Advances in
eISSN: 2377-4290


Mini Review Volume 10 Issue 3
1Center for Research and Teaching in Health Sciences, CIDOCS, Hospital Civil de Culiacán, Universidad Autónoma de Sinaloa, Mexico
2Neurosciences, Hospital Ángeles Culiacán, Mexico
3Neuroscience Laboratory, Center for Research Applied to Public Health (CIASaP), Faculty of Medicine, Autonomous University of Sinaloa, Mexico
Correspondence: Abel Ramón-Concepción, Center for Research and Teaching in Health Sciences, CIDOCS, Hospital Civil de Culiacán, Universidad Autónoma de Sinaloa, Mexico, Tel 6673453755
Received: May 21, 2020 | Published: June 1, 2020
Citation: Ramón-Concepción A, Romo-García E, Álvarez-Félix JR, et al. Micro RNAs in neurodegenerative eye disease. Adv Ophthalmol Vis Syst. 2020;10(3):54-57 DOI: 10.15406/aovs.2020.10.00384
Introduction: Neurodegenerative diseases affect the central nervous system and cause progressive dysfunction. These debilitating and incurable conditions are characterized by loss of neuronal cell function and are often associated with atrophy of the affected nervous system structures. The retina and the optic nerve are considered an extension of the central nervous system due to their embryonic origin and myelination by oligodendrocytes. Thereby, common physiopathological mechanism may be implicated in the neurodegeneration progress, regulated by key molecules as microRNAs.
Aim: To determine the expression profile of microRNAs in peripheral blood in patients with neurodegenerative eye diseases such Glaucoma and Alzheimer´s disease.
Methods: Observational, multicentric, comparative study. 50 patients distributed into four groups were included for this study: 10 (20%) with Alzheimer's disease, 20 (40%) with Glaucoma and 20 (40%) healthy control individuals.
Results: miR-155 was found to be with tendency to overexpression in the Glaucoma group. miR-483 was found to be significatively overexpressed by five times in the Glaucoma group compared to control individuals. No significant changes were observed for Alzheimer’s disease group in both microRNAs.
Conclusion: The preliminary results observed in this study, may suggest that the miR155 and miR483 microRNAs show peripheral blood levels that can be measured in a repeatable and reliable way, this opens the way to a new route of early diagnosis and possible treatment for neurodegenerative eye disease like glaucoma.
Keywords: miR-155, miR-483, retina, optic nerve, neurodegeneration
Neurodegenerative diseases like Alzheimer's disease (AD), glaucoma and age-related macular degeneration (AMD) represent a real challenge in diagnostic and therapeutic strategies.1,2 AD is associated with the formation of amyloid plaques that are the result of extracellular accumulation of the β-amyloid peptide (Aβ42/Aβ40) and with neurofibrillary tangles made by the aggregation of hyperphosphorylated Tau protein.2 These pathological mechanisms cause the progressive and massive loss of neurons in specific areas related to memory and learning. Recent evidence has shown that the generation of the Aβ42 / Aβ40 peptides is also present in the retina of the eye; and that the accumulation of these peptides in a toxic way can contribute to the pathogenesis of glaucoma and AMD. This ophthalmopathies present common etiological characteristics with AD, such as: excitotoxicity, oxidative stress, neuroinflammation, chronic gliosis, trans-synaptic degenerative changes and genetic risk factors.1,3 The prevalence of glaucoma in Alzheimer's patients is significantly higher than in patients without dementia.3 The strongest evidence linking these two neurodegenerative diseases is that in models of glaucoma and in the retina of post-mortem patients; there is a significant increase in the levels of the Aβ42 / Aβ40 peptides, which is associated with the development of apoptosis of the CGR.4 It is possible that this neurodegenerative ophthalmopathy is part of a pre-clinical stage of Alzheimer's, or a non-memory-related manifestation, so it is important to determine the presence of biomarkers for Alzheimer's in glaucoma patients (without clinical signs of Alzheimer's). A more accurate diagnosis of AD requires behavioral examinations, detection of Aβ and p-Tau peptides in cerebrospinal fluid, and neuroimaging techniques (MRI and PET). However, these techniques are very expensive and sensitive only when the pathology is already advanced. Thus, it is necessary to use new biomarkers that can help early diagnosis in the pre-clinical stage, such as may be glaucoma. Furthermore, clarifying the molecular mechanisms that converge between these neurodegenerative diseases will help to clarify whether Alzheimer's treatments protect neurodegeneration in the retina or vice versa. Although the pathological molecular genesis of amyloid peptides can be used as a diagnostic and or prognostic biomarker, this strategy has several disadvantages like being a very invasive procedure and may be low sensitive at early asymptomatic stages.2 Furthermore, these pathological events are regulated by other molecular mechanisms; among which molecules such as microRNAs stand out.4,5 Thereby, they have been proposed as early biomarkers with great advantages, such as, being detected in practically all human biofluids, making them accessible and non-invasive.6 Several specific microRNAs have been shown to be involved in the pathological increase of Aβ42 / Aβ40 peptides . In the present report, an exploratory study of two AD related microRNAs,7 miR-155 and miR-483, was perform.
MicroRNAs (miRNAs)
They are a new class of non-coding single-stranded small RNAs (18-25 nucleotides) involved in regulatory post-transcription of gene expression. They are involved in almost all biological processes such as proliferation, development, apoptosis, inflammation, and their expression is highly regulated by enzymes or epigenic mechanisms. They have a specific distribution in the tissues and their proliferation has been found in chronic diseases. They are found in all human biological fluids such as plasma and peripheral blood. They can be measured by different methods, the most used are DNA Chip and quantitative PCR.8
Role in neurodegeneration and eye disease
Recently, microRNA-type biomarkers are being studied in chronic pathologies, cancer, infections and degenerative diseases. In neurodegenerative diseases like Alzheimer's the role of markers such as MiR-9, MiR-155 and MiR-34 have been established as involved in the pathophysiology of degeneration of the central nervous system, retina and the optic nerve are considered an extension of the central nervous system due to their embryonic origin and myelination by oligodendrocytes.9,10 These markers can be measured in peripheral blood.11
Study design
A pilot observational, multicentric, comparative study was performed. Identification of patients of three centers with a diagnosis of age-related macular degeneration or glaucoma was conducted, a complete ophthalmological evaluation was performed and 1 ml of peripheral blood was collected, prior filling and explanation of informed consent. Cold storage and transportation of samples to the Neuroscience Laboratory at the Universidad Autónoma de Sinaloa were carried out for analysis to quantify microRNA levels from plasma and using the qRT-PCR technique (Reverse Transcription coupled to Real- Time Polymerase Chain Reaction).
MicroRNA quantification
Total miRNA purification was performed with the miRNeasy-serum/plasma extraction kit (Qiagen, Hilden, Germany) according to the manufacturer’s protocol. Samples were eluted with RNase-free water for subsequent cDNA synthesis and stored at -80°C. The microRNA quantification was performed by qRT-PCR (Reverse Transcription coupled to Real- Time Polymerase Chain Reaction). Total RNA was reverse transcribed using a miScript II RT Kit (Qiagen, Hilden, Germany) in a StepOne Plus (Thermo Fisher Scientific, MA, USA) according to the manufacturer’s protocol. Quantification of miR-155 (Cat. No. 00031486) and miR-483 (Cat. No. 00009758) mature miRNAs were determined using the miScript SYBR Green PCR kit (Qiagen, Hilden, Germany) and SNORD68 (Cat. No. 00033705) as normalizer. PCR reactions were each performed in a final volume of 15µl in the StepOne Plus thermocycler. The Melting Curve was performed to observe the specificity of reactions. Relative quantification was obtained using the Pfaffl method, which describes the use of reaction efficiencies obtained using LinReg PCR software.12 Statistical tests were performed using GraphPad Prism. Data were examined for normality. Mann-Whitney U and Student’s t-test were used to examine the statistical difference in between groups respect to miRNA expression. Statistical significance was set at p< 0.05.
In order to explore changes in the expression of two AD related microRNAs in the glaucoma context a pilot observational, multicentric, comparative study was performed. The population consisted of 50 patients distributed in three groups: 10 (20%) patients with Alzheimer's disease, 20 (40%) with Glaucoma and 20 (40%) healthy control individuals. The gender was 30 (54.54%) women and 25 (45.45%) men. The microRNAs miR-155 and miR-483 were quantified by qRT-PCR technique and the expression levels related to the control healthy group. The miR-155 expression showed a tendency to up-regulation in the glaucoma group compare to control group (Figures 1&2). No changes were observed in the AD group, perhaps due to the study population size. Respect the analysis of miR-483, it was observed a five-time significant up-regulation in the level expression in the glaucoma group compare to control group. No significant changes were observed in the AD group. Together, these results suggest that AD related microRNAs, like miR-483, may be altered in other neurogenerative diseases like glaucoma; and perhaps cross talking in the early pathophysiological mechanisms for neurodegeneration in central nervous system.
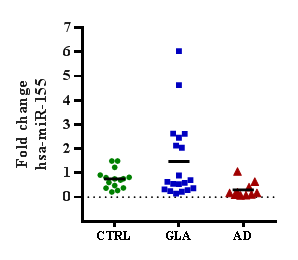
Figure 1 The microRNA miR-155 expression in glaucoma and Alzheimer´s disease patients. Expression of miRNA miR-155 measured by qRT-PCR in GLA or AD people and referenced to control individuals. Data are plotted as relative value for each individual, and means for each group. GLA, glaucoma; AD, Alzheimer´s disease; CTRL, control.
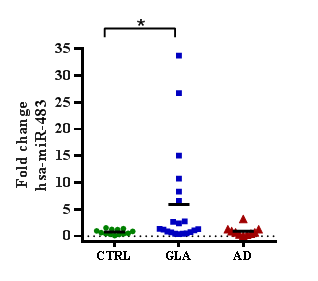
Figure 2 The microRNA miR-483 is up-regulated in glaucoma patients. Expression of miRNA miR-483 measured by qRT-PCR in GLA or AD people and referenced to control individuals. Data are plotted as relative value for each individual, and for each group statistically analyzed by Mann-Whitney U test (*p<0.05). GLA, glaucoma; AD, Alzheimer´s disease; CTRL, control.
The particular role of different microRNAs is currently being studied. Chen et al found retinal biomarkers in patients with age-related macular degeneration including drusen, pseudodruse, macular atrophy, and retinal pigmented subepithelial deposits that correlate with visual acuity and suggest the need to search these patients for genetic indicators that have relate to neurodegeneration.13 This is supported by the theory that the retina and optic nerve are direct extensions of the central nervous system, so that genetic indicators that have been found in the brain of patients with neurodegenerative diseases such as Alzheimer's could also be found at the ocular level and in blood. Alexandrov studied a group of 6 microRNAs associated with plasma neurodegeneration from patients with degenerative disease and found dysregulations in miR502, miR103, miR301, miR142, miR200, none of them similar to those studied in the present work25. Opening an important area of interest since it showed that in peripheral blood there are also sufficient levels and that they can be measured with special techniques such as PCR, this directed search in blood samples and not in biopsies of the central nervous system are postulated to begin investigating tests serological as pre-established genetic markers that allow early and less invasive diagnoses, as well as the cost reduction of studies such as nuclear magnetic resonance imaging for the evaluation of established damage in the white and gray matter of patients with neurodegeneration.14 Although there are different neurodegenerative diseases, it was preferred to use Alzheimer's disease as it is the most studied and prevalent neurodegenerative disease, Zhuang and Yan published several works involving microRNAs as genetic elements that must be involved from the beginning of the disease and are postulated for a new early therapy, for the purpose of this work it served as a cornerstone to study those same microRNAs already related to cognitive neurodegeneration but now in patients with degenerative retinal disease such as age-related macular degeneration and optic nerve degenerative processes such as Glaucoma.15,16
Gonzalez and Guorng Li studied microRNAs in the trabecular meshwork, however we cannot find any specific correlation because they are different types, in terms of concentration we can say that in peripheral blood it is lower since there may be a phenomenon of dilution in plasma or even lysis because they are fragments of very small genetic material.17 The preliminary results observed in this study, may suggest that miR155 and miR483 microRNAs show peripheral blood levels that can be measured in a repeatable and reliable way, this opens the way to a new route of early diagnosis and possible treatment; and thus postmortem biopsies of the central nervous system, expensive studies such as magnetic resonance imaging are no longer the only way to study and address neurodegeneration inside and outside the central nervous system.18,19 Peripheral venous blood was a reliable route for microRNA research, as it is minimally invasive compared to other classic diagnostic routes such as lumbar puncture. Serological samples can be obtained that are easily transportable with a conventional cold network and that, with the appropriate equipment, in this case RT-PCR, can be evaluated safely and repeatably with very small volumes.
The microRNAs included in the analysis of this work, miR483 and miR155, are potential biomarkers of genetic dysregulation and open the way for the search for therapeutic options that impact the evolution of neurodegeneration. New studies with a larger number of patients, different measurement techniques and longer follow-up are suggested, although most of the reports at the moment include a small sample due to the difficulty of excluding other metabolic diseases that modify the measurements of microRNAs and the high cost of equipment for genetic study.20
None.
My research project was not partially or fully sponsored.
Author declares that there is no conflict of interest.

©2020 Ramón-Concepción, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.