Advances in
eISSN: 2377-4290


Purpose: To report the efficacy and safety of suprachoroidal injection of triamcinolone acetonide using custom made needle in a 76 years old male with macular edema due to retinal vein occlusion.
Methods: This was interventional case report, of a symptomatic 76years’ old male with history of macular edema due to branch retinal vein occlusion, underwent for clinical examination and follow up with optical coherence tomography at baseline, one week, 4weeks and 8 weeks of treatment with single injection of triamcinolone acetonide in suprachoroidal space using custom made needle with evaluation efficacy and possible of ocular complications.
Results: Improvement of best corrected visual acuity from baseline 20/100 to 20/30 at 8 weeks with resolution of macular edema and intraretinal hemorrhages at 8weeks from baseline with no signs of ocular complications.
Conclusions: Injection of triamcinolone acetonide in suprachoroidal space using custom made needle seems to be safe and effective in treatment macular edema due to retinal vein occlusion.
Retinal vein occlusion is a common vascular disorder which may lead to blindness, retinal vein occlusion can be classified as branch, central, ischemic or non-ischemic, vein blockage can lead to high pressure in the capillaries causing hemorrhage and formation of cystoid macular edema which is the most common cause of onset of visual reduction from retinal vein occlusion.
Treatment of cystoid macular edema secondary to retinal vein occlusion used to be laser1 then intravitreal triamcinolone was used for the treatment2 today monthly intravitreal vascular endothelial growth factor blockage agents are used as first line therapy, as they showed efficacy and safety,3 however there are intravitreal steroids (dexamethasone or fluocinolone) in a form of implants (slow release devices) has shown durability and efficacy4 especially in cases where intravitreal VEGF blockage agents are contraindicated or non-responsive mainly in cases where inflammatory cascade is the main pathological element.
In this case report a suprachoroidal injection of 0.05 ml triamcinolone acetonide using custom made needle was used to treat cystoid macular edema secondary to retinal vein occlusion in a 76years old male.
A 76years old male presented to Marashi Eye Clinic with complains of onset reduction of vision in his left pseudophakic eye 4 days ago, his best correct visual acuity 20/80 at presentation with a referaction -1.00/-0.50 x 18 where his right eye has no light perception due to uncontrolled glaucoma, although patient did not report any family history but he reported an ischemic stroke 45days ago and myocardial infarction 2years ago. His anterior chamber examination was unremarkable with no signs of rubeosis iridis with well centered intra ocular lens, on gonioscopy the angle was open with no signs of neovascularization, blood in schlemm's canal or fibrosis, intraocular pressure was 19. The funds examination showed scattered clusters of dot and blot hemorrhages limited to the inferior half of the posterior pole with significant cystoid macular edema and some hard drusen, and the diagnosis was made of branch retinal vein occlusion with cystoid macular edema; the optic disc shows cup to disc ratio up to 0.8.
Optical Coherence Tomography (OCT) shows hyperreflectivity of inner retinal tissues along with empty cystic formation, increased central macular thickness and disturbed ellipsoid zone; RPE shows formation of hard drusen.
The OCT of the optic disc in map and circle scans shows reduction of ganglion cell complex volume along with reduction of RNFL thickness in superior, inferior, and nasal to the optic disc.
A discussion was made with patient and his sibling about the pros and cons of each treatment plan including monthly intravitreal injection of antiVEGF, intravitreal injection of triamcinolone as intravitreal dexamethasone implant is not affordable, or suprachoroidal triamcinolone and the latter was chose for the treatment, and informed consent was signed, the suprachoroidal was administered under sterile conditions, by disinfecting the skin using povidone iodine 10% and the conjunctiva using povidone iodine 4% under topical anesthesia , the injection is carried out after placing sterile drape and lid speculum isolating eye lashes in the superior temporal quadrant, injection site was measured with calipers 4 mm from the limbus, using needle which is custom made 30 gauge with a stopper (23 gauge sleeve) will only let 1000 micron penetration of the needle in to the sclera (Figure 1) which help 0.05 ml triamcinolone to reach the suprachoroidal space with gentle pressing on the sclera.
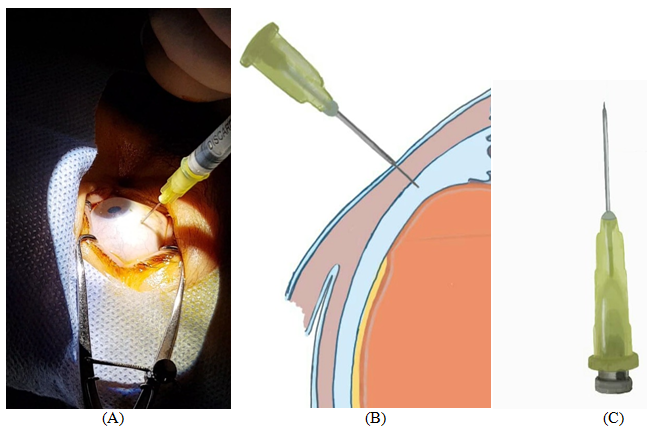
Figure 1 (A) Injection technique B) schematic shows how the needle is only penetrating the suprachoroidal space (C) showing the needle itself with stopper installed.
Intra ocular pressure was 18 after the injection and remained stable without increased spikes at 4weeks observation, best corrected visual acuity was improved to 20/50 in the 1st week, 20/40 at 4th week, and 20/30 at 8 weeks.
OCT showed remarkable thickness reduction within 48hrs with minimal cystic formation and improved hyperreflectivity in inner retinal layers, however OCT after 4weeks shows improvement in retinal structural changes with normal thickness, while results maintained in the 8weeks (Figure 2).
OCT of the optic disc map and circle shows no significant changes in term of ganglion cell volume and thickness of the RNFL (Figure 3).
Fundus exam shows reduction of scattered clusters of dot and blot hemorrhages without macular edema, with 8weeks there was no evidence of intraocular inflammation or toxicity nor systemic complication (Figure 4).
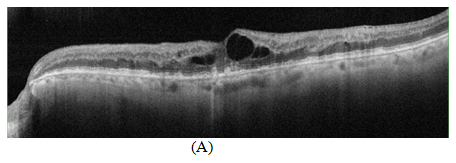

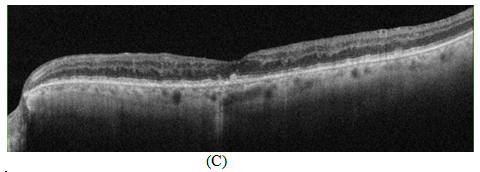
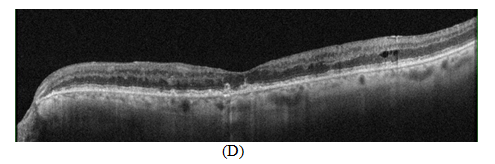
Figure 2 OCT scan showing (A) Cystoid macular edema with increased hyper reflectivity in the inner retinal tissue due to retinal vein occlusion with RPE alternation related to drusen formation (B) 48hrs later after suprachoroidal injection of triamcinolone acetonide showing decreased in central retinal thickness and resolution of cystic spaces (C, D) 4weeks and 8weeks OCT scans showing sustained effect of treatment with suprachoroidal triamcinolone acetonide.
Branch retinal vein occlusion can result from compression of the vein at the arteriovenous crossing and other factors like vascular wall degenerative changes or hematological diseases thus will lead to instability of fluid in vessels and disruption of blood retinal barrier, increasing production of VEGF and initiating inflammatory cascade inducing the formation of cystoid macular edema.5
Treatment of cystoid macular edema secondary to branch retinal vein occlusion usually done by monthly intravitreal injections of AntiVEGF,6 switching to intravitreal steroids7 recommended in cases where AntiVEGF is not responding or contraindicated especially in high risk patients such as ischemic stroke or myocardial infraction; however intravitreal steroids can cause increased intraocular pressure along with cataract formation.8
In this case patient is a high risk for vascular events if managed with monthly intravitreal injection of AntiVEGF as Branch Vein Retinal Occlusion happened 45days after ischemic stroke, the OCT scans of the optic disc shows compromised ganglion cell complex and RNFL so any intra ocular pressure spikes may lead to damage, and intravitreal triamcinolone may induce high intraocular pressure, while intravitreal dexamethasone implant may have a safer profile but they cannot be reimbursed by the patient.
Suprachoroidal space (SCS) rout to administer triamcinolone was elected in this case because does not increase the risk of vascular events, injection of triamcinolone in the SCS may reduce the risk of inducing spikes of high intraocular pressure because triamcinolone is directed to the choroid and retina which enhance it efficacy and may not interfere with angle of the anterior chamber in contrast to intravitreal rout where only 40 % of the triamcinolone is directed in the retina where rest of triamcinolone is disseminated in to the crystalline lens and anterior chamber angle.9
A study conducted on injection of intravitreal aflibercept in a combination with injection triamcinolone acetonide in suprachoroidal space or intravitreal aflibercept alone to treat macular edema secondary to retinal vein occlusion and concluded that combination of both intravitreal aflibercept and triamcinolone acetonide suprachoroidal was tolerated and required fewer injections of intravitreal aflibercept in 8 weeks follow up with better central retinal thickness and visual acuity outcome.10
Using custom made needle for Injection of triamcinolone acetonide in suprachoroidal space to treat macular edema due to retinal vein occlusion seems to be safe and effective however larger randomized trails should be conducted to evaluate durability and long term safety and efficacy.
None.
Author declares there is no conflict of interest towards the manuscript.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.