Advances in
eISSN: 2377-4290


Editorial Volume 8 Issue 1
Department of Ophthalmology, Yuksek Ihtisas University, Turkey
Correspondence: Burak Turgut, Professor of Ophthalmology, Yuksek Ihtisas University, Faculty of Medicine, Department of Ophthalmology, 06520, Ankara, Turkey, Tel +90 312 2803601, Fax +90 3122803605
Received: February 21, 2018 | Published: February 28, 2018
Citation: Turgut B, Lamina cribrosa measurements for glaucomatous eyes: a new focus point in optical coherence tomography. Adv Ophthalmol Vis Syst. 2018;8(2): 0027 DOI: 10.15406/aovs.2018.08.00271
Lamina cribrosa, measurements, optical coherence tomography, glaucoma, anterior surface depth, curvature index, laminar thickness, prelaminar tissue thickness
LC, Lamina cribrosa; RGC, retinal ganglion cell; IOP, intraocular pressure; GON, glaucomatous optic neuropathy; RNFL, retinal nerve fiber layer; OCT, optical coherence tomography; EDI-OCT, enhanced imaging OCT; SS-OCT, swept source OCT; ALCS, anterior LC surface; ALCSD, anterior LC surface depth; PTT, prelaminar tissue thickness; LCT, LC thickness; BM, Bruch’s membrane; BMO, BM opening; LCCI, LC curvature index; LCCD, LC curve depth.
Lamina cribrosa (LC) is the part of the sclera with a mesh-like structure and it is the exit zone from the globe of retinal ganglion cell (RGC) axons. It has a lot of pores formed by the multi-layered network of collagen fibers. It has been considered that LC maintains the pressure gradient between intraocular pressure (IOP) and the periocular pressure and it structurally supports RGC axons. As LC is structurally weaker than other parts of sclera which are thicker and denser, it is highly sensitive to the effects of the IOP elevation.1,2 LC is a dynamic structure responding with thinning or thickening to IOP changes. Following sudden IOP increase, optic cup excavation associates with thinning in prelaminary tissue. The IOP elevation causes the compression, posterior displacement or bulging of the LC, deformation of the pores, kinking and pinching of the RGC axons and blood vessels traversing through the laminar pores. It has been thought that the blockade of axoplasmic flow within RGC or ischemic damage to RGC axons due to compressional LC deformation caused by the distortion or posterior displacement of the LC might lead to glaucomatous optic neuropathy (GON). Additionally, it has been demonstrated in experimental studies that posterior displacement of LC precedes early surface-detected structural damage and retinal nerve fiber layer (RNFL) loss. Thus, LC is the putative primary region of the axonal injury in RGC in GON.1,2
Enhanced depth imaging optical coherence tomography (EDI-OCT) and swept source OCT (SS-OCT) have provided better visualization of the vitreoretinal interface, retina, optic disc, choroid and choroidoscleral junction. In recent years, these imaging modalities have been also used in the imaging of LC in especially patients with glaucoma and healthy subjects, and the posterior displacement and focal defects in LC detected by OCT have been linked to GON.3–10 Additionally, it has been also reported that the LC moved anteriorly with the reduction in the depth of LC following IOP lowering in the patients with glaucoma. The reversibility of the LC has been related to both the anterior displacement of the LC and the thickening of the prelaminary tissue.11–13 Moreover, it has been demonstrated in experimental glaucoma that the displacement of the LC might precede structural LC damages and early RNFL defects.14 The measurement of the parameters related with LC such as anterior LC surface (ALCS) depth, prelaminar tissue thickness (PTT) and LC thickness (LCT) may reveal posterior LC displacement and the thinning of LC (Figure 1).15–21 ALCS depth (ALCSD) is defined as the distance from the Bruch’s membrane opening (BMO) level to the ALCS. BMO is defined as the termination point of Bruch’s membrane (BM) and it seemsas a high-reflection and high-contrast line in EDI-OCT. ALCS is the surface between the prelaminar tissue and the lamina cribrosa. Recent reports suggest that ALCSD might vary according to sex and axial length and it should be measured at various planes and sections.7,8,11,12,15-17
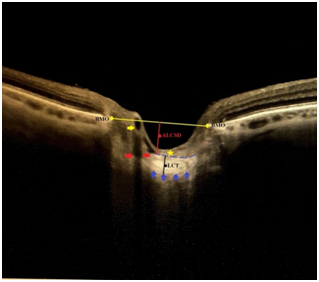
Figure 1 Some terms and parameters to be used in lamina cribrosa morphology in glaucoma. BMO, bruch membrane opening; ALCSD, anterior lamina cribrosa surface depth; LCT, lamina cribrosa thickness. Yellow and red arrows indicate the vascular trunk in the optic disc and its shadowing, respectively. Yellow star points out the prelaminary tissue. Break blue line indicates anterior lamina cribrosa surface. Blue arrows point out the possible posterior lamina cribrosa surface.
LCT is defined as the distance between the anterior and posterior borders of the hiper-reflective region visible beneath the ONH in a cross-sectional B-scan SS-OCT or EDI-OCT image. It has been reported that LCT was thinner in eyes with primary open angle glaucoma and normal tension glaucoma compared with normal eyes.11–13 However, the clear detection of the posterior border of the LC may always not be possible in OCT and its exact borders may be only confirmed histologically.18 The prelaminar tissue is composed primarily of RGC axons and glial cells which support them. PTT is measured as the distance between ALCS and vitreous-optic nerve head interface or anterior prelaminar surface in the region enclosed by the Bruch's membrane opening.11, 13,19 In a recent study, it has been demonstrated that prelaminar tissue in healthy eyes had significantly thicker than the eyes with glaucoma or glaucoma suspect, and it was significantly thicker in the eyes with glaucoma suspect than glaucomatous eyes. Additionally, it has been reported that of the anterior LC boundary was significantly more visible in glaucomatous eyes compared to healthy eyes.19 Lamina cribrosa curvature index (LCCI) is an LC-related index defined by Lee et al to quantify the posterior bowing of the LC using with SS-OCT.7 LCCI is calculated by dividing the lamina cribrosa curve depth (LCCD) by the width (W) of the ALCS reference line and multiplying by 100 (LCCI=(LCCD/W) X 100). LCCD is determined as the maximum depth from this reference line was set in each B scan by connecting the two points on the ALCS that met with the lines drawn from each Bruch’s membrane termination point perpendicularly to the BMO reference line to the ALCS. ‘’W’’, width, is the length of this reference line. It has been reported by Lee et al that LCCI has discriminating capability between glaucoma and healthy eyes.7
All of these LC related measurements or calculations may be beneficial in the diagnosis and the evaluation of treatment and prognosis in glaucoma patients. However, it should not forget that above-mentioned measurements especially, the measurement of LCT and especially, the visualization of posterior LC surface might be affected by OCT signal ratio, individual variations in PTT, optic disc pit, focal LC defects, neuroretinal rim, thick prelaminary tissue, shadowing due to vascular trunk in optic disc and LC segmentation errors.7–10,15–21 Further studies on LC morphology and indices will be able to provide more significant knowledge for the usage of LC parameters for early diagnosis and follow-up in glaucomatous eyes.
None.
The author declares that there is no conflict of interest regarding the publication of this paper.
The author received no financial support for the research, authorship, and/or publication of this article.
Concept and Design, Data Collection, Literature Search, Writing: Analysis, Interpretation: Burak Turgut.

©2018 Turgut. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.