eISSN: 2469-2778


Research Article Volume 10 Issue 3
1Clinical hematology and BMT department, Al Maadi military medical compound, Egypt
2Clinical Hematology Unit, Internal Medicine Department, Faculty of Medicine, Oncology Center, Mansoura University, Egypt
Correspondence: Mohamed Mabed, The Hematology Unit, Oncology Center, Faculty of Medicine, Mansoura University, Egypt
Received: August 07, 2022 | Published: August 30, 2022
Citation: Elshamy A, Elmohsen EA, Denewer M, et al. Post-transplantation cyclophosphamide as graft versus host disease prophylaxis in patients with acute leukemia received fully matched allogeneic HSCT or haplo-identical HSCT. Hematol Transfus Int. 2022;10(3):78-84. DOI: 10.15406/htij.2022.10.00285
Post-transplant Cyclophosphamide (PT-Cy) has proved efficacy as GVHD prophylaxis regimen after HSCT. However, experiences are limited with controversial results. We herein, assess the efficacy of PT-Cy compared to Methotrexate regimen. Eighty patients with acute leukemia received a fully matched allogeneic HSCT or a Haplo-identical HSCT were analyzed. Group I (Historical group) included 40 patients received Methotrexate, Cyclosporine and MMF. They were transplanted with fully matched allogeneic transplantation. Group II included 40 patients received PT-Cy, Cyclosporine and MMF. They were subdivided to 2 subgroups. Subgroup IIA included 22 patients received fully matched allo- HSCT and subgroup IIB included 18 patients received Haplo- HSCT. Group IIA showed significantly lower incidence of cGVHD when compared to group I with an incidence of 22.7% and 67% respectively (P = 0.002). Group IIA was associated with reduced risk of extensive cGVHD compared to MTX group (P =0.003). No significant differences were found in the incidence of aGVHD, relapse rates, relapse or non-relapse related mortalities, OS and DFS data among the different groups (P>0.05). In conclusion, PT-Cy with addition of IS drugs has statistically significant difference in reducing the incidence of cGvHD in both fully matched and Haplo SCT with less hepatic and renal toxicity.
Keywords: acute graft-versus-host-disease, acute leukemia, chronic graft-versus-host-disease, cyclophosphamide, immunosuppressives, prophylaxis
Cyclophosphamide is an alkylating agent metabolized by the cytochrome P450 in the liver into two metabolites, phosphoramide mustard and acrolein that prevents cellular division by crosslinking DNA strands. The alkylating effect of the active metabolites is most evident during the G1 and S phases of cell division.1 The rapidly proliferating cells subjected to DNA crosslinking are more susceptible to cyclophosphamide due to their reduced ability to renew damaged DNA. The hematopoietic stem cells are resistant to the effect of cyclophosphamide as they are rich in aldehyde dehydrogenase, the enzyme required for the conversion of phosphoramide mustard into the inactive metabolite carboxycyclophosphamide. Consequently, cyclophosphamide can be administered after allogeneic HSCT targeting Proliferating T- cells without affecting engraftment.2 The mechanism for induction of tolerance by post-transplant cyclophosphamide (PT-Cy) can be explained by three steps scheme. Early, the multiplying alloreactive donor and recipient T -cells are destroyed. Next, the increased T-regs (because T-regs have aldehyde dehydrogenase which cause drug efflux) counter balance the effect of any remaining alloreactive mechanisms. Finally, the intrathymic clonal deletion that is delayed but long-lasting and permit anti-host T cells that maintains long term tolerance.3
Adapted GvHD prophylaxis has been proposed with the increased use of HSCT from un-manipulated haploidentical donor.4 Among those Luznik et al,5 pioneered the use of high-dose PT-Cy with other immunosuppressives (IS) and reported a low rate of acute and chronic GVHD with low rates of transplant-related mortality.
The feasibility of PT-Cy in the haploidentical setting paved the way for its use for GVHD prophylaxis in recipients of HSCT from matched sibling donors (MSD) or matched unrelated donors (MUD).6 Luznik et al,7 reported 43 and 10% of grades II–IV and III–IV aGvHD, respectively, and 10% of cGvHD in 117 patients receiving bone marrow (BM) transplantation from MSD with myeloablative regimen. However, the administration of PT-Cy alone in the setting of HSCT with peripheral blood stem cell (PBSC) grafts from either HLA identical sibling or unrelated donors, was associated with severe aGVHD and related deaths.8 Ruggeri et al, analyzed PT-Cy alone or in combination with other IS as GvHD prophylaxis in a large cohort of patients transplanted for acute myeloid leukemia (AML) or acute lymphoblastic leukemia (ALL). The study showed that the addition of IS drugs to PT-Cy enhances its effect and reduces the risk of severe cGvHD, reducing mortality and improving survival.9 Taken together, these clinical data suggest that PT-Cy has been successfully translated from basic and preclinical models into clinical practice. The data to date have been sufficient to encourage further exploration and development of this approach in further trials.3,10
We herein, assess the efficacy and compare the complications of PT-Cy as GvHD prophylaxis compared to Methotrexate regimen in the setting of fully matched sibling donor transplantation and haplo-identical transplantation.
Patients
In a matched paired controlled study, 80 patients with acute leukemia were recruited from Al Maadi Military Hospital in Egypt. The study was approved by the Institutional Research Board (IRB) of Mansoura Faculty of Medicine with an approval number MS.18.08.236. They received either a fully matched allogeneic HSCT or a Haplo-identical HSCT. According to the regimen of GvHD prophylaxis they were divided into 2 groups:
Group I (Historical group): included 40 patients received, Methotrexate: D+2 (15 mg/m2), D+4 (10 mg/m2), D+6 (10 mg/m2), Cyclosporine: 5mg/kg from day -3 till day +90 and MMF: 2000 mg – 3000 mg /day from day +1 till day +30 as GvHD prophylaxis. They were transplanted with fully matched allogeneic transplantation and were preconditioned with Fludarabine 30 mg/M2/ day from day -6 to day -2, Melphalan 140 mg/ M2 from day -4 to day -2 and Etoposide 30 mg/kg/day from day -6 to day-2.
Group II: included 40 patients received Post allogeneic stem cell High Dose Cyclophosphamide: (50 mg /kg/day) at day +3 and day +4; Cyclosporine: 5mg/kg from day +5 till day +30 and MMF: 2000 mg – 3000 mg /day from day +5 till day +30 as GvHD prophylaxis. They were divided into 2 subgroups according to the type of transplant.
Subgroup A: included 22 patients received fully matched allo- HSCT and preconditioned with Fludarabine 30 mg/M2/ day from day -6 to day -2; Melphalan 140 mg/ M2 from day -4 to day -2 and Etoposide 30 mg/kg/day from day -6 to day -2.
Subgroup B: included 18 patients received Haplo- HSCT and preconditioned with Fludarabine 30 mg/M2/ day from day -6 to day -2 from stem cell infusion (day zero); Melphalan 140 mg/ M2 from day - 4 to day -2 and Cyclophosphamide 14.5 mg/kg from day -6 to day -5.
Assessment of complications
GvHD was diagnosed and classified according to EBMT−NIH−CIBMTR Task Force position statement, 2018.12 Evaluation of nephrotoxicity was done through measurements of blood urea nitrogen (BUN), serum creatinine, glomerular filtration rate and creatinine clearance. Evaluation of Hepatotoxicity done through elevation of liver enzymes and serum bilirubin. Evaluation of SOS/VOD was done according to the European Society for Blood and Marrow Transplantation (EBMT) classification.13 Hemorrhagic cystitis was graded as: grade 0 = none; grade I = microscopic hematuria with urinary symptoms; grade II = macroscopic hematuria; grade III = macroscopic hematuria with clots and grade IV = macroscopic hematuria with intervention for clot evacuation and/or urinary retention.14
Statistical analysis
Data were analyzed with the use of IBM SPSS software version 21. The qualitative data were described as numbers and percentages. The continuous variables were presented as ranges (minimum, maximum) and medians for quantitative parametric data and categorical data were presented as counts and percentages. The statistical significance of the difference between the means of two study groups was assessed by the student t test. The Chi-Square test was used to examine the relationship between two qualitative variables. The Fisher’s exact test was used to examine the relationship between two qualitative variables when the expected count is less than 5 in more than 20% of cells. To assess the strength of association between two quantitative variables a correlation analysis was done. Logistic regression analyses were used for prediction of risk factors, using generalized linear models. Cox regression analysis of factors potentially related to survival was performed to identify which independent factors might jointly have a significant influence on survival. The overall survival was defined as the time from diagnosis until death from any cause or end of the study. DFS was defined as the time from remission until disease relapse, death from any cause or end of the study. Kaplan–Meier test was used for survival analysis and the statistical significance of differences among curves was determined by Log-Rank test. All the tests were 2-sided and a P-value < 0.05 was considered statistically significant.
The demographics and the engraftment data (Table 1)
The three groups were matched for age and gender. The diagnosis of post-transplant MTX (group I) included 25 AML patients (67.5%), 13 ALL patients (32.5%) and 2 biphenotypic patients (5%). Those with PT-Cy (group IIA) were 18 AML cases (81.8%), 4 ALL cases (18.2%). Haplo-identical HLA stem cell transplant with PT-Cy (group IIB) patients included 14 AML (77.8%), 3 ALL (16.7%) and 1 biphenotypic (5.6%). No significant differences were found in the diagnosis among the three groups. No significant differences were found in stem cell dose among the studied groups (p>0.05). Neutrophil and platelet recovery time were significantly prolonged in PT-Cy groups received fully matched transplant compared to MTX group (P=0.002, 0.024 respectively). No significant differences were found in Neutrophils and platelets recovery between fully matched and haploidentical HLA typing groups (P>0.05). No significant differences were found regarding chimerism assessment at D +30 among the studied groups (P>0.05).
Post-transplant Cyclophosphamide |
|||||
Post-transplant methotrexate (group I) |
Fully matched HLA typing (group IIA) |
Haploidentical HLA typing (group IIB) |
P1 |
P2 |
|
N=40 |
N=22 |
N=18 |
|||
Age (years) Mean ± SD |
28.3±8.5 |
28.2 ± 8 |
24.8 ± 6.1 |
0.9 |
0.28 |
Male N (%) |
26 (65%) |
13 (59.1%) |
14 (77.8%) |
0.65 |
0.21 |
Female N (%) |
14 (35%) |
9 (40.9%) |
4 (22.2%) |
||
Diagnosis N (%): |
|||||
AML |
25 (62.5%) |
18(81.8%) |
14 (77.8%) |
0.23 |
0.83 |
ALL |
13(32.5%) |
4 (18.2%) |
3 (16.7%) |
||
Biphenotypic |
2 (5.0%) |
0 (0%) |
1 (5.6%) |
||
Stemcell dose(X106/Kg) Mean ± SD |
6 ± 0.7 |
6.5 ± 2.3 |
6.5 ± 1.3 |
0.07 |
0.98 |
Engraftment data: |
|||||
Neutrophil recovery mean±SD days |
12.7±1.9 |
14.6±2.6 |
14.4±3.2 |
0.002 |
0.89 |
Platelet recovery mean±SD days |
12.3±1.9 |
14.6±6.2 |
14.2±4.3 |
0.024 |
0.84 |
Full chimerism at day 30 N (%) |
35 (87.5%) |
19 (86.4%) |
17 (94.4%) |
0.659 |
0.61 |
Table 1 The patient’s characteristics and demographics
P1: comparison between group I and group IIA; p2: comparison between group IIA and group IIB. SD: standard deviation.
The transplant related morbidities and the Graft-versus-Host-Disease incidence rates (Table 2)
Group IIA showed significantly more frequent central venous line (CVL) culture growth and fungal mucositis compared to group I. However, it showed significantly less frequent blood culture growth when compared to group IIB. No significant differences were found in CMV reactivation among the studied groups (P>0.05). No significant renal or hepatic toxicities were seen among the studied groups. Post-transplant cyclophosphamide groups (group IIA and IIB) were significantly associated with hemorrhagic cystitis when compared to post-transplant methotrexate group (P=0.006; 0.006 respectively).
Post-transplant Cyclophosphamide |
|||||||||
Post-transplant methotrexate (group I) |
Fully matched HLA typing (group IIA) |
Haploidentical HLA typing (group IIB) |
P1 |
P2 |
|||||
N=40 |
N=22 |
N=18 |
|||||||
N |
% |
N |
% |
N |
% |
||||
Blood culture growth |
19 |
47.50% |
6 |
27.30% |
11 |
61.10% |
0.12 |
0.03 |
|
Sputum culture growth |
2 |
5% |
6 |
27.30% |
4 |
22.20% |
0.012 |
0.71 |
|
Urine culture growth |
4 |
10% |
1 |
4.50% |
2 |
11.10% |
0.692 |
0.58 |
|
CVL culture growth |
3 |
7.50% |
5 |
22.70% |
5 |
27.80% |
0.034 |
0.73 |
|
Fungal Mucositis |
13 |
32.50% |
13 |
59.10% |
9 |
50% |
0.043 |
0.57 |
|
CMV Reactivation |
19 |
47.50% |
6 |
27.30% |
8 |
44.40% |
0.177 |
0.26 |
|
Liver toxicity |
23 |
57.50% |
8 |
36.40% |
4 |
22.20% |
0.11 |
0.33 |
|
Renal Toxicity |
14 |
35% |
3 |
13.60% |
2 |
11.10% |
0.083 |
0.81 |
|
Hemorrhagic cystitis |
2 |
5% |
2 |
9.10% |
9 |
50% |
0.006 |
0.006 |
|
VOD |
3 |
7.50% |
0 |
0% |
2 |
11.10% |
0.644 |
0.196 |
|
aGVHD |
any |
19 |
47% |
6 |
27% |
9 |
50% |
0.485 |
0.145 |
II-III |
6 |
15% |
1 |
4.50% |
3 |
16.70% |
0.499 |
0.31 |
|
III-IV |
12 |
30% |
3 |
13.60% |
2 |
11.10% |
0.056 |
0.81 |
|
cGVHD |
Any |
27 |
67% |
5 |
22.70% |
3 |
16.70% |
0.002 |
0.709 |
Mild |
4 |
10% |
4 |
18.20% |
2 |
11.10% |
0.499 |
0.673 |
|
Moderate |
11 |
27.50% |
0 |
0% |
0 |
0% |
<0.001 |
- |
|
Severe |
12 |
30% |
1 |
4.50% |
1 |
5.60% |
0.003 |
0.884 |
|
Table 2 The transplant related morbidities and the Graft-verus-Host-Disease incidence rates
P1, comparison between group I and group IIA; p2, comparison between group IIA and group IIB
CVL, central ven ous line; CMV, cytomegalovirus; VOD, veno-occlusive disease
The incidence of aGVHD was 47%, 27% and 50% in group I, IIA and IIB respectively. Thirty Percent of group I experienced severe aGVHD while 13.6% of group IIA and 11.1% of group IIB had severe aGVHD with no significant difference among the three groups. Group IIA was associated with significantly lower incidence of cGVHD when compared to group I with an incidence of 22.7% and 67% respectively (P = 0.002). Group IIA was associated with reduced risk of extensive cGVHD to 4.5% compared to 30% in MTX group (P =0.003). No significant difference between both group IIA and IIB.
The outcome and survival data (Table 3; figure 1 and 2)
No significant differences were found in relapse rates among the studied groups (P>0.05). No significant differences were found regarding the number of deaths, non-relapse related mortalities and relapse related mortalities among the different groups (P>0.05). Three cases lost follow up at Post-transplant methotrexate group. No significant differences were found in OS and DFS data among the studied groups. The cumulative 4 years DFS and OS were 43.4% and 43.7 respectively in MTX group (group I), while they were 41.1% and 46.4% respectively in group IIA and they were 50% and 47% in group IIB, respectively. The mean DFS and mean OS were 62.9, 65.3 months respectively in group I, while they were 22.8 and 30 months respectively in group IIA and they were 27.7 and 25.9 months respectively in group IIB.
|
Post-transplant methotrexate |
Post-transplant Cyclophosphamide |
|
|
|
Fully matched HLA typing |
Haploidentical HLA typing |
P1 |
P2 |
||
N=40 |
N=22 |
N=18 |
|||
Relapse N (%) |
7 (17.5%) |
8 (36.4%) |
4 (22.2%) |
0.189 |
0.332 |
Death N (%) |
21 (56.8%) |
11 (50%) |
9 (50%) |
0.632 |
0.855 |
Non- relapse related mortality |
15 (40.5%) |
5 (22.7%) |
7 (38.9%) |
0.323 |
0.197 |
Relapse related mortality |
6 (16.2%) |
6 (27.3%) |
2 (11.1%) |
|
|
4 years DFS: |
|
41.1 |
50 |
0.453 |
0.957 |
4 years OS: |
43.7 |
46.4 |
47.1 |
0.619 |
0.653 |
Table 3 The outcome and the survival data
P1, comparison between group I and group IIA; p2, comparison between group IIA and group IIB
DFS, disease free survival; OS, overall survival
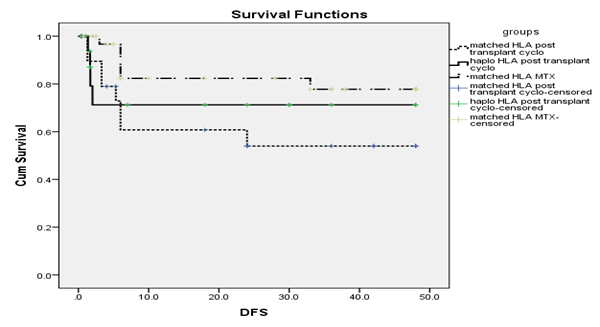
Figure 1 DFS in relation to GVHD prophylaxis and HLA typing.
No significant differences were found in DFS data among the studied groups. The cumulative 4 years DFS was 43.4% in MTX group (group I), while it was 41.1% in group IIA and 50% in group IIB, respectively. The mean DFS was 62.9 months in group I, while it was 22.8 months in group IIA and 27.7 in group IIB respectively.
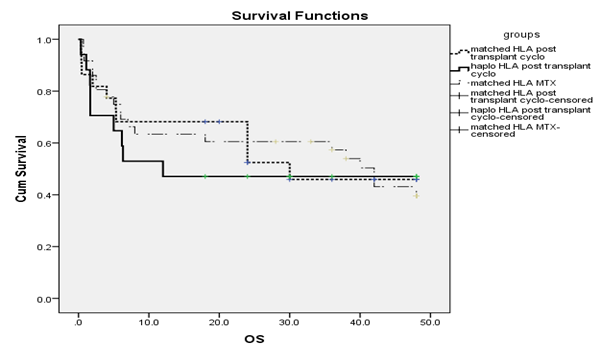
Figure 2 OS in relation to GVHD prophylaxis and HLA typing.
No significant differences were found in OS data among the studied groups. The cumulative 4 years OS was 43.7 in MTX group (group I), while it was 46.4% in group IIA and 47% in group IIB, respectively. The mean OS was 65.3 months in group I, while it was 30 months in group IIA and 25.9 months in group IIB respectively.
The causes of death (Table 4)
Sepsis as a cause of death was significantly associated with Post-transplant Cyclophosphamide (group IIA) when compared to Methotrexate groups (group I) (P=0.043). Otherwise, no significant differences were found in cause of death among the studied groups (p>0.05).
|
Post-transplant methotrexate |
Post-transplant Cyclophosphamide |
|
|
||||
Fully matched HLA typing |
Haploidentical HLA typing |
P1 |
P2 |
|||||
N=40 |
N=22 |
N=18 |
||||||
N |
% |
N |
% |
N |
% |
|||
Sepsis |
2 |
5% |
4 |
18.2% |
4 |
22.2% |
0.043 |
0.751 |
aGVHD |
3 |
7.5% |
1 |
4.5% |
1 |
5.6% |
0.644 |
0.884 |
cGVHD |
4 |
10% |
0 |
0% |
0 |
0% |
0.116 |
- |
Hemorhagic cystitis |
1 |
2.5% |
0 |
0% |
3 |
16.7% |
0.615 |
0.083 |
Nephro-toxicity |
1 |
2.5% |
0 |
0% |
0 |
0% |
0.314 |
- |
Hepatic-toxicity |
3 |
7.5% |
0 |
0% |
0 |
0% |
0.241 |
- |
Intra-cranial hemorrhage |
2 |
5% |
0 |
0% |
0 |
0% |
0.494 |
- |
Chest infection |
2 |
5% |
1 |
4.5% |
0 |
0% |
0.556 |
0.360 |
ARDS |
0 |
0% |
1 |
4.5% |
0 |
0% |
0.314 |
0.360 |
CNS relapse |
0 |
0% |
1 |
4.5% |
0 |
0% |
0.314 |
0.360 |
Relapse |
5 |
12.5% |
4 |
18.2% |
2 |
11.1% |
0.745 |
0.673 |
Lost follow up |
3 |
7.5% |
0 |
0% |
0 |
0% |
0.241 |
- |
Table 4 The causes of death
P1, comparison between group I and group IIA; p2, comparison between group IIA and group IIB
ARDS, acute respiratory distress syndrome
Regression analysis (Table 5 and 6)
Cox regression analysis was conducted for prediction of DFS and OS within all the studied patients using age, gender, diagnosis, type of donor, infection, aGVHD and cGVHD. Non was considered as a predictor for DFS or OS (Table 5). Logistic regression analysis was conducted for prediction of GvHD occurrence within all studied groups using age, gender, diagnosis, type of GVHD prophylaxis, type of donor and infection. Post-transplant cyclophosphamide was considered as a protective drug against cGvHD occurrence (Table 6).
|
DFS |
OS |
||||
|
P |
HR |
95% CI |
p |
HR |
95% CI |
Age |
0.329 |
1.019 |
0.981-1.058 |
0.290 |
1.021 |
0.982-1.062 |
Gender |
0.137 |
0.48 |
0.241-1.957 |
0.146 |
0.491 |
0.245-1.986 |
ALL versus AML |
0.622 |
1.191 |
0.595-2.385 |
0.398 |
1.354 |
0.670-2.735 |
Cyclophosphamide versus MTX |
0.426 |
1.284 |
0.695-2.372 |
0.704 |
1.130 |
0.601-2.123 |
Haplo vs fully matched HLA donor |
0.957 |
0.977 |
0.411-2.322 |
0.656 |
1.222 |
0.505-2.954 |
Infection |
0.337 |
1.658 |
0.591-4.653 |
0.390 |
1.574 |
0.560-4.425 |
aGVHD |
0.548 |
1.211 |
0.648-2.262 |
0.578 |
1.203 |
0.627-2.309 |
cGVHD |
0.144 |
0.505 |
0.260-1.981 |
0.136 |
0.479 |
0.241-1.953 |
Table 5 Cox regression analysis for prediction of DFS and OS within the studied subjects
DFS, disease free survival; OS, overall survival
|
p |
OR |
95% CI |
Age |
0.401 |
1.025 |
0.967-1.087 |
Gender |
0.563 |
0.758 |
0.296-1.941 |
ALL versus AML |
0.242 |
0.541 |
0.193-1.517 |
Cyclophosphamide versus MTX |
0.024 |
0.343 |
0.135-0.871 |
Haplo vs fully matched HLA donor |
0.358 |
1.806 |
0.512-6.363 |
Infection |
0.610 |
0.714 |
0.196-2.605 |
Table 6 Logistic regression analysis for prediction of GvHD within all the studied groups
Acute and chronic GvHD continues to compromise the overall success of allogeneic HSCT. The rates and severity of acute GvHD have decreased with improvements in the criteria of donor selection, pharmacologic prophylaxis, and supportive care. However, the rates of chronic GvHD have remained stable at 35% to 50% for many years. Chronic GvHD contributes to long-term morbidity and mortality after allo-HSCT. Therefore, strategies to prevent this complication without affecting thr beneficial graft-versus-tumor (GvT) effects are required.14-16
With the use of PT-Cy in allogeneic HLA-matched bone marrow transplant and the haploidentical setting, significant improvement in the prevention of GVHD has been made. Our study was conducted on 80 patients with acute leukemia received either fully matched allogeneic HSCT or Haplo-identical HSCT. They were compared according to the regimen of GvHD prophylaxis. We have shown that PT-Cy was effective as MTX in prevention of aGVHD. Mielcarek et al,16 used PT-Cy for 43 patients who received PBSC from related and unrelated donors. They reported a cumulative incidence of grades II-IV and III-IV acute GvHD of 77% and 0%, respectively. In the study of Mehta et al,17 41 patients received PT-Cy, tacrolimus and mycophenolate mofetil (MMF) for GVHD prophylaxis; 72 patients received conventional prophylaxis with anti-thymocyte globulin, tacrolimus and methotrexate. The incidence of grade II–IV (37% vs. 36%, P = 0.8) and grade III–IV (17% vs. 12%, P = 0.5) acute GVHD was similar at day +100. Even more, Moiseev et al,18 reported 86 patients with acute leukemias treated with PT-Cy, Tacrolimus and MMF. The main focus of their study was a retrospective comparison with an historical control group of patients treated with ATG, calcineurin inhibitors, and methotrexate. The cumulative incidences of grades II-IV acute GVHD (19% versus 45%, P = 0.003) and grades III-IV (4% versus 27%, P < .0001).
A salient finding of our study is that PT-Cy was associated with considerable decrease of cGvHD when compared with MTX. PT-Cy was associated with reduced risk of severe cGvHD to 4.5% in patients received HLA matched transplant compared to 30% in MTX group. Rashidi et al,19 in a study included 336 patients received PT-Cy–based haplo-HSCT and 869 received MSD using calcineurin inhibitor–based GvHD prophylaxis stated that the Haplo-HCT group had a considerable reduced rate of chronic GvHD. This finding was validated in logistic regression analysis for prediction of GvHD occurrence using age, gender, diagnosis, type of GVHD prophylaxis, type of donor and infection. Post-transplant cyclophosphamide was considered as a protective drug against cGvHD occurrence in all the studied cases.
87.5% of group I, 86.4% of group IIA and 94.4% of group IIB were engrafted with full donor chimerism at Day +30 with no statistical differences among the different groups. Times to neutrophil and platelet recovery were significantly prolonged in Post-transplant Cyclophosphamide group received fully matched donor compared to MTX group (P=0.002, 0.024 respectively). No significant differences were found in times to Neutrophils and platelets recovery between matched and haploidentical HLA typing groups received post-transplant cyclophosphamide (p>0.05). Mielcarek et al,16 used post stem cell cyclo for prevention of GVHD with a median time to platelet engraftment and neutrophil engraftment of 14 (range,10 to 74) days and 19 (range,16 to 37) days respectively with no effect on engraftment. Mehta et al,17 compared PT-Cy with conventional methods (anti‐thymocyte globulin, tacrolimus and methotrexate) and showed prolonged neutrophil and platelet engraftment in PT-Cy group than conventional methods and this was pronounced in patients who used bone marrow as a source of stem cells.
Hematopoietic cell transplant recipients are at substantial risk for a variety of infections depending upon the degree of immunodeficiency, exposure to pathogens and the time of neutropenia. Blood stream infections (BSI) are the most frequent bacterial infections in HSCT patients. They occur in 20–50% of allo-HSCT patients, with higher rates before engraftment, and are associated with increased morbidity and mortality.20-23 Fayard A et al,23 performed a retrospective study on 381 patients undergoing Haplotransplant with Post-transplant cyclophosphamide from 30 centers. A total of 1058 infectious episodes were diagnosed, affecting 90.3% of the cohort. Around 41% of the documented infections were of bacterial origin and 35% of viral origin. Among which 48.8% of the patients were presented with CMV reactivation. Our results showed incidence of Blood culture growth of 47.5%, 27.3% and 61.1% in group I, group IIA and IIB respectively. Haploidentical group showed significant more frequent blood culture growth when compared to fully matched (P = 0.031). Patients in group IIA who received PT-CY plus matched HLA transplant showed significantly higher incidence of sputum culture growth, CVL culture growth and fungal mucositis. This difference may be attributed to the difference in times to neutrophils recovery.
Hemorrhagic cystitis (HC) is one of the important complications which may occur after allo-HSCT. The incidence of HC after haplo with post-HSCT Cy is high and is associated with high morbidity rates, especially in those with a previous transplant history and with impaired immune reconstitution.24, 25 This was in agreement with our results that the PT-Cy group was significantly associated with higher incidence of HC when compared to MTX. Haplo subgroup was significantly associated with higher incidence of HC when compared to fully-matched group. On the other hand, our results showed no significant hepatic, renal or SOS among the different groups.
Mielcarek et al,16 used post PT-Cy for prevention of GVHD with relapse rate of 17%. The cumulative incidence of relapse in our study at 2 years after transplantation was 17.5 % for group I, 36.4% for group IIA and 22.2% for group IIB with no significant difference. Mehta et al,17 compared PT-Cy with conventional prophylaxis with ATG, tacrolimus and methotrexate showed no difference in relapse rate between both groups. The cumulative 4 years DFS and OS did not differ significantly among our patients. The cumulative 4 years DFS and OS in group I were 43.4% and 43.7% respectively while they were 41.1% and 46.4% in group IIA and 50% and 47.1% in group IIB respectively. Cox regression analysis was conducted for prediction of DFS and OS within all the studied patients, using age, gender, diagnosis, type of donor, infection aGVHD and cGVHD. Non was considered as a predictor for DFS or OS in all the studied cases.
The data from the Center for International Blood and Marrow Transplant Research (CIBMTR) and The European Society for Blood and Marrow Transplantation (EBMT) indicated that OS and PFS after PT-Cy haplo SCT were comparable with transplantation from HLA-matched related donor (MRD) or HLA-matched unrelated donor (MUD) in patients with acute leukemia. Almost all of these studies demonstrated the lower incidence of cGVHD in patients after PT-Cy haplo SCT along with similar relapse rates.26-30 A meta-analysis by Gu et al,31 verified these results. Nine case–control studies included 2258 patients (827 cases within the haplo-HCT with PT-Cy group; 748 controls within the MRD group; and 683 controls within the MUD group). They compared outcomes of PT-Cy haplo SCT with those of HLA-matched SCT. No differences were found between PT-Cy with haplo SCT and HLA-matched SCT regarding the incidences of acute GVHD, NRM, relapse rate, DFS, and OS. However, PT-Cy haplo SCT was associated with a significantly lower incidence of moderate to severe chronic GVHD which nearly matches our results.
PT-Cy with addition of IS drugs has statistically significant difference in reducing the incidence of cGvHD in both fully matched and Haplo SCT with less hepatic and renal toxicity. Meanwhile, PT-CY was equally effective as the conventional MTX containing regimen in aGVHD prevention. No effect on engraftment, relapse, DFS and OS when compared with methotrexate-based regimen. Further prospective, randomized studies with larger sample size are recommended to validate the efficacy of PT-Cy in reducing the incidence of GvHD either alone or when combined with other IS drugs.
The authors thank the patients and the clinical study team who participated in this research.
The authors declare that there is no conflict of interest.

©2022 Elshamy, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.