Advances in
eISSN: 2377-4290


Case Report Volume 4 Issue 2
Faculty of Medicine of Health Sciences Center, Federal University of Rio de Janeiro UFRJ, Brazil
Correspondence: Luiz Reis Barbosa Junior, Faculty of Medicine of Health Sciences Center, Universidade Federal do Rio de Janeiro, Rua Rodolpho Paulo Rocco, 255-Cidade Universit ria Ilha do Fundão Rio de Janeiro RJ, Brazil, Tel (55 21) 3938-9600
Received: January 15, 2016 | Published: February 11, 2016
Citation: Reis L, Zangalli A, Dantas AM. Synophthalmus, agnathia and synotus. Adv Ophthalmol Vis Syst. 2016;4(2):34-36. DOI: 10.15406/aovs.2016.04.00099
Synophthalmus is the partial fusion of the two eyes - an incomplete form of cyclopia. Cyclopia (single midline eye) is a complete fusion of the two eyes with formation of a single median eye. The term cyclopia is derived from the cyclopes of Greek mythology, a race of fierce one-eyed-giants. Synophthalmus as well as cyclopia is part of a complex of severe anomalies caused by a disturbance of the prosencephalic organizing center during the first month of gestation. This results in deformities of the anterior part of the brain and mesodermal midline structures that are lethal.We describe two case reports of synophthalmus and its main features.
Keywords: synophthalmus, cyclopia, embryology, anomalies
In synophthalmus as well as in cyclopia, the eyes are partially or completely fused, forming a single median eye enclosed in a single orbit. There is usually a tubular nose (proboscis) inferior to the eye. 'Both anomalies are attributable to failure of a wedge-shaped area of the neural plate and the head mesoderm.1,2 The findings from Spemann 19073 on the induction constituted an important phase for the understanding of the theratogenesis. After that many experiments in animals had admitted that the cyclopia-synophthalmus are consequences of a partial or total inactivity of the organizing centers located at the prechordal mesoblast, responsible for the induction of the prosencephalon, and of the olfactory and visual systems. They result from the lack of the development of the medial structures of the face dependent on the frontal bud.4
The epiblast near the prechordal plate is fated to contribute to the forebrain and eyes, and it is this centrally located region, the eye field, that has acquired the potential to develop as ocular tissue. Inductively active mesodermal cells of the midline (prechordal plate, notochord) are necessary to divide the eye field along the midline to generate twoseparated eye fields. A key inductive signal in this process, is the sonic hedgehog gene. Failure of midline formation results in synophthalmus or cyclopia, deformities in which the midline structures are missing, producing two eyes or a single, centrally located eye. Homozygous mutants in the mouse sonic hedgehog generated by targeted mutagenesis result in multiple embryonic defects, includinq severe cyclopia.5,6 Although the mouse heterozygotes show no abnormalities, human patients with cyclopia or holoprosencephaly have been shown to carryheterozygous mutations in the sonic hedgehog gen.7
Agnathia (also known as otocephaly) is characterized by virtually absent mandible, extremely low-set, ventro-medially fusing ears (synotia) and microstomia with microglosia. A small oral opening to 3 mm wide leads directly into the pharynk. Agnathia is said to represent the most extreme form of first visceral arch defects, being incompatible with a variety of malformations, holoprosencephaly being the most common reported association. The association of synophthalmia and cyclopia with agnathia has been reported several times. In every case where the brain was available for study, brain defects were represented by alobar holoprosencephaly.8,9
The auricle develops around the first branchial groove. Its tissue is furnished both by the first (mandibular) branchial arch and the second (hyoid) arch. During the sixth week six hillocks appear on these parts - three on the caudal border of the first arch and three on the second arch. For many years it was held that the auricle develops in a rather precise manner from these six elevations and from an auricular fold of the hyoid integument. Later restudies of the problem differ in some details, even to asserting that the entire auricle, except the tagus, is of hyoid origin.10
The almost equally rare condition of synotus shows the ears fused, or near the midventral line at the upper part of the neck; it is associated with agnathus, and illustrates the primitive location of the primordial ear before being wedged apart by the growing mandible. The classification of malformations recognized under the heading holoprosencephalies has been described by De Myer and Zeman.11 Prosencephalic and median facial development are intimately related. Therefore, the more severe degrees of brain malformation may be predicted on the basis of the roentgen features, particularly as they refer to the midline facial configuration.12
In alobar holoprosencephaly, the most severe form of embryonic midline defects in inter-hemispheric cerebral fissure or lobes are present. This tectogenesis and arrest in cleavage result in:
In semilobar holoprosencephaly, lobes are recognized but no inter-hemispheric fissure is present. In lobar holoprosencephaly, lobes and an inter-hemispheric fissure are well formed; but one or more anomalies reflect a tendency for incomplete prosencephalic development. These anomalies include midline continuity of the frontal neocortex, agenesis of the olfactory bulbs and tracts, communication of the lateral ventricles, and agenesis of the corpus callosum with or without a dorsal sac. Molecular studies have led to the identification of several holoprosencephaly-related genes, including sonic hedgehog (shh).6
In the first case report, the body was of a male newborn weighing 2000 grams and 40 cm high. Cephalic perimeter 34cm and thoracic 30cm (Figure 1). This full-term infant died. His 25 years old mother had suffered no complication during the pregnancy and she had taken no drugs. The head had median two eyes and a nose that resembled an animal's trunk. The neck was short and each ear was small and curved. The two eyes were almost fused but centered in the middle of the face at the level of normal eye with rudimentary eyelids. The two corneas presented on the anterior surface of the median eye were fused along their medial margins.
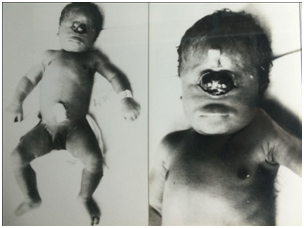
Figure 1 Fetus with synophthalmia and a proboscid. The second image shows the same case in a large size.
The opening of the skull revealed the presence of a large volume of limpid liquid; there were no cerebral hemispheres and the cerebral aqueduct could be easily opened at the cranial cavity. The exam of the nervous central system revealed a normal cerebellum occupying the posterior fossa. The following structures were not found: optic nerves, bulbs and olfactory tracts. All the remainder pairs of the cranial nerves were found. In the second case, the body was of a male newborn weighing 1100 grams and 30 cm high. Cephalic perimeter 28cm and thoracic 20cm (Figure 2). The full-term infant was born dead and his 20 years old mother has suffered complication during the pregnancy. She had taken drugs.
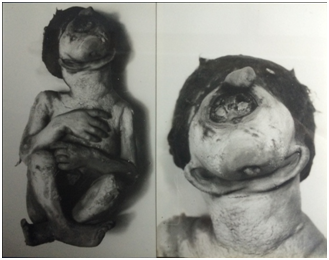
Figure 2 Fetus with synophthalmia, proboscid and a rare case of neck short with the ear in a primitive region, that is, synotus combined with microstomus and agnathus.The second image shows the same case in a large size.
The microcephalic head had two median eyes almost fused and a proboscid. The neck was short and each ear was in the primitive region, that is, synotus combined with microstomus and agnathus. The two eyes were fused but centered in the middle of the face at the level of normal eye, the two corneas present on the anterior surface of the median were fused. The opening of the skull revealed an alobar holoprosencephaly with absence of the optic and olfactory nerves, and presence of a rudimentary midbrain.
Synophthalmia and cyclopia, which are anomalies of organogenesis of the eye, result from arrest of development of the anterior end of the neural plate.13–15 Thus, they are always associated with the abnormalities of the brain. The anomaly was classified by De Myer et al.16as the most extreme form of holoprosencephaly when they graded the disorders in which there is impaired underline cleavage of the embryonic forebrain. The same authors correlated the appearance of the face and the type of brain malformation in holoprosencephaly. Confirming that view, Torczynski et aI.,13 reported data based on the analysis of a series of 35 cases of synophthalmia-cyclopia.
In the early development of the embryo certain groups of cells (anlage) differentiate into masses destined to give rise to definite organs or tissues. Injury or abnormality of these specialized cell masses at certain critical stages during their development can result in congenital malformation of the organs or tissues17,18 suggested that these anomalies are a lethal autosomal recessive trait19 suggested that some cases of holoprosencephaly may be caused by an autosomal recessive gene in na individual with normal karyotype, and suggested that the responsible gene is located on the short arm of the chromosome 18.
It is important to know that in several cases chromosomal stud es were normal.20Exposure to drugs or to other potentially teratogenic environmental factors during organogenesis has been regarded as the basis of these anomalies. These factors were ionic radiation, contraceptives, viraemia plus corticosteroids and salicylates, rubella vaccine, antibiotics, and amidopyrine (amiropyrine).21,22 Genetic errors with chromosomal abnormalities such as trisomy-D, monosomy-G mosaicism, translocation affecting chromosome 3 and group C chromosome, and chromosome 10 short arm deletion have also been reported in cyclopia-synophthalmus.13,23-32
None.
No conflicts to declare.
None.

©2016 Reis, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.