Advances in
eISSN: 2377-4290


Review Article Volume 8 Issue 3
Department of Ophthalmology and Visual Science, Yale University School of Medicine, USA
Correspondence: Brian M DeBroff, Department of Ophthalmology and Visual Science, Yale University School of Medicine, 46 Prince Street, Suite 404, New Haven, CT 06519, USA, Tel +203-375-5819; +203-913-1605
Received: May 02, 2018 | Published: May 17, 2018
Citation: DeBroff BM. The role of vision in the science and art of the putting stroke in the sport of golf. Adv Ophthalmol Vis Syst. 2018;8(3):156?159. DOI: 10.15406/aovs.2018.08.00292 DOI: 10.15406/aovs.2018.08.00292
Keywords: golf, putting, putting science, sports vision, sports visualization, sensorimotor, sports visual processing, target aiming, depth perception in sports, slope determination, plumb bobbing, target gaze position, gaze pattern, gaze variability, target focusing, perception-action coupling, sports visual devices, sports contrast sensitivity
DME, diabetic macular edema; CSME, clinical significant macular edema; OCT, optical coherence tomography; VEGF, vascular endothelial growth factor; BCVA, best corrected visual acuity; VA, visual acuity; ETDRS, early treatment diabetic retinopathy.
Clinically significant macular edema (CSME) cause early onset reduction of vision in patients with diabetes1 however CSME can be classified into central involving the fovea and non-central sparing the fovea where the former cause central visual acuity (VA) reduction the later patients presented with good VA, but whenever Non-central CSME progress to central a VA reduction occurs. The treatment of choice of central CSME is monthly intravitreal injection of AntiVEGF, which has proven safety and efficacy,2 however, laser photocoagulation as proposed by ETDRS (Early Treatment Diabetic Retinopathy) proved to reduce the risk of visual loss especially in cases of non-central CSME.3 Conventional laser treatment induce retinal burns that distract photoreceptors and retinal pigment epithelium, which interim will cause retinal scars that may spread to the fovea and lead to visual loss or cause sub retinal fibrosis or choroidal neovascularization. However, it is not essential to induce retinal burns to treat macular edema; clinical trials showed4 that using subthreshold diode micropulse photocoagulation is effective and safe treatment procedure to treat CSME without causing scars. Clinical trials that used subthreshold diode micropulse photocoagulation was at 810 nm and yellow 577 nm wave length, this clinical trial will determine if Sub Threshold Laser at 532 nm can reduce macular thickness in non-central CSME and stop the progression of non-central CSME to central CSME without causing any retinal scars in the macular area.
This is a randomized single center and open label single arm interventional study that adhered to the declaration of Helsinki, the study registered at clinical trials.gov (NCT03226951),Study received approval by institutional review board, and patients obtained signed consent after explaining the possible complications.
Study participant
Inclusion criteria
Exclusion criteria
Treatment protocol
One or both eyes can be included in this study, where eyes treated with subthreshold laser done at the base line after confirming the diagnosis of non-central CSME with OCT, which defined as retinal thickening above 299 micron within the ETDRS circle sparing the central 500 micron, which represent the foveola. Subthreshold laser applied using 532 nm single spot laser (spot size is 125m ) with 5% duty cycle 200 ms duration and 200 ms interval the power was defined using titration method i.e. laser applied with 5% duty cycle in non-edematous peripheral area and increased until a threshold tissue reaction is noted. Half of the power used for laser application and when approaching foveal avascular zone then only 1/3 of the power used where duration is 200 ms Laser application was done using low intensity high density protocol in painting fashion covering all the area of the edema Retreatment with subthreshold laser only in cases of macular edema worsening during follow up where AntiVEGF intravitreal injection is considered if edema enters the foveal avascular zone and reducing vision or vitreous hemorrhage precluding retinal view.
Follow up and outcome measures
All patients undergone dilated fundus exam and BCVA (best-corrected visual acuity) at baseline, and every 4 weeks using Snellen chart then converted to LogMAR to monitor visual acuity changes for 24 weeks. SD-OCT(OTI:OCT/SLO Heidelberg Engineering) was obtained at baseline and week 8 and then every 8 weeks to evaluate status of Non central CSME using OCT map topography, and HbA1C was obtained every 3 months to monitor glycemic control.
Statistical methods
Clinical data obtained and analyzed using SPSS V.20.0 Paired student t Test Calculator for 2 Independent Means used to analyze the difference between the mean 24 weeks outcome and mean baseline values for visual acuity and macular thickening.
Eight eyes out of seven patients has been included in these study 5 women and two men mean age 55.57±51Table 1 shows summarized baseline characteristics, all patients received Subthreshold laser treatment in the edematous area and followed up for 24 According to Table 2 there is no significant change in BCVA follow up from baseline where best-corrected visual acuity converted from Snellen to LogMAR for statistical analysis.
Age, Years (Mean±SD) |
55.57 ± 51 |
Sex, N (%) |
2 (29%) |
Eyes: N (%) |
4 (50%) |
Race: N (%) |
4 (57%) |
Lens: N (%) |
8 (100%) Phakic |
HbA1C: (%)(Mean±SD) |
7.84 ±1,01 |
Table 1 Characteristics of patients at presentation. According to (Table 1) seven patients were analyzed with the mean age of patients is 55.57 ± 51 of whom two are Men and five are Women.
|
Baseline |
Change |
P value |
BCVA, LogMAR Units |
0.05±0.05 (20/20) |
-0.02 (20/20) |
0.29 |
Retinal Thickness |
384.5±64.5 |
-73.17±51.7 |
0.028 |
Table 2 Mean changes of BCVA in LogMAR units, and Retinal thickness after subthreshold hold 532nm laser treatment from baseline and after 24 weeks.
Where retinal thickness was decreased from baseline of 384.5±64.5µm to 311.33±51.7µm to 24 weeks follow up (P=0.028) (Figure 1), 19.03% is the overall decrease in retinal thickness reduction from baseline to 24 weeks follow up (Figure 2). Six eyes were treatment success, reduced retinal thickness at the area of edema, and prevented visual loss from progression of edema into central 500 microns. Four eyes of three patients required re treatment after 16 weeks of treatment due to poor glycemic profile in two patients. However the other two eyes did not require additional treatment and their glycemic control maintained throughout 24 weeks. Two patients excluded from the study because they required intravitreal injection of Bevacizumab due to development of vitreous hemorrhage obscuring fundus view from subtle neovascularization from uncontrolled glycaemia and blood pressure, the other patient due to treatment failure with subthreshold laser and edema progressed into the central 500 microns causing reduced vision. There were no complications from subthreshold laser and there was no evidence of laser burns in the treated area in clinical examination.
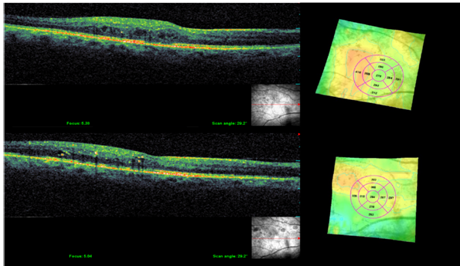
Figure 2 Shows NON central CSME in topography and tomography before (above) and after (bottom) treatment with subthreshold 532 nm laser showing that laser has reduced macular thickening and maintained BCVA 20/20 without causing central edema progression or retinal thermal damage, this patient had an average HbA1C 8% there for required repeat treatment at week 16.
This study showed that 532 nm subthreshold laser for non-central CSME as a monotherapy reduce retinal thickness at the area of the edema within 24 weeks with no sign of laser burns at the macular area, nor there was changes observed in retinal tomography in the external retinal layers such as external limiting membrane, ellipsoid zone or retinal pigment epithelium. Sub threshold laser stimulates retinal pigment epithelium to repair inner blood barrier without causing damage to photoreceptors via sub lethal injury by inducing heat shock protein without destroying retinal pigment epithelium and thus upregulating pigment epithelium-derived factor and down regulation of VEGF.5–7 Neural tissue protection can be achieved by subthreshold laser because energy delivered in microsecond envelope with “ON” time 100 microsecond of micropulsed power and “OFF’ 1900 micro second without power which gives time for the heated tissue to cool down and thus will activate retinal pigment epithelium without causing neural tissue damage. Due to retinal and pigment epithelium are close together a millisecond laser can cause damage to photoreceptors and retinal pigment epithelium to achieve a therapeutic effect, as the heat shock will diffuse to inner retinal tissues.
In contrast traditional laser achieve therapeutic effect when a white burn is seen because of retinal pigment epithelium thermal injury and thus can lead to fibrosis and possible choroidal neovascularization.8 Perente et al.9 compared the treatment with 532nm traditional focal laser or observation for non-central CSME, the laser arm achieved better outcome in term of vision and retinal thickness within 12 months, and retinal thickness reduced by average 22.6µm, when comparing it to this study were thickness reduced by average 73.17µm topography without causing retinal scars. Clinical trials tested effect of subthreshold laser using both 577 nm and 810 nm, to our knowledge this is the first study that uses 532nm subthreshold laser. When comparing results the 810 nm subthreshold laser reduced retinal thickening10 from358± 94 to 346± 113µm while 577 nm11 358±46 to 340±56µm, in this study a 532nm, where this study showed that subthreshold laser reduced retinal thickening from 384.5±64.5µm to 311.33±51.7µm.
When choosing 532 nm subthreshold laser for treatment non central CSME, it is important to consider the following
The weakness of this study is the following

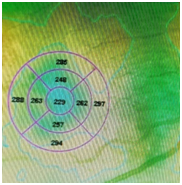
Figure 3 shows NON central CSME in topography before (Left) and after (Right) treatment with subthreshold 532 nm laser showing that laser has reduced macular thickening and maintained BCVA 20/20 without causing central edema progression or retinal thermal damage within 8 weeks however the central retinal thickness is normal whenever it is below 250 microns.
This study showed that 532nm subthreshold laser is effective in reducing retinal thickness and preventing the risk of progression of non-central CSME to central CSME and visual loss especially in cases of edemas with retinal thickness less than 400µm and with good glycemic control without causing retinal scars or visual loss.
However, long-term and larger studies are required to evaluate the true efficacy and durability of 532nm subthreshold laser.
None.
The author declares that there is no conflict of interest.

©2018 DeBroff. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.