International Journal of
eISSN: 2381-1803


Research Article Volume 16 Issue 2
Department of Zoology, Mizoram University, India
Correspondence: Ganesh Chandra Jagetia, 10 Maharana Pratap Colony, Sector 13, Hiran Magari, Udaipur-313 002, India
Received: March 16, 2023 | Published: March 24, 2023
Citation: Jagetia GC. Antidiabetogenic action of jamun Syzygium cumini Skeels: a review. Int J Complement Alt Med. 2023;16(2):88-97. DOI: 10.15406/ijcam.2023.16.00636
This review delineates the utility of Jamun, Syzygium cumini in the treatment of Diabetes and hyperlipidemia. The review has been written by making thorough internet search on Google Scholar, PubMed, SciFinder, ScienceDirect and many other websites. Diabetes is a common disorder inflicting children and adults equally. Diabetes mellitus is mainly of two types insulin dependent (IDDM-Type I) and non-insulin-dependent Diabetes mellitus (NIDDM, Type II). Type I Diabetes is more common in children, whereas the Type II Diabetes is highly prevalent infecting adults. Jamun or Syzygium cumini belongs to family Myrtaceae and bears sweet fruits that are eaten as such or used to prepare juice, jam, and cookies. Jamun is considered as a Holy tree which grows in India, Bangladesh Ceylon, Myanmar and Pakistan and is used in traditional medicine to treat Diabetes. Jamun contains cardiac glycosides, anthraquinones, catechins, alkaloids, flavonoids, glycosides, steroids, phenols, tannins, and saponins. In Ayurveda, Jamun has been used for the treatment of Diabetes since ancient times. Numerous preclinical and clinical investigations have validated the use of Jamun as an effective treatment regimen against Diabetes. Jamun inhibits different free radicals and increases phase-I enzymes and reduces lipid peroxidation. It also activates PPARα, PPARγ, and AKT, and alleviates the expression of ACC1, Foxo-1, PGC1α, Scid 1, SREPB1c, endoplasmic reticulum protein retention receptor (KEDL), and GPR98 and causes depletion in activities of glucose 6-phosphatase, adenosine deaminase, 5'-nucleotidase, phosphorenolpyruvate carboxykinase, and fatty acid synthase at the molecular level that may be responsible to cure diabetic condition.
Keywords: Diabetes, hyperlipidemia, Jamun, phytochemical, blood sugar
Dysregulated metabolism characterized by hyperglycemia and glucose intolerance leads to Diabetes mellitus. Alteration in the regulation of hormones and defective cellular functions that raise fasting blood glucose concentration higher than 110mg/ dL (6.1mmol) and postprandial glucose concentration of 200mg/dL (11.1 mmol) or more causes Diabetes.1 Diabetic individuals commonly show disrupted homeostasis in hormone regulation which shoots up the level of sugar in the blood. Pancreatic hormone insulin along with glucagon, growth hormone, corticosteroids, and epinephrine tightly regulate glucose level in the blood.2 Diabetes is mainly caused by the malfunction of the pancreas that inhibits insulin secretion, or causes abnormal action of insulin, or both leading to a diabetic condition. The classification of Diabetes depends on the causes and clinical manifestations and is categorized into four types. The insulin-dependent Diabetes mellitus (IDDM, Type I) and non-insulin-dependent Diabetes mellitus (NIDDM, Type II). Gestational Diabetes and other specific types of Diabetes are also found.3,4 IDDM or Type I Diabetes is an autoimmune disorder that occurs due to the damage to β-cells of islets of Langerhans by the body’s T-lymphocytes leading to local inflammation and blockage of insulin secretion and usually requires insulin treatment.2,5,6 Children are more prone to IDDM than adults and have a genetic predisposition to IDDM, and its incidence is on the rise.7,8 The more prevalent Diabetes is Type II, which is caused by increasing resistance to peripheral insulin and diminished pancreatic insulin secretion. The salient features of type II Diabetes include intermediate stages of dysregulated fasting glucose and reduced glucose tolerance, it is also called prediabetes.
Diabetes is the 9th killer disease in the world and causes numerous complications. There are 537 million individuals in the age group of 20 to 79 years who suffered from Diabetes in the world during the year 2021. The number of diabetic individuals may increase to 643 million by 2030 which may further go up to 783 million by the year 2045. Usually, 1 individual died every 5 seconds of Diabetes in the year 2021 taking a total death toll to approximately 6.4 million individuals. 140.9 million diabetic patients are found in China, which is the highest number in the world, next comes India where in the year 2021 approximately 74 million individuals were diabetic. Pakistan has 33 million diabetic patients ranking third in the world. The number of diabetic patients in the USA has been 32.2 million and it ranked fourth in the year 2021 suggesting that Diabetes is one of the important health problems in the world including the USA. Diabetes has become one of the important public health concerns globally and its healthcare cost has been whooping 966 billion USD.9 Modern medicine offers numerous treatment strategies to control both type I and II Diabetes. However, Ayurveda, an ancient system of medicine, also gives a detailed account to treat Diabetes by Jamun and this review will focus mainly on the role of Jamun, Syzygium cumini to cure Diabetes.
Botanical description
Jamun belongs to Kingdom: Plantae; Division: Magnoliophyta; Class: Magnoliopsida; Order: Myrtales; Family: Myrtaceae; Genus: Syzygium and Species: cumini. Jamun is found in the Indian subcontinent including India, Bangladesh, Ceylon, Myanmar, Pakistan, and Madagascar. Jamun grows in various places of the world including the United States of America. Jamun grows well in deep, loamy, well-drained soils.10–12 It is also cultivated in West Indies, Israel, California, and Algeria.13 Jamun becomes a fully grown tree in 40 years and become usually 100 feet (30 m) tall. Jamun canopy expands up to 36 feet (11m) and its trunk diameter reaches up to 2-3 feet (0.6-0.9 m) (Figure 1). Jamun has numerous branches that start shortly from the ground. The stem of Jamun bark is usually cracked, rough, flaking, and its color fades at the lower end (Figure 2). The stem bark becomes smooth and light gray with increasing height. The Jamun leaves are opposite, oblong-oval or elliptical, blunt or tapering to a point at the apex (Figure 3). The size of leaves varies between 2 to 10 inches (5-25cm) in length, and 1 to 4 in (2.5-10cm) in width. The nascent leaves are pink and the mature leaves of Jamun become dark green above, lighter below, glossy, leathery, with a yellowish midrib, and have got turpentine like smell. March-April is the flowering time of Jamun. The Jamun bears flowers in bunches of a few or 10 to 40 in number. The shape of Jamun flowers is round to oblong, 1 to 4 inches long (2.5-10cm) and 1/2 inch (12.7mm) wide, scented, with 0.16 inch (4mm) long funnel-shaped toothed calyx and 4 to 5 petals conjugated as a small disk (Figure 4). Initially, the flowers are greenish-white that turn rose pink eventually.11–14 The Jamun starts fruiting in clusters of a few to 10 or even 40 during summer, which becomes fully ripened in June-July. The fruits of Jamun are oblong or round and their size varies between 1/2 to 2 inches (1.2 to 5cm). The fruits are green initially and turn light to dark purple or even black colored (Figure 5) after ripening.10–15 Jamun fruits are sweetish sour and the tongue becomes purple after eating Jamun. The seeds of Jamun are oblong, whitish purple colored that becomes brown once dried (Figure 6). Hindus and Buddhists regard Jamun as a holy tree and is generally found in the compounds of Hindu temples. Lord Krishna loves Jamun and its leaves and fruits form part of the offering to Lord Ganesha (Elephant God) during worship.11,12

Figure 5 Jamun Syzygium cumini fruits in natural form on the tree (top) and ripened furits (bottom).
Phytochemical analysis
Jamun stem, fruit, leaf, roots, and seeds synthesize different phytochemicals (Table 1). The hexane, petroleum ether, chloroform, ethanol, ethyl acetate, methanol, and water extracts of Jamun leaves have been reported to contain low to high quantities of cardiac glycosides, flavonoids, alkaloids, glycosides, anthraquinones, saponins, phenols, steroids, phytosterols, triterpenoids, tannins, terpenoids, proteins, amino acids, volatile oils, carbohydrates, fixed oils, mucilage, and fats. Jamun contains magnesium, iron, manganese, calcium, copper, zinc, phosphorus, potassium, nitrogen, and sulfur as micronutrients.16–23 Flavonoids, terpenoids, tannins, and carbohydrates have been detected in the ethanol extract of Jamun leaves whereas carbohydrates were detected in the chloroform, ethyl acetate, and methanol extracts.24 Acetone extract of Jamun leaves possessed glycosides, phenols, saponins, proteins, and resins whereas stem bark extract contained alkaloids and flavonoids. All these phytochemicals were detected in the root extract except saponins. The chloroform leaf extract of Jamun showed the presence of alkaloids, steroids, and proteins while the root extract did not show proteins. The alkaloids, tannins, phenols, proteins, and carbohydrates were present in the seed extract, whereas alkaloids and tannins were only found in the stem bark chloroform extract. Methanol extract of the leaves and stem bark showed alkaloids, glycosides, flavonoids, phenols, steroids, saponins, tannins, and carbohydrates, while proteins and resins were additionally present in the root extract. Jamun stem bark n-hexane extract exhibited the presence of alkaloids, tannins, and proteins, and leaf extract in addition contained alkaloids. Resins and alkaloids were found in the root extract and proteins and carbohydrates were present in the seed extract.25 The ethanol extract of Jamun stem bark has been reported to possess alkaloids, catechins, anthraquinone glycosides, phytosterols, phenols, saponins, quinones, steroids, terpenoids, tannins, and amino acids, in addition, flavonoids were present in the methanol extract. The water extract of Jamun stem bark showed catechins, flavonoids, quinones, and phenols.26
|
S.No. |
Parts used |
Extract type |
Phytochemicals |
Reference |
|
1. |
Leaves |
Methanol, ethanol, aqueous, chloroform petroleum ether, acetone, and hexane |
Alkaloids, anthraquinones, flavonoids, glycosides, steroids, phenols, tannins, saponins phenols, triterpenoids, cardiac glycosides, phytosterols, resins |
16-24 |
|
2. |
Stem bark |
Aqueous, ethanol, methanol, hexane and chloroform |
Catechins, phenols, quinones, flavonoids,terpenoids, anthraquinone glycosides, alkaloids, catechins, phenols, quinones, saponins, phytosterols, steroids tannins and amino acids |
|
|
3. |
Seed and fruit pulp
|
Ethyl acetate, methanol. ethanol, and hydroalcoholic
|
Alkaloids, flavonoids, proanthocyanins, tannins, saponins, phenols, cardiac glycosides, terpenoids, phytosterols, steroids, anthocyanins, tannins, proanthocyanidins, amino acids, monosaccharides, reducing sugars |
27-33 |
|
4. |
Root |
Aqueous, and hexane |
Volatile oil, alkaloids, flavonoids, glycosides, saponins, steroids, tannins, terpenoids, carbohydrates and mucilage |
Table 1 Various phytochemicals present in Jamun Syzygium cumini
Methanol and aqueous Jamun seed extracts did contain alkaloids, phenolics, flavonoids, phytosterols, saponins, tannins, and amino acids.27 The ethanol, methanol, and aqueous extracts of Jamun seed have been reported to possess alkaloids, glycosides, flavonoids, phenol, proanthocyanidins, tannins, terpenoids, saponins, steroids, reducing monosaccharides, and reducing sugars. The chloroform seed extract contained flavonoids, phenols, proanthocyanidins tannins, terpenoids, reducing monosaccharides, and reducing sugars. Flavonoids, tannins, phenols, proanthocyanidins, reducing monosaccharides, and reducing sugars have been detected in the diethyl ether Jamun seed extract. The n-hexane Jamun seed extract contained flavonoids and steroids whereas flavonoids and terpenoids have been found in the benzene seed extract.28 Jamun seeds extracted in ethyl acetate and methanol consisted of glycosides, alkaloids, flavonoids, saponins, steroids, triterpenoids, and tannins.29 The alkaloids, cardiac glycosides, proanthocyanins, flavonoids, phenols, phytosterols, saponins, tannins, terpenoids, steroids anthocyanins, and amino acids have been reported from the Jamun seed and fruit pulp ethanol extracts.30–32 Jamun hydroalcoholic seed extract has alkaloids, flavonoids, steroids, and tannins.33 Jamun aqueous root extract possessed glycosides, alkaloids, flavonoids, saponins, steroids, terpenoids, tannins, carbohydrates, mucilage, and volatile oil.22. Some of the active components isolated from Jamun are presented in Figure 7.
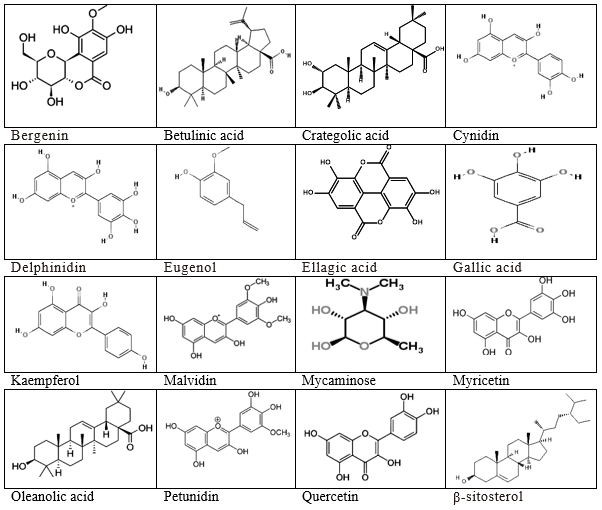
Figure 7 Chemical structures of some important phytochemicals present in different parts of Jamun, Syzygium cumini.
Antidiabetic activity
A sizeable population in the world is inflicted with Diabetes. However, China and India have the highest number of individuals suffering from Diabetes. Although Diabetes was not prevalent in ancient India, the Ayurveda pharmacopeia describes the antidiabetic effect of Jamun seed powder in detail to manage high blood sugar levels. Jamun has been researched for more than 130years for its antidiabetic effect in the Western world however, the clinical trials have been inconclusive as Diabetes was cured in some patients while the response to treatment in others was not encouraging.34 Several investigations have been undertaken to prove the antidiabetic potential of different parts of Jamun in preclinical animal models since the nineteenth century, with mixed results (Table 2). Many studies found that Jamun did decrease blood sugar levels, while some other studies did not find any significant change.11,12,15,34 A 1947 study was unable to detect a depletion in blood sugar levels in the alloxan-induced diabetic rats given Jamun seed extract.35 After that several investigations were carried out in different animal models, that reported a reduction in blood sugar levels by Jamun.12,15 The aqueous Jamun seed extract diminished blood glucose levels in rabbits.36,37 The stem bark extract of Jamun depleted blood glucose in spontaneous diabetic rats.38 Feeding rats with ethanol extract of Jamun bark alleviated glucose level in the blood.39 Various investigations conducted on diabetic rats have conclusively reported an attrition in blood glucose level by aqueous seed powder extract.40–42
|
S.No. |
Parts used |
Extract type |
Species |
Reference |
|
1. |
Seed |
Aqueous, powder |
Rabbits and Rats |
|
|
2. |
|
Aqueous |
Mice |
|
|
3. |
|
Powder |
Humans |
66-69 |
|
|
|
Ethanol, Human |
Rabbits and Rats |
|
|
|
|
Ethyl acetate Methanol |
Rats |
|
|
4. |
Stem |
Ethanol/Methanol |
Rats |
|
|
5. |
Fruit pulp |
Lyophilized |
Rat |
|
|
|
|
Aqueous Ethanol |
Rat |
|
|
6. |
Leaf |
Aqueous/ethanol Aqueous/methanol |
Humans Rats |
|
|
7. |
Root |
Aqueous |
Rats |
Table 2 Effect of different Jamun extracts in the treatment of Diabetes in various study systems
Diabetic rats and mice administered with aqueous seed extract lyophilized powder of Jamun alleviated blood glucose level in the serum.43,44 Alloxan-induced diabetic rats fed with water soluble seed extract with gummy fibers effectively reduced Diabetes. In contrast, the aqueous seed extract devoid of gummy fiber was not effective.45 Administration of different doses of Jamun seed powder in water to streptozotocin-induced diabetic rats led to depletion in fasting blood glucose level.46 The alloxan-induced diabetic rats and rabbits fed with ethanol extract of Jamun seeds/kernel reduced fasting blood glucose levels.47 The ethanol seed kernel extract of Jamun diminished blood glucose, AST, ALT, urea, and cholesterol levels and also raised glucose tolerance in streptozotocin-induced diabetic rats. It also regained the normal levels of reduced glutathione (GSH), superoxide dismutase (SOD), catalase, glutathione peroxidase (GPx) activities in the streptozotocin-induced diabetic rat liver and kidney.48,49 Jamun seed methanol and ethyl acetate extracts and mycaminose (an isolated compound) decreased glucose levels in the blood of streptozotocin-induced diabetic rats.50
The glucose level declined significantly in the serum of alloxan-induced mild and severe diabetic rats administered with the active principles separated on Sephadex gel from the ethanol Jamun seed extract.51 The blood glucose level has been brought to near normal by the water extract of Jamun seeds by elevating peroxisome proliferator-activated receptor gamma (PPARγ) and PPARα proteins expression in the liver of streptozotocin-induced type–II diabetic rats.52 Jamun seeds extracted in methanol depleted serum glucose levels in alloxan-induced diabetic rats.53 The administration of aqueous and ethanol extracts of Jamun fruit pulp in alloxan-induced diabetic rats resulted in a decline in blood glucose level where the aqueous extract was more effective than the ethanol extract.54 A Brazilian study reported that the lyophilized powder of Jamun fruit pulp extract was ineffective in controlling raised blood sugar levels in the serum of the streptozotocin-induced diabetes rats.55 Streptozotocin-induced diabetic female Wistar rats administered with Jamun fruit pulp aqueous extract revealed a reduction in the serum glucose. The concomitant treatment of Jamun fruit extract and stem bark extract of Cinnamon zeylanicum was relatively more efficient than the individual treatment.56
Aqueous and methanol extracts of Jamun root, stem bark, leaf, and seeds lowered glucose in the serum of alloxan-induced diabetic Sprague Dawley rats.57 The administration of an aqueous extract of Jamun seeds in alloxan-induced diabetic mice reduced glucose level in the serum.58 The Jamun fruit pulp/seed/kernel extracted in methanol reduced the activity of α-amylase, and the greatest inhibition of α-amylase activity was recorded for kernel extract in vitro.59 Feeding diet consisting of Jamun fruits and seeds extracted in 50% ethanol for 60 days to diabetic rats led to attrition in serum glucose levels and a rise in insulin levels.60 Attrition in serum glucose levels has been reported in the alloxan-induced diabetic rats administered with different doses of Jamun seed methanol extracts for 14 days.61 The administration of different doses of aqueous Jamun seed extract orally once daily continuously for 21 days to streptozotocin-induced diabetic Wistar albino rats alleviated serum insulin, glucose, TNF-α, and HOMA-IR levels and rise in the PPARγ and PPARα expression in diabetic rat liver. The activities of catalase, GPx, and SOD increased whereas lipid peroxidation and streptozotocin-induced damage to pancreatic β-cells were decreased in the pancreas of rats receiving aqueous Jamun seed extract.62 Single daily administration of triterpenoid-enriched Jamun fruit extract (60mg/mouse) constantly for ten days in the streptozotocin-induced diabetic C57BL/6 mice led to a depletion in the fasting serum glucose and insulin levels and an increase in HOMA-β index. It also increased serine/threonine kinase (AKT, a protein kinase) phosphorylation and p-AKT/AKT expression followed by attrition in the glucose 6-phosphatase (G6Pase), phosphoenolpyruvate carboxykinase (PEPCK), peroxisome proliferator activated receptor gamma coactivator 1-alpha (PGC1α), and Forkhead box protein-1 (Foxo-1) in the streptozotocin-induced diabetic mouse liver.63 Feeding of 200 and 400 mg/kg body weight aqueous Jamun root extract or isolated compound 4-(2-amino-2-(2-(2-hydroxy-3- methyl butyl)octahydropyrrolo.12-a pyrazin-7-yl) ethyl)-2-ethylphenol for 15 days once daily depleted serum glucose level in streptozotocin-induced diabetic rats.22
The diabetic patients given Jamun aqueous leaf extract showed diminished activity of adenosine deaminase (ADA) and serum glucose level.64 The ethanol (80%) Jamun leaf extract reduced 5'-nucleotidase (5’NTase) and ADA activities, triglycerides, glucose, and lipid peroxidation levels, accompanied by increased catalase activity in diabetic patients.65 The diabetic patients given Jamun seed powder daily for 30days revealed alleviated fasting and postprandial blood glucose levels.66 The 99 diabetic patients given 5 grams of seed powder twice daily for 90days before meals reduced decreased serum glucose levels in a randomized double-blind placebo-controlled clinical study.67 Type-II Diabetes patients given 2 g of roasted Jamun seed powder for sixty days showed a decline in serum glucose levels.68 An another double-blind randomized clinical trial undertaken between March 2018- March 2019 showed almost similar effects.69 The antidiabetic activity of n-hexane, ethyl acetate, and ethanol extracts of Jamun pulp and seed was evaluated in vitro by evaluating the α-amylase and α-glucosidase activities. The IC₅₀ value of 75.85 and 74.72mg/ml for α-amylase and 55.79 and 59.85mg/ml for α-glucosidase activity have been reported indicating the ability of Jamun to treat Diabetes.70 The above listed studies demonstrate the potential of Jamun to cure Diabetes.
Antihyperlipidemic activity
Cardiovascular morbidity is a consequence of hyperlipidemia caused by Diabetes and different parts of Jamun are known to decrease cholesterol level in the serum. Attrition in the serum low density lipoprotein cholesterol (LDL-c) level, total cholesterol/high density lipoprotein cholesterol (HDL-c) ratio and depression in HMG-CoA reductase activity have been found in the alloxan-induced diabetic rabbits treated with Jamun seed ethanol extract.47 The Jamun seed kernel ethanol extract decreased serum LDL-c and very low density lipoprotein cholesterol (VLDL-c) and raised the HDL-c levels in streptozotocin-induced diabetic rats.49 Two grams of Jamun seed powder given to diabetic patients for 60days alleviated serum cholesterol, LDL, VLDL-c, and triglycerides.68 A depletion in triglycerides, total cholesterol, and an increase in HDL-c has been reported in the alloxan-induced diabetic rats administered with active principles removed from the fraction of Jamun ethanol seed extract by Sephadex gel.51 Jamun seed water extract prevented hyperlipidemia by bringing the total cholesterol and triglycerides levels to normal and elevating the HDL-c in the alloxan-induced diabetic mice.58 The feeding of high cholesterol diet and Jamun seed and fruit ethanol extract to rats diminished triglycerides, and LDL-c and increased the HDL-c.71 Administration of fruit pulp extract of Jamun to streptozotocin-induced diabetic rats caused a similar effect.56 The administration of triterpenoid-rich Jamun fruit extract (60 mg/mouse) once daily for ten days to streptozotocin-induced diabetic C57BL/6 mice resulted in a decline in the serum free fatty acid (FFA) contents and triglyceride and also in the liver of diabetic mice. This was accompanied by attrition in the mRNA of stearoyl-CoA desaturase-1 (Scd1), acetyl-CoA carboxylase 1 (ACC1), fatty acid synthase (Fas) and cluster of differentiation 36 (CD36), and expression ACC1 protein. An elevated expression of p-ACC1 protein and normalization of extracellular signal-regulated kinase (Erk1/2) and p-Erk1/2 expression has been recorded in diabetic mice.63 Intermediate hyperglycemic patients fed with 4.5g Jamun seed powder capsules once daily for 84 days showed significantly reduced cholesterol and LDL-c levels.72
Mechanism of action
The action of Jamun in reducing blood sugar and hyperlipidemia may not be due to a single mechanism and several putative mechanisms in concert with each other seem to be involved in controlling Diabetes and hyperlipidemia. The excess formation of free radicals/reactive oxygen species (ROS) is one of the most important events in causing Diabetes,73,74 therefore, the passivation of free radicals by Jamun seems to be one of the crucial mechanisms of action in controlling Diabetes.75–77 At the molecular level reduction in glucose level may be attributed to the ability of Jamun to activate PPARα, PPARγ, and AKT, and subsequently downregulate the expression of ACC1, Foxo-1, PGC1α, Scid 1, SREPB1c, endoplasmic reticulum protein retention receptor (KEDL), and GPR98 that cause depletion in G6Pase, ADA, 5’NTase, PEPCK, and Fas, activities (Figure 8).62–64,66 The (Nuclear factor E2-related factor 2) Nrf2/Keap1/ARE (Antioxidant Response Element) signaling pathway dysregulation has been detected in Diabetes78 and Jamun seems to sequestrate Nrf2/Keap1 and translocates Nrf2 into the nucleus thus restoring this pathway to normal. The elevation of Nrf2 and its translocation into the nucleus activates ARE that stimulates heme oxygenase-1 (HO1) and NAD[P]H: quinone oxidoreductase-1 (NQO1), which subsequently leads to a rise in the antioxidants including GPx, GSH, glutathione reductase (GR), SOD, catalase, glutathione-s-transferase (GST) in diabetic condition followed by reduced lipid peroxidation (Figure 9).79–81 Several other unknown mechanisms may be utilized by Jamun to bring about antidiabetic action.

Figure 8 Jamun scavenges free radicals (reactive oxygen species ROS) that downmodulate TNF-α and upregulate protein kinase (AKT), peroxisome proliferator activated receptor (PPARγ) and PPARα that downregulates Forkhead box protein-1 (Foxo-1), peroxisome proliferator activated receptor gamma coactivator 1-alpha (PGC1α), (SREBP1c), Acetyl-CoA carboxylase (ACC1), stearoyl-CoA desaturase-1 (ACC1), endoplasmic reticulum protein retention receptor (KEDL), G protein-coupled receptor (GPR98) fatty acid synthase (Fas), sterol regulatory element-binding protein glucose 6-phosphatase (G6Pase), and phosphoenolpyruvate carboxykinase (PEPCK), Adenosine deaminase (ADA)and 5'-nucleotidase (5’NTase).
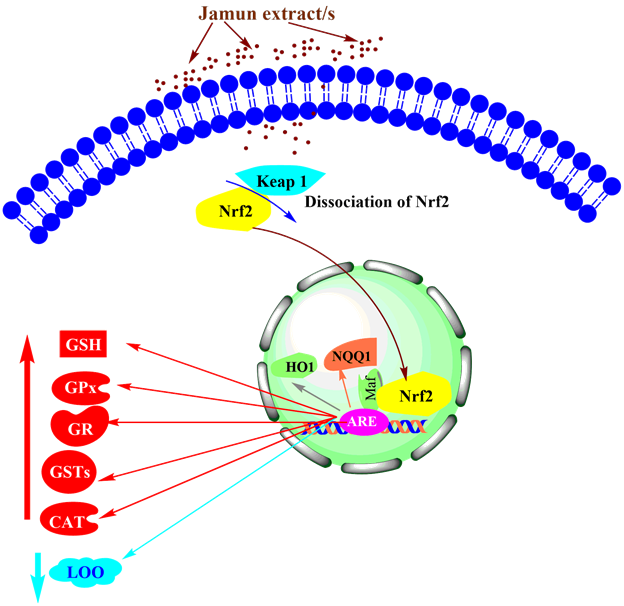
Figure 9 The antidiabetic action of Jamun is due to its ability to dissociate (nuclear factor E2-related factor 2) Nrf2/Keap1 that translocates Nrf2 into the nucleus. Once Nrf2 is in the nucleus it activates antioxidant response element (ARE), heme oxygenase-1 (HO1) and NAD[P]H: quinone oxidoreductase-1 (NQO1) which increases glutathione (GSH), glutathione peroxidase (GPx), glutathione reductase (GR), glutathione-s-transferase (GST) and catalase (CAT) and reduces lipid peroxidation (LOO).
Jamun is native to South Asia and reaches a height of 100 feet. In traditional medicine, Jamun finds its application in the treatment of different diseases including Diabetes. The ability of Jamun to synthesize numerous phytochemicals seems to be mainly responsible for its antidiabetic and hypolipidemic action. The attrition in 5’NTase, ADA, PEPCK, Fas, G6Pase, and glucose concentration seems to be due to a rise in the gene expression of AKT, PPARα, and PPARγ and a decline in the expression of GPR98, Foxo-1, ACC1, PGC1α, SREPB1c, Scid 1, and KEDL in diabetic condition. The augmented levels of GPx, GSH, catalase, GST, and SOD, and the decline in LOO seems to be due to the activation of the Nrf2 signaling pathway by Jamun in diabetic conditions. It will be purposeful to undertake future studies to unfurl molecular mechanisms of Jamun in vitro and in vivo in diabetic conditions.
My sincere thanks go to my wife Mrs. Mangla Jagetia for her unstinted support during the writing of this manuscript.
We declare there are no conflicts of interest.
The financial assistance from the University Grant’s Commission, New Delhi, India vide grant No. F4-10/2010(BSR) is thankfully acknowledged.

©2023 Jagetia. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.