Advances in
eISSN: 2377-4290


Research Article Volume 1 Issue 3
1Department of Ophthalmology, Queen Mary's Hospital, UK
2Department of Pediatric Ophthalmology and Strabismus Kettering General Hospital UK
Correspondence: Wai Hong Chan, Consultant Ophthalmologist, Kettering General Hospital, Kettering Northants, NN16 8UZ, UK
Received: October 25, 2014 | Published: November 8, 2014
Citation: Rodrigues IA, Chan WH. A Comparison of the Icare pro rebound tonometer with applanation tonometry in healthy infants. Adv Ophthalmol Vis Syst.2014;1(3):70-73. DOI: 10.15406/aovs.2014.01.00016
Aims: To study the accuracy and patient tolerability of the new Icare Pro rebound tonometer (ICPRO) compared with applanation tonometry in infants aged under 24 months.
Methods: Prospective observational study of 50 consecutive patients with expected normal intraocular pressure (IOP). One eye of each patient was examined sequentially using an ICPRO and Perkins applanation tonometer (PAT) to obtain a single adequate IOP measurement for each device. Agreement was assessed using Bland-Altman analysis. An objective assessment of patient tolerance of IOP measurement was also recorded.
Results: Mean age of patients was 12.6 months. Correlation of IOP measurement between PAT and ICPRO was moderate (r=0.384, p=0.0059). ICPRO underestimated IOP in 88% of cases: IOP was measured significantly lower with ICPRO (9.18±1.57 mmHg) compared to PAT (12.4±2.95 mmHg, p<0.001). The 95% limit of agreement (LOA) was -8.6 to 2.2 mmHg. Subgroup analysis showed ICPRO underestimated IOP most in infants over 12 months (mean: 4.8mmHg, 95% LOA:-9.5 to 0.2mmHg, p<0.0001). Measurement of IOP with the ICPRO was significantly better tolerated, causing distress and crying in 6%, compared to 62% with PAT (p<0.0001). IOP underestimation with ICPRO was significantly greater when infants were crying during PAT measurement (5.26±2.83) compared to when infants were not crying during PAT measurement (1.97±1.83, p<0.0001).
Conclusion: ICPRO appears to be safe, tolerable and of adequate accuracy for the screening of IOP in infants. It consistently significantly underestimated IOP, therefore high or borderline IOPs measured with the ICPRO should be repeated with applanation tonometry to prevent missing cases of raised IOP.
Keywords: intraocular pressure, glaucoma, applanation tonometry, infants, icare pro
ICPRO, icare pro rebound tonometer; IOP, intraocular pressure; LOA, limit of agreement; PAT, perkins applanation tonometer.
The measurement of intraocular pressure (IOP) in children is an important component of the ocular examination, but can be challenging, especially in younger children and infants. Clinical suspicion of raised IOP necessitates examination under anaesthesia. This is costly,1 associated with potentially serious adverse events,2 including developmental delay,3 and uses valuable paediatric theatre resources. It is therefore important to have devices to measure IOP in infants that are both well tolerated and accurate. Paediatric ophthalmic examination of infants in the hospital environment also requires such devices to be portable, easy to use and transportable between wards, theatre, outpatient’s services and for use during on-call and emergency situations. There are a number of methods to measure IOP in children, all with benefits as well as drawbacks in terms of accuracy, ease of use and patient tolerance.4,5
Historically, the Perkins applanation tonometer (PAT) has been the most suitable and widely used instrument,6 and has shown to be highly correlated to Goldman applanation Tonometry.7 However it requires topical anaesthesia, which can cause discomfort and rarely allergic reactions.8,9 An experienced tonometrist is also required to avoid inter-observer variability, and it may be difficult to perform in children, patients with blepharospasm and those with corneal abnormalities. Pneumatic non-contact tonometry is easy to use, but requires bulky and immobile equipment and is therefore primarily used by optometrists. Digital contact tonometry (such as the Tonopen) is portable and does not require significant experience or training to use. It does however involve prolonged direct contact with the eye to take 10 separate readings and therefore requires topical anaesthetic like applanation tonometry. The length of contact required with the eye when using the Tonopen may cause similar distress to applanation tonometry, and it has been shown to disagree with PAT measurements of IOP in children, with significant overestimation when the IOP is greater than 16mmHg.10
An alternative to PAT that uses the principle of rebound tonometry has been available for a number of years.11 I-care tonometry involves a magnetized probe that is propelled towards the eye using a solenoid. The motion and impact of the probe colliding with the eye and bouncing back is detected by the solenoid. A moving magnet in the probe induces voltage in the solenoid, and the motion parameters of the object are monitored. I-care tonometry has the advantages of not requiring topical anaesthesia, minimising corneal injury and avoiding risk of cross-infection through use of disposable probes. The accuracy of rebound tonometry has been found to be independent of corneal thickness, but not corneal hysteresis or corneal resistance factor, which represents the overall resistance of the cornea.12
The original I-care was limited by requiring the patient to be in the upright position in order to perform IOP readings, to prevent the probe dislodging. This made it challenging for neonates with minimal tone in their back muscles, who are unable to sit upright. The newly developed Icare PRO (ICPRO; Icare Finland Oy, Espoo, Finland) has a built-in inclination sensor that allows measurement of the eye in supine patients as well as standard measurements whilst upright. Jablonski et al. recently compared the ICPRO to applanation tonometry and found it to be reliable and show good correlation.13 These findings were confirmed by Kim et al who also found the correlation between the ICPRO and applanation tonometry to be independent of the central corneal thickness, age, axial length or spherical equivalent.14
All the previously mentioned tonometers have been extensively tested and calibrated for measuring IOP in adults,15 however, their accuracy in children and particularly infants without general anaesthesia is poorly documented. We have previously reported on the accuracy of applanation, pneumatic, rebound and digital tonometers in children.16 The original Icare rebound tonometer has been shown to have good correlation with applanation tonometry in healthy children,17 as well as children with known or suspected glaucoma.18,19 The mechanical properties of the cornea in infants however have been shown to significantly differ to that of the adult.20−22 Since all current tonometers are calibrated using the normal parameters of the adult cornea for thickness, rigidity, and hysteresis, there is potential for introduction of clinically significant inaccuracies when measuring IOP in infants using different tonometers. The aim of our study therefore was to determine the accuracy of IOP measurement using the ICPRO tonometer in infants, in comparison to the PAT. In order to determine if it is an acceptable alternative method, we also evaluated how well tolerated these devices are by infants and if increased distress of the patient influenced the accuracy of the IOP measurements.
This was a prospective observational study. We included infants under the age of 24 months who were referred to our outpatient department for ophthalmic review. The acquisition of the data was in order to verify the accuracy of this device in this age group. This was discussed with the local ethics committee and was deemed to fall under the category of service evaluation and did therefore not require any further formal ethical approval.
IOP measurements were obtained in either an upright or supine position. Topical anaesthesia with 1 drop of G. proxymetacaine 0.5% was instilled and a wire speculum was placed. IOP was measured with the ICPRO or PAT with disposable single-use probes. One accurate measurement with each device for one eye of each patient was recorded sequentially during the same examination by a single examiner (WHC). The average IOP value was obtained using the ICPRO from 6 consecutive measurements where the numerical standard deviation was within 95%. The PAT was a Perkins hand-held applanation tonometer designed for IOP measurement in the supine position (Hand-held applanation tonometer MK-II; Clement Clarke, Harlow, UK).
The order of the IOP measurement was alternated between ICPRO followed by PAT and PAT followed by ICPRO. Significant distress observed as crying experienced during IOP measurement was noted. The means and standard deviations of the IOPs measured by both tonometers were compared. The Bland-Altman plot was used to compare the bias and 95% limits of agreement between the ICPRO and PAT measurements. In the Bland-Altman analysis the difference between each IOP measurement was plotted against the mean. Linear regression of the Bland-Altman analysis showed whether any overestimation or underestimation of IOP had arisen within the measured range. All statistical analyses were performed using Analyse-it for Microsoft Excel, Cambridge, UK.
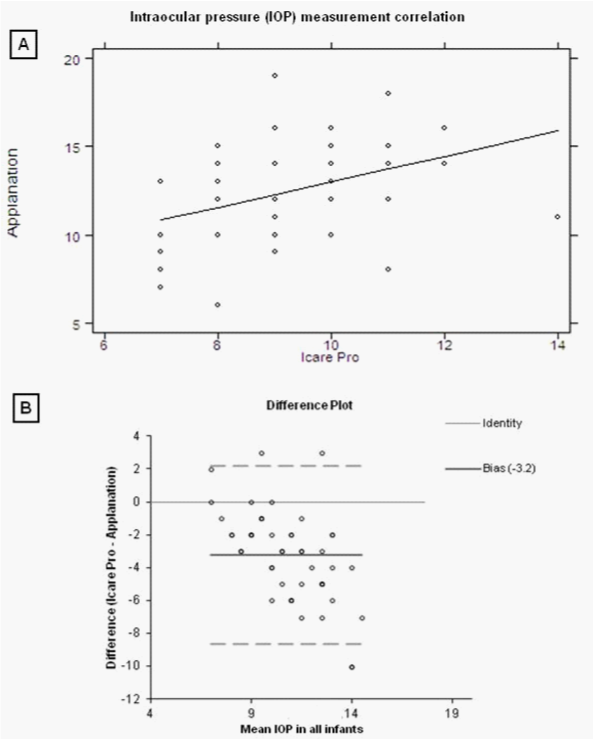
Our study included 50 consecutive infants with a mean age of 12.6 months, whose full demographics are summarised in table 1. Most eyes had no underlying pathology (n=24) with the remaining having a horizontal strabismus. Mean IOP was 12.4 mmHg when measured with PAT and 9.18 mmHg with ICPRO. A moderate correlation between the two methods of IOP measurement was found (r=0.384, p=0.0059, (Figure 1A)), with the IOP difference±3mmHg in 58%. However ICPRO underestimated IOP in 88% of cases, by an average of 3.22 mmHg (SD=2.76–2.44, p<0.001). A Bland-Altman plot comparing PAT and ICPRO showed the 95% limit of agreement to be -8.6 to 2.2 mmHg, with the difference constant over the range of IOPs measured (Figure 1B). A summary of the Bland-Altman analyses performed is provided in Table 2.
Variable |
Mean ±SD (range) |
Age (months) |
12.64± 5.33 (3–21) |
PAT IOP (mmHg) |
12.4±2.95 (6–19) |
ICPRO IOP (mmHg) |
9.18± 1.57 (7–14) |
Diagnosis (n) |
24 |
Normal |
19 |
Esotropia |
7 |
Exotropia |
Table 1 Patient demographics (n = 50)
IOP, intraocular pressure; PAT, perkins applanation tonometer; ICPRO, icare pro rebound tonometer
Group |
Bias (mmHg) |
95% Limit of Agreement |
All (n = 50) |
- 3.2* |
-8.6 to 2.2 |
Age <12 months (n = 24) |
- 1.5* |
-5.2 to 2.3 |
Age >12 months (n = 26) |
- 4.8* |
-9.5 to -0.2 |
Infants not crying (n = 31) |
- 2.0* |
-5.6 to 1.6 |
Infants crying (n = 19) |
- 5.3* |
-10.8 to 0.3 |
Table 2 Summary of Bland-Altman Anayses, Bias of intraocular pressure measurement with Icare Pro rebound tonometer compared to Perkins applanation tonometer
*p < 0.001
A subgroup analysis of infants under 12 months showed a small underestimation of IOP by the ICPRO of 1.5 mmHg (p=0.0012), with the Bland-Altman 95% limit of agreement -5.2 to 2.3 (Figure 2A). The underestimation of IOP was greater in infants over 12 months at 4.8 mmHg (p<0.0001), with the Bland-Altman 95% limit of agreement -9.5 to 0.2 (Figure 2B). Recording IOP of infants with the ICPRO was well tolerated with only 6% crying during measurement, all of whom were also crying during PAT measurement. There were significantly more infants who became distressed during IOP recording with the PAT at 62% (p<0.0001). Subgroup analysis of infants that were not crying found the ICPRO to underestimate IOP by 2.0 mmHg (p<0.0001), with the Bland-Altman 95% limit of agreement -5.6 to 1.6 (Figure 3A). In infants who were crying during IOP measurement, there was a difference of 5.3 mmHg, with the Bland-Altman 95% limit of agreement -10.8 to 0.3 (Figure 3b). The underestimation of IOP with the ICPRO was significantly greater in infants who were crying (3.30 mmHg, SD 4.00 – 2.76, p<0.0001).
It is important that IOP measurement in paediatric ophthalmology is both accurate and well tolerated. Although our data show that IOP measurement with ICPRO was correlated with PAT, the ICPRO significantly underestimated IOP. The IOP was underestimated in the overall cohort, but this was also the case, irrespective of the age being above or below 12 months, or if the infant was crying or not. The use of ICPRO however, did appear to be better tolerated by infants than PAT. A number of previous studies have evaluated the accuracy of the original Icare rebound tonometer in children. They all found there to be an overestimation of IOP compared with applanation tonometry.15−17 The only published studies evaluating the ICPRO were on adults, which in contrast found less difference in IOP measurement12 and slight underestimation of IOP compared to applanation tonometry.11 The larger average underestimation of ICPRO IOP by 3.22mmHg from our data is likely to be due to the population studied and their inherently different corneal properties such as corneal thickness and elasticity.18−20 Such a disparity is unlikely to be highly clinically significant when the IOP is well within the normal range, given that Goldmann applanation tonometry in adults has inter-observer variability of over 2.5 mmHg.23 However, caution should be taken in cases with higher IOPs where the ICare Pro could potentially underestimate true IOP by much more.
One other study to date has evaluated rebound tonometry in infants. Lundvall et al used the original Icare in healthy infants aged between 3 to 18 months and found low inter-observer variation, ease of use and good tolerability by infants, but did not compare it to other methods of tonometry.24 The significantly greater underestimation of ICPRO IOP when the infants were crying could in fact be a falsely elevated PAT IOP owing to distress caused by instillation of the anaesthetic drop and the more lengthy and demanding technique involved in obtaining the PAT recording. The ICPRO may be a more accurate reflection of true IOP because the quicker measurement allows less opportunity for the infant to squeeze and create artifactually high IOP during applanation.
IOP measurement using ICPRO was well tolerated by the infants in our study and has been found to be better tolerated by children than applanation tonometry in a number of previous studies.25,26 This together with its superiority over previous versions in being able to measure IOP on supine patients, gives it advantage when assessing patients in intensive care units, those unable to cooperate and in trauma cases where applanation tonometry may be considered relatively contraindicated. This study, although simple and on a relatively small cohort of patients, provides the first data on a patient group where the ICPRO has the potential to be of significant clinical benefit. The data represents a real clinical setting and is therefore directly applicable to clinical use in screening IOP in infants. It is limited by the unmasked nature of the data collection where PAT IOP recording could be biased when the examiner is already aware of the ICPRO reading. However, corroboration of our findings of underestimation of IOP with the ICPRO could lead to a useful correction factor to give an applanation tonometry equivalent IOP for infants. Future work is also needed to compare the ICPRO and PAT on anaesthetized children where the effect of patient distress during IOP measurement can be eliminated. Further validation of the ICPRO in infants with abnormal IOP is also required.
In conclusion, the ICare PRO rebound tonometer appears to be safe, tolerable and of adequate accuracy for screening of IOP in infants. However, when high IOPs are recorded with the ICPRO, the true IOP may be significantly underestimated. Until more data is available about the use of the IcarePro in the paediatric population we suggest that it may be prudent to confirm the IOP measurement in such patients in a clinical setting that would enable accurate applanation tonometry.
None.
The authors have no proprietary interests or research funding relevant to this research and this work has not previously been presented at any meetings.

©2014 Rodrigues, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.