. 2021;8(3):114-120. DOI: 10.15406/ijrrt.2021.08.00302
Psoriasis vulgaris is a chronic benign disease of the whole thickness of the epidermis with symptomatic plaque formation impacting quality of life. Relatively low doses of fractionated radiotherapy (RT) have been used in the past to a total dose of four to six Gray (Gy).
We present the case of an older male with seven recalcitrant plaques each treated with the same prescribed dose of RT. Superficial radiotherapy (SXRT) with a 100kV beam from a Xstrahl 300 machine to a total dose of 20 Gy in 10 fractions (#s) at five #s per week was used for six areas - four on the scalp and one on each elbow. At six months post treatment, four scalp areas had a continuing complete response (CR) whilst the elbows had a continuing partial response (PR).
A seventh area, measuring 21 x 16cm on the lower back, was treated with volumetric modulated arc therapy (VMAT) using six megavoltage photons (MV) at the same prescribed dose. At four weeks, an infield CR was observed, but at eight weeks there was some infield recurrence. Pre- and post-RT biopsies from this field were compared. In the VMAT area with clinical recurrence, RT caused a histological PR. In the VMAT area with clinical CR, there was no histological evidence of disease.
These findings raise radiobiological questions. Was the difference in clearance due to lower disease burden, kilovoltage backscatter from bone giving a higher relative dose, a difference in oxygenation or chronic sun exposure of tissues, or a difference in the relative biological effectiveness (RBE) between SXRT (1.2) relative to MV photons (1.0)?
We recommend that future studies of RT in psoriasis start with 20Gy in 10#s, preferably with a modality that can penetrate the whole plaque epidermis, which can be quite thickened in patients suffering from psoriasis.
Keywords: Radiotherapy, radiobiology, superficial radiotherapy, volumetric modulated arc therapy, psoriasis, psoriatic plaques, benign, case study, histopathology
Psoriasis vulgaris is a chronic benign skin disease affecting around two percent of the adult world population.1 It is caused and maintained by an interplay between genetic risk foci, environmental factors, and immunological abnormalities,2 especially those involving T lymphocytes and dendritic cells. These are present focally in skin and are what leads to localised plaque formation.3 Within these plagues, the epidermis is thickened, and the keratinocyte turnover time is shortened.4 The keratinocytes move through the epidermis over four to five days, which is a tenfold acceleration.3 The psoriatic process happens throughout the whole thickness of the epidermis which has lost the usual maturation pattern found in normal skin.
Psoriasis usually presents clinically as often multiple and symmetrically distributed, red, scaly plaques with well-defined edges. Without treatment, the plaques persist.5 Plaques can be symptomatic with itch and exfoliative desquamation and cosmetically undesirable, thus significantly impacting a person’s quality of life.6
Radiotherapy (RT) has been used to treat psoriatic plaques.7-11 We present a single case involving seven areas of psoriatic plaques treated with RT at the same dose. The administered dose was higher than in the previously cited studies as the plaques were recalcitrant. Some interesting radiobiological phenomena were seen.
A well 72-year old man presented with a long history of psoriasis involving multiple skin plaques and occasional psoriatic arthritis. He had never taken biologicals but had used multiple topical treatments when the plaques were particularly symptomatic. After some time, the lesions would recur in the same anatomical loci.
dThe patient had many plaques amenable to RT. Seven were chosen for an initial course of RT. Those selected were the most obvious, and all had a history of symptomatic relapse following local therapy and could therefore be considered recalcitrant.
Six were treated with superficial radiotherapy (SXRT): the right and left temple, right and left scalps above both pinnas, and both elbows. These were treated with SXRT with a 100kV beam from a Xstrahl 300 machine to a total dose of 20 Gy in 10 fractions (#s) at five #s per week. At six months post treatment, the SXRT treated scalp areas achieved a continuing complete response (CR; Figure 1). At six months, the elbows had a continuing partial response (PR; Figure 2).
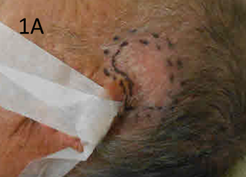
Figure 1A Photo at SXRT planning phase showing scalp above the left pinna. Note the infield erythematous desquamating psoriatic skin.
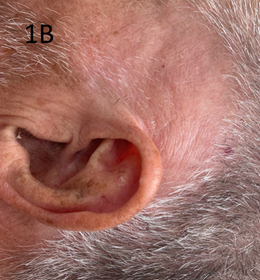
Figure 1B Scalp above the left pinna 8 weeks after treatment with 20 Gy in 10#s. Note the complete resolution of the infield psoriasis.
Figure 1 SXRT of scalp above left pinna.
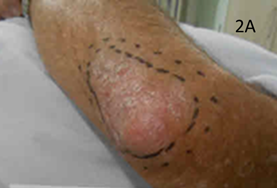
Figure 2A Photo at SXRT planning phase of left the elbow. Note againthe infield erythematous desquamating psoriatic skin.
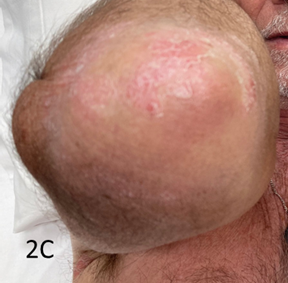
Figure 2C Left elbow at 8 weeks following 20 Gy in 10#s.Note the partialresolution of infield psoriasis.
Figure 2 SXRT of the left elbow.
A large area measuring 21 x 16 centimetres (cm) on the lower back was treated with a VMAT technique12 using six megavoltage photons (MV) to the same fractionated dose of 20 Gy on 10#s. An infield biopsy was taken prior to RT to act as baseline. At four weeks, a CR was observed infield. At eight weeks, however, there was some recurrence infield (Figure 3). Two infield biopsies were taken of what appeared to be normal skin and recurrent disease at eight weeks (Figure 4).
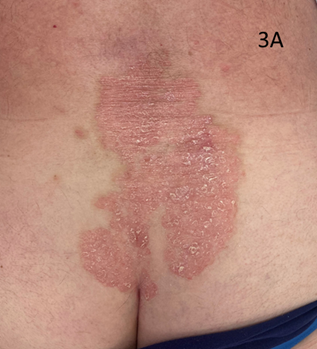
Figure 3A A large psoriatic plaque measuring 21x16cm on the lower back. Note again the erythematous desquamating psoriatic skin.
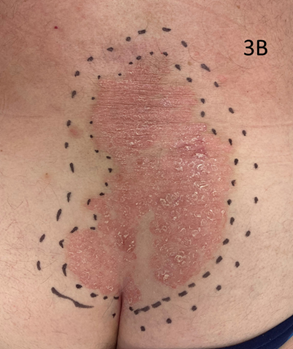
Figure 3B Treatment planning. The radiation oncologist has marked out the clinical target volume (CTV)with a dotted line. This line is within another dotted line which is the planning target volume (PTV).13

Figure 3C Wires have been placed during planning on the marks so they can be captured on the planning computed tomogram (CT) scan. The black arrow marks a bandage that coversthe the biopsy site prior to RT.
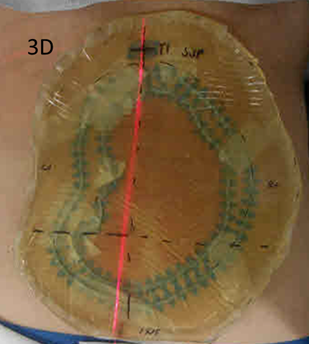
Figure 3D Prior to the planning scan, bolus is placed over the entire field with a margin to ensure 100% dose to skin.
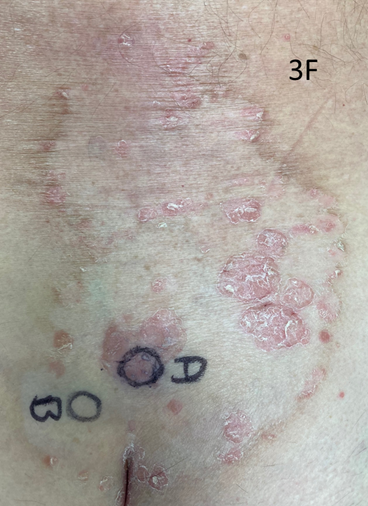
Figure 3F Biopsies were taken of the recurrent disease from position A, and a biopsy of “normal skin” was taken from position B. Position A was very close to the pre-RT biopsy site (see Figure 3C).
Figure 3 A large area measuring 21x16cm on the lower back. This areawas treated with a VMAT technique.
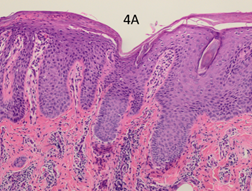
Figure 4A Pre-RT x 100 showing psoriasiform hyperplasia, loss of the granular layer and inflammation.
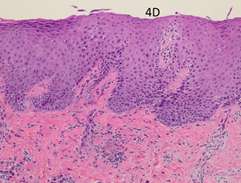
Figure 4D Biopsy taken from the recurrent infield RT area (position A in Figure 3F). Post RT x 100showing that the psoriasiform hyperplasia and inflammation are less marked in this recurrent plaque. The papillary dermal vessels are not as plump, and no neutrophil aggregates are present in the epidermis.
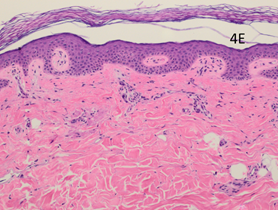
Figure 4E AtPost-RT x 100. Biopsy taken from normal looking infield RT area (position B in Figure 3F). Histopathologically it appears as normal skin.
Figure 4 Comparison of thehistopathology of the pre-RT biopsy of the VMAT-treated field to the biopsy performed at 8 weeks post RT. The addition of VMAT has decreased the severity of the psoriasis when comparing the first and second biopsies. See text for descriptors.
The pre-RT baseline biopsy was compared with the biopsies taken at eight weeks. The classic histological features of psoriasis are shown in the pre-RT biopsy (Figure 4A-C). There is psoriasiform hyperplasia and inflammation. The epidermis is thickened (a finding known as acanthosis), there is a loss of the normal granular layer, there is a thickened stratum corneum (a finding known as hyperkeratosis), and retention of nuclei in the upper layers and stratum corneum (a finding known as parakeratosis). There is suprapapillary thinning. There are abundant neutrophils in the epidermis and stratum corneum. In the dermis, there are abundant mononuclear cells, predominantly myeloid cells and T cells. There are several dilated dermal blood vessels which give the psoriasis plaques their characteristic erythema.3
The post-RT biopsy of the clinically recurrent psoriasis is seen in Figure 4D. This shows the histopathology of the recurrent lesion at eight weeks. The findings of psoriasis are present to a lesser degree than in the pre-RT biopsy. The post-RT biopsy of the clinically normal skin is seen in Figure 4E and shows essentially normal skin whereas, prior to RT, there was florid psoriasis.
We describe a case of an older male with multiple plaques of symptomatic recalcitrant psoriasis. He underwent radiotherapy to seven of the plaques at the same dose using different treatment modalities. The observed outcomes of complete response, partial response and biopsy-proven infield recurrence differed from plaque to plaque (Table 1). A complete response was noted in four of the seven fields. These results raise interesting questions about the radiobiology of psoriasis and whether the administered dose was fortuitously close to the minimum required to control recalcitrant psoriasis.
Field |
Location |
Field |
Modality |
Dose |
RBE Dose |
Infield control |
1 |
Right |
5x3 =15 |
SXRT |
20/10 |
24 |
CR |
2 |
Left |
6x3 = 18 |
SXRT |
20/10 |
24 |
CR |
3 |
Right Temple |
5.5x |
SXRT |
20/10 |
24 |
CR |
4 |
Left Temple |
6x3 =18 |
SXRT |
20/10 |
24 |
CR |
5 |
Right Elbow |
10x6=60 |
SXRT |
20/10 |
24 |
PR |
6 |
Left Elbow |
10x7=70 |
SXRT |
20/10 |
24 |
PR |
7 |
Lower Back |
21x16 |
VMAT |
20/10 |
20 |
Recurrence |
Ave/Total |
|
17.6 for scalp v 65 for elbows |
|
|
|
4/7CR |
Table 1 Field size, treatment modality, dose, and results of the treated plaques at eight weeks
For areas treated with SXRT, all four of the scalps achieved a CR, whereas the best response for elbows was a PR. Was this differential effect between scalp and elbows due to:
When comparing areas treated with SXRT as opposed to those with VMAT, SXRT gave a better result despite superior dose homogeneity with VMAT. Was this due to:
Treatment of psoriasis has traditionally been directed against the immune system with systemic drugs such as cyclosporine, or to the keratinocytes with drugs such as methotrexate. These oral systemic therapies are important treatment options for patients with moderate to severe psoriasis either as monotherapy or, in recalcitrant cases, as combination therapy with phototherapy to localised plaques. Local topical therapies, like steroid cream, can help with acutely symptomatic plaques. Long-term treatment is needed to maintain sufficient disease control in psoriasis, but continuous use of systemic treatments is limited by adverse events and cumulative toxicity.20 The recent advent of biological therapies that target the responsible cytokines have helped. They all induce remission, but not cure.21 These biologicals are administered through regular injections with a course lasting from 6 - 40 weeks.22 Relapse can follow without further on-going treatment.23
Some patients are unable to have, or decline, biological treatments and simply want problematic plaques dealt with for functional and cosmetic reasons. RT has been used for psoriatic plaques but has been mainly limited to problematic anatomical areas including the nails, palms and plantar surfaces, and scalp.7-11
Dose and volume are important considerations with radiotherapy. RT has activity against lymphocytes.24 Cutaneous lymphoma is usually treated with radiotherapy doses in the 24 to 40 Gray (Gy) range.25 RT has activity against benign dividing keratinocytes. The post-operative dose for keloids is 10 to 15 Gy.26 Rosacea has been controlled at 45Gy.27 Doses used for psoriasis, however, have been modest, usually four to six Gy, given as a fractionated course.7-11
In terms of volume, the whole thickened epidermis is the target. A modality is needed that gives a homogeneous and conformal dose of radiation through the epidermis. Most studies, even some randomised trials (RCTs), have used Grenz rays, which have very short penetration into tissue. Even though these studies reported some success, only a small amount of the Grenz rays would have penetrated through the whole thickened epidermis. The treatment modalities used in this case study were superficial radiotherapy (SXRT) and volumetric modulated arc therapy (VMAT; Figure 5). These modalities have a greater ability to give a homogeneous, conformal, and therefore sufficient dose, to the whole thickened epidermis than Grenz rays (Figure 6).
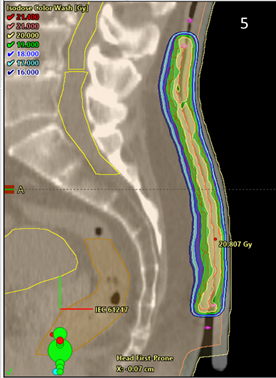
Figure 5 Sagittal planning scan of the lower back area treated with VMAT showing excellent conformality and homogeneity of the dose in dose colour wash mode to the affected epidermis. Note the bolus on the surface of the epidermis.
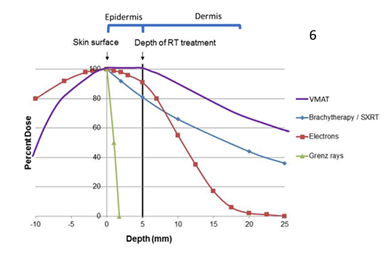
Figure 6 Schematic only. This graph shows the depth doses of different RT modalities through the epidermis. The thin grey vertical line at zero represents the skin surface. The thick black vertical line represents 5 millimetres (mm) into tissue, which is the deepest point that skin appendages penetrate.14 This can be considered as the volume that contains the epidermis. Megavoltage modalities (purple line = volumetric modulated arc therapy [VMAT] and red line = electrons [MeV]) need build up (BU) or “bolus” to ensure full dose to the skin surface.
The finding of complete clearance using VMAT on the back at four weeks followed by recurrence at eight weeks is surprising. VMAT probably gives a more homogeneous and conformal dose than SXRT, especially over larger fields, and therefore it would be expected to be more efficacious. The observed difference in clearance may be due to the relative biological effectiveness (RBE) of the two modalities, which is thought to average around 1.2 for kilovoltage x-rays compared to 1.0 for MV photons.28 This difference in efficacy is because MV primarily deposits energy in tissue by the Compton effect, whereas kV does so by a photoelectric effect. The latter more readily deposits energy in tissue, thus giving more cell kill per Gy.16
This effectiveness is also a function of linear energy transfer (LET). According to Fowler17 in 1981, LET (and RBE) is higher for 250 kVp photons compared with megavoltage photons and electrons. In 2010, Hansen et al18 reported that the RBE of orthovoltage X-rays is 10–15% higher than the RBE of MV, and advised a dose increase of 10–15% when using MV compared to orthovoltage doses. The dose of 20 Gy with SXRT may have been just enough to clear the plaques, but not so with VMAT. Unfortunately, no in-vivo dosimetry was carried out to verify the prescribed dose for any of the treatments which was a substantial oversight.
Another explanation for these findings is that skin may have different radiation sensitivities depending upon its location on the body. This may be related to the amount of chronic sun exposure. The phenomenon of increased radiation sensitivity in skin with chronic sun exposure has been noted when using VMAT for extensive skin field cancerisation. Areas with more chronic sun exposure, for example the scalp, appear to be more radiation sensitive.29
Whatever the cause, it seems that 20Gy in 10 fractions as administered in this case report was very close to the minimum dose required to induce a complete response in recalcitrant psoriatic plaques. The main finding of this case report therefore is that the RT dose needed for complete recalcitrant plaque clearance may be higher than traditionally thought in previous studies, and perhaps even higher than that used in this case report. We suggest that any study of RT for local control of psoriasis should begin with a dose of at least at 20Gy. The treatment modality used should also have adequate penetration to cover all the epidermis, preferably with a homogenous and conformal dose. This is particularly so in psoriasis where the epidermis can be quite thickened.
In the treatment of seven areas of psoriasis in an older fit male, we administered superficial radiation therapy (SXRT; 6 areas) or volumetric modulated arc therapy (VMAT; 1 area) at the same prescribed. At six months post treatment, four scalp areas had a continuing complete response (CR) and both elbow areas had a continuing partial response (PR). A seventh area treated with VMAT achieved a CR at four weeks but at eight weeks there was some infield recurrence. Although the reason for this difference in efficacy is unknown, we propose that it may be due to a lower disease burden, kilovoltage backscatter from bone giving a higher relative dose, a difference in oxygenation or chronic sun exposure of tissues, or a difference in the relative biological effectiveness between SXRT and MV photons. We recommend that future studies of radiotherapy as a treatment for psoriasis start with 20 Gray in 10 fractions using a modality that can penetrate the whole and quite thickened epidermis in this setting.
The authors wish to thank the patient involved for consenting to have his clinical details and photos used. The authors also thank Aileen Eiszele of A&L Medical Communications for editing the manuscript and for overseeing the journal submission process. Recognition and thanks should also be extended to the ever-innovative GenesisCare radiotherapy department at the Mater Hospital, Sydney, Australia. Finally, the authors would like to thank Xstahl Ltd (UK) for funding the medical writer and the cost of open access publication.
The authors have no conflicts of interest to declare.

©2021 Fogarty, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.