eISSN: 2469-2778


Short Communication Volume 11 Issue 3
Department of Histopathologist, Punjab University Chandigarh, India
Correspondence: Anubha Bajaj, Department of Histopathologist, Punjab University Chandigarh, India
Received: June 25, 2023 | Published: July 12, 2023
Citation: Bajaj A. The convoluted conduit-striated duct adenoma. Hematol Transfus Int. 2023;11(3):73-74. DOI: 10.15406/htij.2023.11.00307
Striated duct adenoma is an exceptionally encountered, benign salivary gland neoplasm. Tumefaction is constituted of ductal articulations coated with mono-layered epithelial cells and a cytological resemblance to normal striated ducts. Striated duct adenoma may recapitulates normal configuration of salivary ducts. Striated duct adenoma was initially scripted by Dardick wherein the neoplasm was designated as ‘ductal adenomas with no visible myoepithelial cells’ in 1996.
Keywords: adenoma, myoepithelial cells, carcinoma, tumor
Contemporary World Health Organization (WHO) classification of benign salivary gland tumours denominates striated duct adenoma within a category pertaining to canalicular adenoma and various ductal adenomas. In contrast to intercalated duct adenoma, striated duct adenoma appears devoid of myoepithelial cells or basal cells. Upon immunohistochemistry, striated duct adenoma appears to lack a distinct myoepithelial cell layer. The benign striated duct adenoma appears immune non reactive to various myoepithelial markers, in contrast to diverse malignant salivary gland neoplasms exemplifying immune non reactivity to myoepithelial markers, indicative of malignant metamorphosis. Striated duct adenoma is associated with decimated incidence, possibly as ascertainment of the infrequently discerned neoplasm is challenging of striated duct origin, tumefaction is devoid of myoepithelial cell differentiation. Upon cogent immunohistochemistry, myoepithelial immune markers appear decimated or absent. However, upon extensive and varied adoption of immunohistochemistry, the neoplasm denominates possible striated duct differentiation.1,2 The infrequent, benign, striated duct adenoma of salivary gland commonly occurs within fifth decade to eighth decade. The encapsulated, well circumscribed neoplasm is encountered within major and minor salivary glands.2,3
Striated duct adenoma may represent as a tumefaction of extended duration confined to the cheek. Parotid gland is commonly incriminated. Grossly, incriminated salivary gland exhibits a solid, well circumscribed, grey/white tumefaction of variable magnitude. Upon cutting open, tumour surface is preponderantly encapsulated and demonstrates a variegated appearance with myxoid areas.2,3 Upon microscopy, a well circumscribed tumefaction composed of intensely articulated ductal configurations appears segregated by scant, intervening, fibrotic stroma. Few ducts exhibit ductal dilatation and permeation with colloid-like intraluminal content.3,4 Morphologically, tumefaction is composed of intensely adherent ducts delineating minimal or absent intervening stroma. Few ducts may exemplify cystic dilation up to 0.1 centimetres.3,4
Neoplastic cells are pervaded with abundant, eosinophilic cytoplasm with prominent cellular membranes, reminiscent of striations encountered within normal striated ducts. Individual tumour cells are impregnated with eosinophilic cytoplasm, bland nucleus, prominent cellular membrane and intracytoplasmic striations. Occasionally, neoplastic cells appear permeated with nuclear grooves and intra-nuclear pseudo-inclusions, simulating nuclear features of papillary thyroid carcinoma.3,4 Histological appearance of striated duct adenoma depicts several ductal structures layered by singular coat of epithelial cells, reminiscent of striated salivary gland ducts. Infrequently, tumour cells appear pervaded with nuclear grooves, intra-nuclear pseudo-inclusions and mild clearing of nuclear chromatin, simulating nuclei of papillary thyroid carcinoma. Prominent capillaries, vascular articulations and red blood cell extravasation are commingled with ductular configurations. Mitotic figures or tumour associated necrosis is absent (Figure 1 & 2) (Table 1).3,4
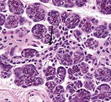
Figure 1 Striated duct adenoma composed of intensely adherent ductal structures encompassed by scant stroma. Ducts are layered by tumour cells imbued with abundant eosinophilic cytoplasm, bland nuclei and intracytoplasmic striations.7
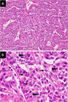
Figure 2 Striated duct adenoma exhibiting closely aggregated ductal structures lined by neoplastic epithelial cells incorporated with abundant eosinophilic cytoplasm, bland nuclei and intracytoplasmic striations. Encompassing stroma is scant and fibrotic.8
|
Low grade |
Intermediate grade |
High grade |
Variable grade |
|
Acinic cell carcinoma |
Myoepithelial carcinoma |
Salivary duct carcinoma |
Mucoepidermoid carcinoma |
|
Basal cell adenocarcinoma |
Sebaceous adenocarcinoma |
Squamous cell carcinoma |
Adenoid cystic carcinoma |
|
Epithelial-myoepithelial carcinoma |
Lymphoepithelial carcinoma |
Small cell carcinoma |
Salivary carcinoma NOS |
|
Secretory carcinoma |
Large cell neuroendocrine carcinoma |
Intraductal carcinoma |
|
|
Polymorphous adenocarcinoma |
Large cell undifferentiated carcinoma |
Carcinoma ex pleomorphic adenoma |
|
|
Hyalinising clear cell carcinoma |
Carcinosarcoma |
||
|
Mucinous adenocarcinoma |
Salivary gland carcinomas with high grade transformation |
||
|
Micro-secretory adenocarcinoma |
|||
|
Sclerosing microcystic adenocarcinoma |
|||
|
Sialoblastoma |
|||
|
Metastasizing pleomorphic adenoma |
|||
Table 1 Histopathological stratification of salivary gland malignancies3
Neoplastic cells of striated duct adenoma are intensely and diffusely immune reactive to S100 protein, CK5 or CK7 whereas singular cells may appear immune reactive to p63.4,5 Tumour cells appear immune non reactive to markers indicating basal cells or myoepithelial cells as alpha smooth muscle actin(α-SMA), calponin, p63 or glial fibrillary acidic protein (GFAP). Striated duct adenoma is immune non reactive to thyroid transcription factor (TTF-1). MIB-1 proliferative index is minimal at ~2%.4,5 Contingent to histological features, striated duct adenoma requires segregation from neoplasms demonstrating a ductal architecture and preponderantly composed of tumour cells incorporated with abundant, eosinophilic cytoplasm as oncocytoma, intercalated duct adenoma, basal cell adenoma or canalicular adenoma.5,6 Besides, demarcation of striated duct adenoma from low grade, malignant neoplasms as polymorphous adenocarcinoma (PAC) or oncocytic epithelial-myoepithelial carcinoma upon examination of miniature tissue samples may be challenging, especially in neoplasms where assessment of tumour periphery is problematic.5,6 Striated duct adenoma can appropriately be managed with comprehensive surgical excision. The essentially benign tumefaction is devoid of associated facial nerve palsy or regional lymph node enlargement or metastasis. Following cogent surgical intervention, emergence of residual tumour appears absent.5,6
Striated duct adenoma is an infrequent, benign salivary gland neoplasm. Parotid gland is commonly incriminated. Tumefaction is constituted of ductal articulations coated with mono-layered epithelial cells and a cytological resemblance to normal striated ducts. The well circumscribed tumefaction is composed of intensely adherent ductal configurations segregated by scant, intervening, fibrotic stroma. Tumour cells are impregnated with eosinophilic cytoplasm, bland nucleus, prominent cellular membrane and intracytoplasmic striations. Upon immunohistochemistry, striated duct adenoma appears to lack a distinct myoepithelial cell layer. Striated duct adenoma is diffusely immune reactive to S100 protein, CK5 or CK7. Striated duct adenoma requires segregation from neoplasms such as oncocytoma, intercalated duct adenoma, basal cell adenoma or canalicular adenoma. Striated duct adenoma can appropriately be managed with comprehensive surgical excision.
None.
The author declares that there is no conflict of interest.
None.

©2023 Bajaj. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.