International Journal of
eISSN: 2576-4454


Research Article Volume 7 Issue 2
Advanced Polymeric Nanostructured Materials Engineering, Graduate School of Engineering, Toyota Technological Institute, 2-12-1 Hisakata, Tempaku, Nagoya 468 8511, Japan
Correspondence: Prof. Masami Okamoto, Advanced Polymeric Nanostructured Materials Engineering, Graduate School of Engineering, Toyota Technological Institute, 2-12-1 Hisakata, Tempaku, Nagoya 468 8511, Japan
Received: April 28, 2023 | Published: May 10, 2023
Citation: Masashi I, Masami O. Structure and properties of 3D resorbable scaffolds based on poly(L-lactide) via salt-leaching combined with phase separation. Int J Hydro. 2023;7(2):73-76. DOI: 10.15406/ijh.2023.07.00341
In tissue engineering, polymer-based scaffolds play an important role via cell adhesion, proliferation, and tissue regeneration in three-dimensional (3D) structures, exhibiting great potential in a variety of tissues. For fabrication of scaffolds, the particulate-leaching method and thermally-induced phase separation (TIPS) are popular procedures. However, a complete interconnectivity of the porous structure has not been ensured. We have prepared PLLA-based 3D scaffolds with high porosity by the combination of TIPS with NaCl salt-leaching technique. Interconnectivity and cellular infiltration were improved by humidity treatment in the preparation of the scaffolds. By optimization of the parameters of scaffold pore morphologies and cellular penetration, potent 3D resorbable polymeric scaffolds using combinatory method was demonstrated.
Keywords: tissue engineering, poly(L-lactide), polymer-based scaffold, thermally-induced phase separation, NaCl salt-leaching, cellular infiltration, two-dimensional diffusivity
3D, three-dimensional; TIPS, thermally-induced phase separation; PLLA, poly(L-lactide); HA, hydroxyapatite; h+, humidity treatment; D, two-dimensional diffusivity; MC3T3-E1, mouse calvaria preosteoblast cells
In tissue engineering, polymer-based scaffolds play an important role via cell adhesion, proliferation, and tissue regeneration in three-dimensional (3D) structures, exhibiting great potential in a variety of tissues. Porosity, pore size and surface area are well understood as immense parameters of the scaffold-highly engineered structures. Architectural points such as pore shape, interconnectivity between pores and pore wall morphology are also pivotal for cell seeding, growth, migration and mass transport in the formation of the tissue.1
In addition, biodegradable polymers are one of the attractive materials for scaffold. With degradation they are formed leaving nothing foreign to the body in the end. Collagen as natural scaffolds is replaced with ultra-porous scaffolds through biodegradable polymers. The central challenges of the scaffold fabrication depend on the design and customizable biodegradable substitutes having advisable properties. The properties promote cell adhesion and cell porosity along with sufficient mechanical properties which well match the host tissue having cytocompatibility and predictable degradation rate.1,2 The cytocompatibility of the scaffold materials is essential, i.e., the substitute does not show an inflammatory response nor demonstrate immunogenicity in cytotoxicity. For fabrication of scaffolds, the particulate-leaching method and thermally-induced phase separation (TIPS) are popular procedures,2 but a complete interconnectivity of the porous structure has not been ensured. Furthermore, a detailed study regarding homogeneous cell distribution in the scaffolds was not addressed.
This study is aimed to find the correlation between the preparation, morphologies, mechanical properties, and cellular penetration of poly(L-lactide) (PLLA)-based 3D scaffolds fabricated by the combination of NaCl salt-leaching with TIPS technique.3 Parameters including salt contents, concentration of polymer solutions and humidity prior to fusion of salt were examined to elucidate their effect on the porous morphologies. The homogeneous cellular permeability was compared throughout in the scaffold including in the surface layer and in the very center, up to bottom layer. By optimization of these parameters, potent 3D resorbable polymeric scaffolds using combinatory method was demonstrated.
Preparation of scaffolds: The mixtures consisted of poly(L-lactide) (PLLA: Mw= 197 kDa) pellets and a natural hydroxyapatite (HA) crystal powder (particle diameter of ~ 1 mm) (PLLA/HA = 100/0 or 50/50 wt.%) with different concentration (5 and 10 wt.% polymer solution) were dispersed in a solvent (1,4-dioxane) by sonication at 100 W (FU−3H) usually for 30 min to obtain a suspension. 1.0 g of salt particles were added into a polypropylene 24-well plate and subjected at 37°C in an atmosphere of 95% relative humidity for 1.5 h to connect the salt surfaces.4 Prepared suspension of 500 µL were poured on the salt surfaces and maintained in a freezer at -20°C overnight (TIPS). The solidified mixtures in 24-well plate were transferred into freeze-dryer vessel (FDU-2200) under 10 Pa at -80°C for 3 h to remove solvent, then the dried mixtures were taken out from 24-well plate and immersed into ultrapure water to extract the salt particles from scaffolds for 24 h. The prepared samples were designated as x%PLLA-h+ or x%PLLA/HA-h+ (x: polymer solution concentration of 5.0 and 10.0 wt.%, h+: humidity treatment). For a comparison, the scaffolds prepared by the same procedure without humidity treatment of salt particles, salt particles-leaching and freeze-drying methods were prepared. These samples were designated as 5%PLLA, 5%PLLA/HA, 5%PLLA-porogen and 5%PLLA-fd, respectively.
Cell culture: Mouse calvaria preosteoblast cells (MC3T3-E1: ATCC) were cultured in Alpha Minimum Essential Medium (α-MEM: Gibco®, Life Technology) supplemented with 10% fetal bovine serum (FBS: Life Technologies) and 1% antibiotic-antimycotic mixture (Nacalai Tesque) until 80100% confluent in an atmosphere of 5% CO2 and 95% relative humidity at 37°C. Prior to culture initiation, the disc-shaped scaffolds were sterilized at 100°C for 24 h and subsequently with ethanol at room temperature, and then placed into 24-well tissue culture-treated polystyrene plates (TCP).
Cellular infiltration and proliferation into scaffolds: 1 mL of the a-MEM containing MC3T3-E1 was dripped carefully onto the surface of the sterilized scaffolds at a density of 2 x 105 cells/well. The seeded samples were incubated at 37°C for an hour to provide the cells a chance to adhere onto the surface of the scaffolds and up to five days in an atmosphere of 5% CO2 and 95% relative humidity at 37°C. The survivability of the seeded cells was quantified using a Live/Dead Cytotoxicity Kit (Invitrogen) according to manufacturer’s instructions. After staining, the labeled cells were viewed under a fluorescence microscope (EVOS® FL Auto, Life Technologies) with 488 nm excitation and a 525/50 nm emission band pass filter for green fluorescence (Calcein-AM) and 630/75 nm for red fluorescence (EthD-1). To quantify cell permeability at the day 1 point, two cross-sectional slices (very center and in the vicinity of corner along the transverse direction of disc-shaped scaffolds) per scaffolds (n=3) were surveyed. Each slice was divided into at least 6 rectangular (3 mm in width x 1 mm in depth) fields from top surface to the bottom. The numbers of alive (green) and dead (red) cells were counted. The fluorescing cells were analyzed by ImageJ software.
Scaffold structure
For 5%PLLA scaffold, typical macrosize pores (~ 300 mm) are preserved due to the result of salt particles dissolving in water. Owing to the salt particles subject to the humidity, the generation of the additional macropores in the wall could be expected. Then the macropores in the wall could be permeable during the leaching process. However, the significant morphological change is not easily recognized in the field emission scanning electron microscope (FE-SEM) image.
The morphological parameters, density, porosity, and water uptake of the scaffolds are presented in Table 1. The pore size changes more obviously with the loading of HA particles than the pore size fabricated without HA particles. The density of the scaffolds increases with HA loading. The HA loading has no effect on the porosity. The water uptake of the scaffolds is promoted by combinatory method. On the other hand, the porosity does not exactly follow the same trend of water adsorption.
For better understanding, the theoretical values regarding density, porosity, and water uptake are calculated for all scaffolds with different pore morphology. The theoretical values of the designed scaffolds were calculated from the feed composition of the mixture. The measured densities in all scaffolds exhibit higher values than the theoretical values, indicating an insufficient interconnectivity of the porous structures (less than 80% against the theoretical interconnectivity).
The water uptake depends on a hydrophobic polymer like PLLA and hydrophilic nature of HA particles, and the balance between them. However, in this experiment, the interconnectivity of the pores is much affective parameter to change the water uptake capability. The combinatory method leads to the large pore size up to ~ 300 mm (for 5%PLLA and 5%PLLA-h+). The important problem regarding this combinatory method is the structural integrity of the scaffold during the leaching process. Especially at high salt contents (~ 95%) with high porosities (~ 97%) the fabricated scaffolds are prone to fragmentation. In this way, the resultant porous scaffolds are damaged in the leaching process and break into several pieces. As reported by Feijen’s group,5 it is not possible to obtain intact porous structures at salt content over 90%, regardless the size range of the salt particles.
Cellular infiltration
The homogeneous cellular permeability into 3D scaffolds is inevitable property for cell seeding to pursue high quality tissue engineering. The cellular behavior is affected by the 3D scaffold properties, i.e., porosity, pore size, size distribution, and interconnectivity. In this study, cross-sectional slice with two different locations (center and edge of the disc-shaped scaffold) were examined to evaluate the cellular infiltration (Figure 1a). After 24 h of incubation into the scaffolds with different porosity and pore size (Table 1), green fluorescence is observed in each section (Figure 1b–f).
|
Samples |
Pore sizea / µm |
ρscb / g/cm3 |
Φpc/ % |
Water uptaked/% |
Interconnectivitye / % |
x cf/ % |
||||
|
TIPS |
Porogen |
obsd. |
calcd. |
obsd. |
calcd. |
obsd. |
calcd. |
|||
|
5%PLLA |
40 ± 15 |
313 ± 47 |
0.041 ± 0.002 |
0.027 |
96.7 ± 0.2 |
97.9 |
2646 ± 189 |
3610 |
73 ± 5 |
52.6 |
|
5%PLLA/HA |
53 ± 24 |
234 ± 53 |
0.064 ± 0.003 |
0.053 |
96.5 ± 0.1 |
97 |
1268 ± 152 |
1836 |
69 ± 8 |
51.6 |
|
5%PLLA-h+ |
60 ± 21 |
315 ± 67 |
0.040 ± 0.000 |
0.027 |
96.8 ± 0.0 |
97.9 |
2771 ± 136 |
3610 |
77 ± 4 |
55.5 |
|
5%PLLA/HA-h+ |
39 ± 13 |
264 ± 47 |
0.068 ± 0.003 |
0.053 |
96.2 ± 0.2 |
97 |
1538 ± 52 |
1836 |
84 ± 3 |
54.7 |
|
10%PLLA-h+ |
43 ± 27 |
342 ± 52 |
0.067 ± 0.001 |
0.055 |
94.7 ± 0.1 |
95.7 |
1412 ± 103 |
1748 |
81 ± 6 |
60.7 |
|
10%PLLA/HA-h+ |
32 ± 13 |
238 ± 75 |
0.14 7± 0.012 |
0.079 |
91.9 ± 0.7 |
95.6 |
741 ± 51 |
1218 |
61 ± 4 |
53.4 |
|
5%PLLA-porogen |
− |
278 ± 80 |
0.061 ± 0.001 |
0.027 |
95.2 ± 0.1 |
97.9 |
1352 ± 288 |
3610 |
38 ± 8 |
49 |
|
5%PLLA-fd |
116 ± 46 |
− |
0.101 ± 0.001 |
0.052 |
92.0 ± 0.1 |
95.9 |
882 ± 10 |
1845 |
48 ±1 |
60.8 |
Table 1 Morphological parameters, density (ρsc), porosity (Φp), water uptake, interconnectivity and degree of crystallinity (Φp) of PLLA-based scaffolds
aThe cross-sectional pore morphology of 3-D scaffolds was observed through field emission scanning electron microscope (FE-SEM)
bThe mass density of the scaffold was measured by the buoyancy method.
cThe porosity is measured using the equation of Φp= [1− (ρsc/ρb)], where ρb is the mass density of bulk.
dThe water uptake of the scaffold was calculated as following equation: (Ws − Wd)/ Wd. Where Ws is the weight of swollen scaffold and Wd is the weight of dry scaffold.
eThe interconnectivity was calculated by the ratio of water uptake value and its theoretical one.
fThe degree of crystallinity were analyzed using the temperature-modulated differential scanning calorimetry (TA 2920, TA Instruments) at a heating rate of 5
°C/min with a heating/cooling cycle of the modulation period of 60 s and an amplitude of +/-0.796 °C, to determine heat of fusion.
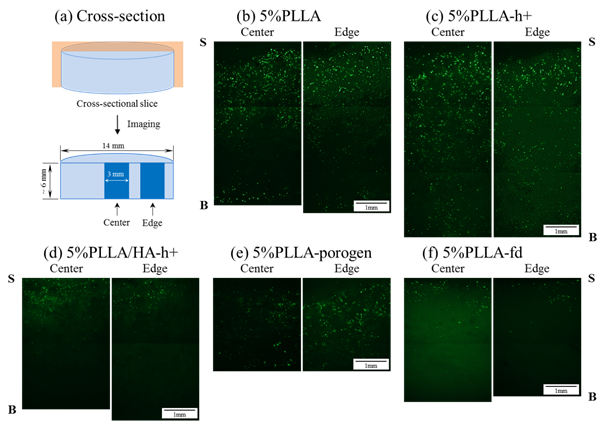
Figure 1 (a) Cross-sectional slice with two different locations, (b)–(f) representative micrographs of cellular infiltration into scaffolds with different porosity and pore size. More green fluorescence is observed at the surface region of both center and edge sections in each scaffold, followed by more diffuse distribution towards the bottom of each scaffold.
The significant cell location is clearly detected at the surface region in each scaffold. As expected, increasing the depth to the bottom of the scaffolds decreases the cell density, indicating the diffusion control is dominant for the cellular penetration. This tendency is almost same as for two different locations, i.e., center and edge of each scaffold. At the same time, the dead cells are hardly observed in the fluorescence, supporting the level of EthD-1 in the image (red fluorescence). Quantitation of percentage cell density shows depth-dependent decrease in the cell density at both locations (Figure 2).
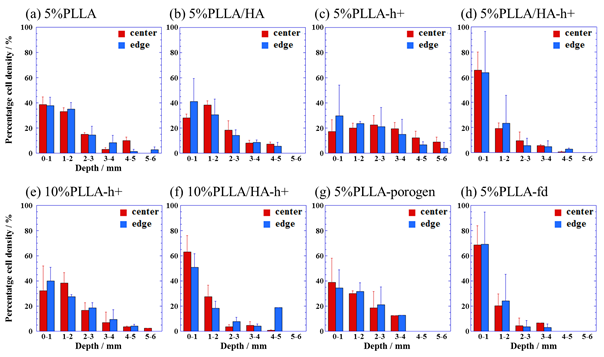
Figure 2 Depth-dependence of cellular infiltration for each scaffold. Results are expressed as mean ±S.D. (standard deviation) (n = 3).
In order to discuss the diffusion of the seeded cells, we present the two-dimensional diffusivity (D) of a cell. According to the time-dependent diffusion equation, as Fick’s second law is given by
(1)
The number of cells per unit area (n(x, t)) at transient space (x) and time (t) is given by
(2)
where N is the total number of the seeded cells and A is the cross-sectional area of infinite length. This D value is useful to discuss the diffusion of the seeded cells in to the scaffolds. The calculated values are presented in Table 2.
|
Samples |
Center |
Edge |
||
|
D a/ nm/s |
R2 b |
D a/ nm/s |
R2 b |
|
|
5%PLLA |
26 |
0.897 |
27 |
0.953 |
|
5%PLLA/HA |
41 |
0.765 |
26 |
0.981 |
|
5%PLLA-h+ |
93 |
0.698 |
56 |
0.962 |
|
5%PLLA/HA-h+ |
8 |
0.975 |
17 |
0.977 |
|
10%PLLA-h+ |
30 |
0.995 |
32 |
0.858 |
|
10%PLLA/HA-h+ |
9 |
0.991 |
10 |
0.718 |
|
5%PLLA-porogen |
35 |
0.992 |
44 |
0.974 |
|
5%PLLA-fd |
7 |
0.985 |
8 |
0.998 |
Table 2 One-dimensional diffusivity (D) of MC3T3-E1 cells seeded into scaffolds with various morphologies
aThe values are obtained using Eq.(2).
bR2 was estimated by the least-squares method of linear regression.
Interestingly, the scaffolds prepared by combinatory method and salt particles-leaching method (5%PLLA-porogen) exhibit large D value one order higher in magnitude compared with that of 5%PLLA-fd. Another interesting feature is that the loading of HA particles has some adverse effect on the diffusivity with a significant decrease in cellular infiltration. This trend is supported by the water uptake behavior as well as rather small pore size created by the salt particles-leaching (Table 1). From the results, optimal pore size for cellular infiltration is found in the scaffolds having large pore size of ~ 200 – 300 µm, which are prepared by using salt particles-leaching method and/or combinatory method. This study supports previous reports that suggested the importance of having pore sizes larger than 300 µm for engineering scaffolds applied to tissue regeneration.6,7
We have prepared PLLA-based 3D scaffolds with different morphology and porosity by the combination of TIPS with NaCl salt-leaching technique. The morphological parameters, density, porosity, and water uptake of the scaffolds were discussed with the correlation of cellular penetration into the scaffolds. The water uptake of the scaffolds was promoted significantly by combinatory method as compare to the scaffolds prepared by salt particles-leaching method and/or freeze-drying. The water uptake depends on the interconnectivity of the pores, but not the loading of HA particles with hydrophilic nature. In this experimental condition, the combinatory method led to the large pore size up to ~ 300 mm (for 5%PLLA and 5%PLLA-h+), at the same time, this method had the problem regarding structural instability for fragmentation in the leaching process.
From an in-depth analysis of cellular infiltration, the diffusion control was dominant for the cellular penetration into the scaffolds prepared by the combinatory method as well as salt particles-leaching. For this reason, there are some limitations to this conventional approach consisted of the combinatory method. These fabricated scaffolds have some properties that may be suitable to support cell in-growth or differentiation. However, the cellular infiltration into the prefabricated scaffolds will eventually lead to the failure of the entire high quality tissue engineered scaffolding system. Hence, more efficient cell seeding techniques are imperative.
This work was supported by the KAKENHI of the Ministry of education, Sports, science and Technology, Japan.
Contributed to the initiating idea and performed most of the experiments: Masashi Ito and Masami Okamoto. Supervised research: Masami Okamoto. The manuscript was written through contributions of all authors.
Author declares that there is no conflicts of interests.

©2023 Masashi, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.