International Journal of
eISSN: 2576-4454


Mini Review Volume 2 Issue 5
1Institute of Geology, University of the Punjab Lahore, Pakistan
2Department of Geology, Abdul Wali Khan University, Mardan Khyber Pakhtunkhwa, Pakistan
Correspondence: Mubashir Mehmood, Institute of Geology, University of the Punjab Lahore, Pakistan
Received: July 13, 2018 | Published: September 18, 2018
Citation: Mehmood M, Yaseen M, Khan EU, et al. Dolomite and dolomitization model - a short review. Int J Hydro. 2018;2(5):549-553. DOI: 10.15406/ijh.2018.02.00124
Worldwide carbonate rocks are occurring abundantly. These carbonate rocks are a major class of sedimentary rocks group. Carbonate are sedimentary rocks formed at (or near) the Earth surface by precipitation from solution at surface temperatures. The two broad categories are limestone, which is composed of calcite or aragonite (different crystal forms of CaCO3) and dolostone, which is composed of the mineral dolomite (Ca Mg (CO3)2). Dolomite is not a simple mineral; it can have a variety of origin, can form as a primary precipitate, a diagenetic replacement, or as a hydrothermal/metamorphic phase, all that it requires is permeability, a mechanism that facilitates fluid flow, and a sufficient supply of magnesium. Dolomite can also form in lakes, on or beneath the shallow seafloor, in zones of brine reflux, and in early to late burial settings. It may form from seawater, from continental waters, from the mixing of basinal brines, the mixing of hypersaline brine with seawater, or the mixing of seawater with meteoric water, or via the cooling of basinal brines. Potential fluid sources are seawater and subsurface fluid of marine and/or meteoric origin: and addition Mg could be released from high-Mg calcite and smectite clays. The only abundant source of Mg2+ ions for early diagenetic surface and near-surface dolomitization is seawater. Dolomitization also creates new crystals, with new rhomb growth following the dissolution of less stable precursors. Dolomitization model and formation depend on the source dolomitization site and lastly, there must exist a favorable condition for a chemical reaction. One particular type of dolomite which may be a cement or a replacement is baroque dolomite, also called 'saddle' or 'white sparry' dolomite and known to mineral collectors as pearl spar. It is characterized by a warped crystal lattice.
Keywords: carbonates, dolomite, calcite, fluid source, dolomitization
Carbonate rocks are a class of sedimentary rocks composed primarily of carbonate minerals. Carbonate are sedimentary rocks formed at (or near) the Earth surface by precipitation from solution at surface temperatures. The two major types are limestone, which is composed of calcite or aragonite (different crystal forms of CaCO3) and dolostone, which is composed of the mineral dolomite (CaMg(CO3)2). Carbonate rocks are depositionally most simple but diagenetically most complex rocks of the world. There are a number of carbonate minerals which are formed by combining of one or more ion with the CO3 2- ion. Dolomite is an anhydrous carbonate mineral composed of calcium magnesium carbonate, ideally CaMg(CO3)2. The term is also used for a sedimentary carbonate rock composed mostly of the mineral dolomite. An alternative name sometimes used for the dolomitic rock type is dolostone. Dolomite is an unusual carbonate mineral. It is common in ancient platform carbonates, yet it is rare in Holocene sediments Table 1.1
Mineral name |
Chemical formula |
Calcite |
Caco3 |
Aragonite |
Caco3 |
Dolomite |
CaMg(co3)2 |
Ankerite |
CaFe(co3)2 |
Magnesite |
Mgco3 |
Siderite |
Feco3 |
Table 1 Some important minerals
On the basis of the mode of formation, dolomites can broadly be divided into two groups: primary dolomite and secondary dolomite.2 Primary dolomite precipitate directly from aqueous solution, mostly at or near room temperature (20-35oC), with no CaCO3 dissolution involved.3 However, dolomite can also form as a secondary phase replacing the precursor mineral calcite (dolomitization process). The widely accepted hypothesis of dolomitization is that limestone is transformed into dolomite by the dissolution of calcite followed by dolomite precipitation.4 That dolomite grows at the expense of calcite was demonstrated by the formation of dolomite on the edges and corners of calcite during dolomitization.5 During dolomitization dissolution of precursor mineral calcite will supply the much-needed Ca+2 wherein Mg+2 (and CO3-2) is supplied by the dolomitizing fluid.6
Formation of dolomite
Dolomite is formed by the replacement of the calcite ions by the magnesium ions. Depending upon the ratio of the Mg ions in the crystal lattice they have different names (Figure 1). Modern dolomite formation has been found to occur under anaerobic conditions in supersaturated saline lagoons in Brazil. In the high-Mg calcite, there is a 0-32 mol% of Mg substitution for Ca. In the protodolomite there is roughly 55-60 Mol% Cain the lattice with incomplete segregation of Ca and Mg into separate layers In the stoichiometric dolomite has a 50:50 of Ca to Mg Ratio with the near perfect ordering of the Mg and Ca in alternate cation layers (Figure 2). Primary dolomite is indeed infrequent and often forms in lakes and lagoons while most of dolomite is from the replacement origin. In the formation of dolomite there are two distinct factor which are the source of Mg+ (Magnesium) what's more, the procedure whereby the dolomitizing liquid is propelled over the carbonate sediments.1 To study the ancient dolomite five wide-ranging classes of dolomitization models are presently existing which are given below:
Every type comprising a unique form of dolomitizing fluid, way of flow and physical settings
Evaporation dolomite
Dolomite is in fact formed in high intertidal supratidal and sabkhas environment. Dolomitic that formed in the supratidal environment are precipitated from evaporated sea water. Early formation of gypsum and aragonite resulting to a sophisticated Mg/Ca proportion of pore water to enhance the formation of dolomite.
Seepage-reflux dolomitization
This process comprises the formation of dolomitizing solutions over vaporization of lagoon water or tidal flat pore water besides then the succession of these solutions into nearby carbonate rocks (Figure 3).
Mixing zone dolomitization
This type of dolomite formed by the mixing of seawater with the fresh water. The source of water may be rainwater (Figure 4).
Burial dolomitization
Burial dolomitization involves prime mechanism which is the dewatering of basinal mud rocks due to compaction and removal of Mg-rich fluids into neighboring shelf edge. Expulsion of water and changes of clay minerals with enlarged burial and increasing temperature suggest that it would liberate Mg ions along with Fe ion. The diagenesis of basinal shales are generally organic-rich and the diagenesis would contribute to CO3 ions.1
Seawater dolomitization
Seawater itself can also be a source of dolomite because it contains the sufficient amount of Mg ions with little modification if a good pumping process exists.
Identification of dolomite
In the lab, it is best to identify by staining it with the Alizarin Red Sulphate Solution.
Result. — The color of calcite will turn into red-brown, whereas dolomite will not be affected. In the field, it gives effervescence with 10% HCl in the powdered form.
Dedolimitization refers to the partial to the whole transformation of former dolomite rocks to a calcian rich rock. The dolomitization can be considered as a kind of process in which Mg is gradually released. The dedolomitization process can be divided into two steps,
Dolomitization is a geological process by which the carbonate mineral dolomite is formed when magnesium ions replace calcium ions in another carbonate mineral, calcite. It is common for this mineral alteration into dolomite to take place due to evaporation of water in the sabkhas area. Dolomitization involves a substantial amount of recrystallization. This process is described by the stoichiometric equation.7
The conditions of dolomite formation are difficult to govern in the laboratory as up to yet no-one has been capable to form it at low temperatures from usual H2O. For undeviating result of dolomite formation the equation is:
For complete dolomitization of a limestone sequence a large quantity of Mg2+ must be propelled over the formation. Beside existence of a favorable environment for chemical reaction, a Mg2+ source and effectual transportation structure are required.
Prospective fluid sources are seawater and underground water of oceanic and/or meteoric source: while addition of magnesium can be added from high magnesium calcite and clays. Sea water is thought to be the prime source of magnesium ion for early diagenetic surface and near-surface dolomitization. It consists 1290 part per million magnesium (0.052 moles I-I) and 411 ppm Ca (0.01 moles I-I), i.e. an Mg/Ca weight ratio of. 3.14 or molar proportion of 5.2. Beside meteoric water generally inconstant possess low values of both; for example, normal river water has 15 ppm Ca and 4 ppm Mg depicting a molar Mg/Ca ratio of 0.44. Therefore ocean water is usually thought to be the origin of the Mg2+ ions for maximum primary dolomitization but this water is generally altered in numerous dolomite forming models in trend.8
Dolomite is one of the complex mineral and its artificial occurrence is difficult to study in lab experimentations. Though it is a very well-ordered mineral and demanding phase to precipitate, yet still dolomite synthesis at sedimentary tempratures using natural waters has not shown. Controlling chemical factors on dolomite formation from water is problematic to clarify and largely have been concluded from high-temperature experimentations9 (Figure 5). Figure 5 Essential elements of any dolomitization model, supply of Mg and the supply of carbonate must be sufficient to form the observed amount of dolomite there must be a delivery mechanism commonly in the form of fluid flow adequate to deliver these ions to the dolomitization site and lastly there must exist favorable condition for chemical reaction. For elucidation of primeval dolomite five extensive divisions of dolomitization model presently exists,
Figure 7 Conceptual models for (a) dolomite precipitation and (b) dolomite replacement scenarios. (a) The thick black lines are the volume fraction of dolomite that is precipitation from the invading solution from the left. The dashed lines represent the dolomite precipitation rates t1 and t2 represent two different time slices. Dolomite precipitation front is moving towards the right as indicated by the open arrow. (b) The replacement front consists of two reaction zones namely zone A and zone B. In Zone A calcite chemically dissolves first releasing Ca+2 in the aqueous solution and producing secondary porosity. The Ca+2 thus released will eventually supersaturate the aqueous solution and precipitate dolomite that will pressure dissolve an equal amount of calcite in zone B. Again t1 and t2 represents two different time slices. Dolomite precipitation front is moving towards the right as indicated by the open arrow.10
Formation of dolomite by replacement of CaCo3 ranges from fabric damaging to retaining and from fabric selective to persistent. The important aspects now are grain mineralogy and crystal extent, the timing of dolomitization and natural surroundings of dolomitizing liquids. Forms of dolomite crystals in replacement assortments differs from anhedral to euhedral shape, with the terminologies xenotopic and idiotopic denoting to the mosaics. Sibley11 put stress on the origin of crystal boundary shapes, identifying non-planar, euhedral and subhedral categories (Figure 8) (Figure 9).
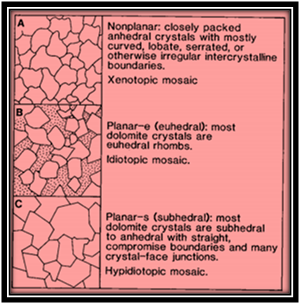
Figure 8 Three common dolomite textures; (A) Non-planar crystals in a xenotopic mosaic; (B) Planar-e crystals (e for euhedral) in an idiotopic mosaic; (C) Planar-s crystals (s for subhedral) in a hypidiotopic mosaic Sibley et al.11
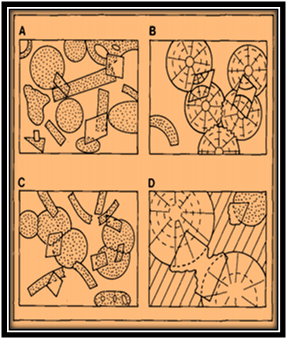
Figure 9 Common dolomite rhomb relationships in limestones; (A) Pre-compaction rhombs, grains in point contact. (B) Compaction after rhomb precipitation so that ghost textures in rhombs are displaced relative to grains; (C)Post-compaction rhombs including fractured grains and stylolites; (D) Pre-sparite rhombs etched and replaced.1
Though dolomite is mainly a replacement, cement of this mineral are common. In Plio-Pleistocene dolomites of the Caribbean-Bahamas, dolomite cement generally formed amongst dolomitized grains and in the spaces. The dolomite can also be found as clear spur, straight analogous to calcite spar, although usually cement is just a single layer of large rhombs in between the spaces of grains. Crystals are too clear and thus they are known as limpid for their white look. Stress has been put on these, proposing they are a distinctive creation of mixing-zone dolomitization. In few cases, dolomite cement comprises regions of syntaxial calcite depicting fluctuation in pore water chemistry (Figure 10).12
This specific dolomite is also known as saddle or white sparry dolomite. Baroque dolomite is known to be forming as a result of replacement. In the minerals collecting community, it is known as pearl spar distinguished by a distorted crystal lattice it has bowed crystal looks, curved cleavage planes, having undulose extinction. Crystal of this dolomite which is larger than a millimeter is generally a compound of sub-crystals, resulting in stepped surface of the crystal. If the dolomite is of replacement origin fluid inclusions, and calcite relics will be present which provides the crystals a gloomy look in thin section and displays shining lustre in hand specimens. Baroque dolomite is calcium rich and usually having Fe content (up to 15 mole % FeCO3. Dissimilarities in configuration of separate crystals are normally distinct, happening amongst dissimilar growing sheets giving a different zonation and inside the regions themselves. This dolomite takes place as together an opening fill and a replacement. In its cavity-fill approach, it typically consumes a xenotropic texture of asymmetrical crystal borders inside the assortment but bowed, scimitar-like ends into the void.13
The objective of this article has been twofold, first to outline and clearly explain the dolomite its types and formation and then to outline clearly the models for dolomitization and to review the different factors associated with dolomite. Dolomite is by formation a very complex which not only forms in the ocean by the simple addition of Mg but also forms as result of Evaporization and burial of magnesium-containing water upon reaction with limestone. Different dolomite types have their own history embedded within it. Baroque a specific category of dolomite which may be cement or a replacement is, also called 'white sparry' dolomite and identified to mineral accumulators as pearl spar. The criteria for choosing between models are not definitive as could be desired and many of these criteria have been interpreted in more than one way. A considerable measure of work and research is anticipating to be completed identified with dolomite, its development and dolomitization process.
None.
The author declares there is no conflict of interest.

©2018 Mehmood, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.