eISSN: 2577-8250


Review Article Volume 5 Issue 3
1Department of Biology, Faculty of Arts and Sciences, Adiyaman University, Turkey
2Department of Pharmaceutical Botanic, Faculty of Pharmacy, Adiyaman University, Turkey
3Gemlik Asim Kocabiyik Vocational School, Uludag University, Turkey
4Department of Biology, Faculty of Arts and Sciences, Marmara University, Turkey
Correspondence: Talip Şahin, Department of Biology, Faculty of Arts and Sciences, Adiyaman University, Turkey
Received: September 10, 2023 | Published: September 22, 2023
Citation: Şahin T, Kiliç O, Acar AG, et al. A review: the role of Streptococcus bovis in colorectal cancer. Art Human Open Acc J. 2023;5(3):165-173. DOI: 10.15406/ahoaj.2021.05.00203
Cancer, ranking second among the most commonly encountered diseases worldwide, is exhibiting an increasing incidence over time. Among cancer types, lung cancer, breast cancer, and prostate cancer hold the top three positions. Following these, digestive system cancers are the most frequently observed. The rising cancer-related mortality rates and potential difficulties during treatment exacerbate the fears and concerns of cancer patients. Throughout history, cancer has been attempted to be explained through theories such as lymphatic, humoral, blastoma, trauma, chronic irritation, and parasitic hypotheses. In contemporary times, a wealth of information exists concerning the roles of viruses and bacteria in cancer development. Among bacteria, the sole member acknowledged as a human carcinogen by the International Agency for Research on Cancer (IARC) is Helicobacter pylori. While there is no conclusive evidence regarding Streptococcus bovis's capacity to induce cancer, substantial suspicions surround this matter. This review delves into the relationship between the Streptococcus bovis group of bacteria, which is associated with cancer but not listed by the IARC, and colorectal cancer.
Keywords: streptococcus bovis, colorectal cancer, carcinogens
Throughout the history of medicine, the disease most extensively researched has been cancer. References to cancer and its fatal consequences are found in Babylonian cuneiform tablets, ancient Indian manuscripts, and Egyptian papyri, including the Ebers Papyrus (15th century BC).1 Today, it stands as a significant issue for all countries worldwide. The projection by the International Agency for Research on Cancer (IARC), a branch of the World Health Organization, for the year 2030 is that cancer will be the leading cause of death.2 Among the most commonly encountered cancer types globally, lung cancer, breast cancer, and prostate cancer hold the top three positions. Following these, digestive system cancers are the most prevalent. The increasing cancer-related mortality rates and potential difficulties during treatment amplify the fears and concerns of cancer patients.3 Between the years 1628 and 1694, the first scientific study on cancer was conducted by Marcello Malpighi. In the past, cancer was explained through theories such as humoral, lymphatic, blastoma, chronic irritation, trauma, and parasitic hypotheses.4 In 1903, Borrel proposed the idea that cancer could have a viral origin. In 1909, Ellerman and Bank demonstrated that leukemia is contagious among chickens, and in 1956, Dr. Stanley indicated that cancer viruses are present in every individual, and some factors trigger their activation. In 1974, Dr. Mak and Dr. Hawatson from the Ontario Cancer Institute in Canada discovered a virus that causes leukemia in humans.5
In recent years, studies have identified associations with viruses, protozoa, helminths, and certain bacteria in various cancer types. Up to this point, particularly in digestive system cancers, certain bacteria (such as Helicobacter bilis, Helicobacter hepaticus, Campylobacter jejuni, Clostridium spp., Bacteroides sp., Salmonella typhi), along with Streptococcus spp. bacteria, have been observed to actively contribute to colorectal cancer (CRC).5–8
Characteristic features of cancer cells include continuous division without dependence on any factor, resistance to growth-inhibitory signals, lack of response to apoptosis, maintenance of telomere sequences allowing unlimited replication, and the ability to migrate to other regions and generate disease.9 Globally, lung and prostate cancers alternately hold the first two places in the deadly ranking. CRC stands as the third most commonly occurring cancer type. Approximately 600,000 individuals worldwide die from colon cancer annually, accounting for around 8% of total cancer deaths worldwide. CRC primarily affects elderly adults; less than 20% of CRCs are found in individuals under 50 years old. However, there is a rapid increase in CRC among the young population, raising questions about lifestyle risk factors and even infectious etiology.10 The American Cancer Society found a yearly increase in CRC incidence of 2.9% in women and 3.5% in men worldwide from 1992 to 2005. In contrast, there is a 20% decrease in CRC incidence in populations over 50 years old. Overall, an individual's lifetime risk of CRC is approximately 5%.11
CRC includes adenocarcinomas, adenosquamous carcinomas, spindle cell carcinomas, squamous cell carcinomas, mucinous carcinomas, signet ring cell carcinomas, and undifferentiated carcinomas, each having distinct clinical characteristics and requiring slightly different treatment approaches.12 CRC formation generally evolves from a pre-existing polyp, progressing through stages of adenoma-dysplasia-carcinoma (Figure 1) and attains a lethal size within a span of 5 to 10 years.13 This timeline underscores CRC as one of the most preventable cancers through early detection. Particularly, the removal of high-risk polyps before malignant transformation significantly diminishes the risk of cancer.12,14

Figure 1 The CRC formation.15
Streptococci
More than 100 species have been identified within the Streptococcus genus to date. Especially with the advancement of next-generation sequencing technologies, this number is likely to increase further. In the past 5 years alone, over 10 new species have been discovered, primarily from the oral cavity and gastrointestinal system of mammals. Streptococci have the potential to cause invasive infections, many of which hold significant public health importance. They can be found as part of the normal flora in the body and can also exist as saprophytes in food items like milk and dairy products.16
Streptococci constitute a diverse group of microorganisms. They are generally characterized as gram-positive cocci, often appearing in chains or pairs. Unlike staphylococci, streptococci yield negative results in catalase reactions. Many of these microorganisms are facultative anaerobes, meaning they can thrive in both oxygen-rich and oxygen-poor environments. The medically significant streptococci are typically non-motile and lack spores, while some species are enveloped by structures such as capsules composed of polysaccharides or hyaluronic acid. These attributes make them amenable to cultivation in nutrient-enriched media, such as those containing blood or serum. In solid culture media, most of these microorganisms form disk-like colonies with diameters of 1-2 mm.17
Streptococcus species
Group A Streptococcus; Streptococcus pyogenes
Streptococcus bacteria are among the most common microorganisms causing diseases in humans. When grown on blood agar, they typically form small, grayish colonies with a wide zone of complete hemolysis (beta hemolysis) surrounding them. Among the cellular components of these bacteria, commonly encountered ones include lipoteichoic acid, M protein, capsular polysaccharide, streptokinase, streptolysin, nucleases, hyaluronidase, and erythrogenic toxins. Additionally, streptococci can produce various enzymes, including proteinases, phosphatases, esterases, amylases, N-acetyl glucosamine amidase, neuraminidase, lipoproteinases, ribonucleases, diphosphopyridine nucleotidase, and esterases.18
Infections caused by Group A Streptococcus can lead to various diseases such as erysipelas, sepsis, endocarditis, puerperal sepsis, toxic shock-like syndrome, skin and subcutaneous tissue infections, streptococcal pharyngitis, scarlet fever, acute rheumatic fever, and acute glomerulonephritis.19
Group B Streptococcus; Streptococcus agalactiae
B group streptococci, known as Streptococcus agalactiae, generally exhibit characteristics typical of the Streptococcus family. When cultured on blood agar, they form larger, purplish colonies compared to A group streptococci and often have a narrow hemolysis zone around them. However, 5% to 15% of them may not exhibit hemolysis. B group streptococci are commonly found in the genital and intestinal flora of humans, particularly in pregnant individuals, nursery personnel, and newborns, without causing disease.20
Infections caused by these bacteria can include sepsis, pneumonia, osteomyelitis, arthritis, and meningitis in newborns. In adults, they can lead to conditions such as endometritis, endocarditis, pyelonephritis, pneumonia, cellulitis, septic arthritis, and meningitis. B group streptococci present in the vaginal flora of women can also be found in 50% of their partners' urethra, potentially causing urethritis.21
Group C Streptococci
Group C Streptococci consist of species like S. equisimilis, S. zooepidemicus, beta-hemolytic S. equi, and S. dysgalactiae, which can also be alpha-hemolytic or non-hemolytic. These bacteria can be responsible for diseases such as pharyngitis, tonsillitis, sepsis, pneumonia, meningitis, osteomyelitis, arthritis, and endocarditis.22
Group D Streptococci
Their microscopic appearance can be in pairs or short chains of diplococci. Enterococcus faecalis, Enterococcus faecium, and Streptococcus durans are microorganisms that are resistant to penicillin and exhibit alpha, beta, or gamma hemolysis characteristics. Streptococcus bovis and Streptococcus equinus, on the other hand, are non-enterococcal Group D streptococci.23
Group D streptococci and enterococci are often part of the normal flora in the intestines, mouths, and sometimes the skin of both humans and some animals. Under suitable conditions, they can cause various diseases in humans, including endocarditis, urinary tract infections, abscesses, and cholecystitis.24
Viridans Streptococci
Viridans group streptococci make up 30% to 60% of the normal oral flora in humans. These bacteria can be found on the surfaces of teeth, in the spaces between gums and teeth, in root canals, on the palate, tongue, and the mucous membranes of the pharynx. To cause infections, they need to leave their natural habitat and encounter reduced host resistance. They exhibit alpha hemolysis when grown on blood agar.25
Viridans streptococci can be isolated as the causative agents of dental root infections, subacute bacterial endocarditis, chronic lung infections, genital and urinary infections, cholangitis, peritonitis, and various abscesses.18,25
Other streptococcus species
Aerococcus
These bacteria have a size of 1.0-2.0 micrometers and form tetrads. They are gram-positive and microaerophilic, being catalase-negative. When grown on blood agar, they exhibit green hemolysis. Aerococcus is generally considered a saprophytic bacterium and has been isolated from certain cases of endocarditis and hospital environments.18
Gemella
Gemella bacteria, on the other hand, appear as diplococci or single cocci with flat, opposing faces. They are gram-positive and form colonies on blood agar that resemble small beta-hemolytic streptococcal colonies. This genus includes only one species, Gemella haemolysans.18
Peptococcus
Peptococcus bacteria are gram-positive cocci that appear singly, in pairs, tetrads, or clusters with a size ranging from 0.3 to 1.3 micrometers. Their colonies on blood agar are small (0.5 mm in diameter), round, shiny, and non-hemolytic, with a black color. Peptococcus niger can be part of the normal flora in the vaginal area and the umbilical region.18
Peptostreptococcus
Peptostreptococcus bacteria, forming irregular clusters or chains of cocci, are gram-positive anaerobic cocci. Isolating Peptococcus and Peptostreptococcus species can be challenging. Particularly, the presence of gram-positive cocci in direct stained preparations of clinical specimens without growth in aerobic cultures, but with suspected gram-positive cocci in anaerobic cultures, indicates the presence of these bacteria.26
Peptostreptococcus species can be isolated from various sources, including the female genital tract in both normal and pathological conditions, blood in cases of puerperal sepsis, the normal respiratory and intestinal flora of humans and some animals, some pyogenic infections, septic war wounds, and appendicitis.27
Classification of species within the streptococcus genus
Species within the Streptococcus genus are classified based on various factors. These factors encompass serological characteristics of the cell wall, the presence of capsular antigens, colony morphology, types of hemolysis exhibited on blood agar, biochemical reactions, and sensitivity to physical/chemical agents.28
Streptococcus bovis
These bacteria, referred to as S. bovis, are categorized as Streptococcus gallolyticus (formerly S. bovis biotype I), Streptococcus infantarius (S. bovis biotype II.1), and Streptococcus pasteurianus (S. bovis biotype II.2). These bacteria typically form small non-hemolytic or alpha-hemolytic colonies, and they cannot grow in a 6.5% salt solution. They yield negative results in the catalase test, test positively for leucine aminopeptidase, and test negatively for PYR (pyrrolidonyl arylamidase). Gram-positive cocci in pairs or short chains define their morphology.29
These species can be found as normal components of the intestinal flora in about 2-12% of healthy adults. However, they are also prevalent in individuals with gastrointestinal cancer. S. bovis group bacteria express the Lancefield Group D antigen and exhibit phenotypic characteristics reminiscent of enterococci.30 The exact pathogenesis mechanism of S. bovis is not fully understood, and thus, the virulence factors associated with these infections are not completely elucidated. The following are some factors that contribute to the survival and growth of this organism within the host:31–33
Many Streptococcus species do not thrive well in traditional growth media like Nutrient Agar, necessitating the use of specialized media containing specific carbohydrates and nutrients. Blood agar and Chocolate agar are commonly used for diagnosing S. bovis, with observation of hemolysis patterns being essential. For more selective isolation, specialized media like Brain Heart Infusion Agar and Trypticase Soy Agar/Broth with defibrinated sheep blood can be utilized (Figure 2).34
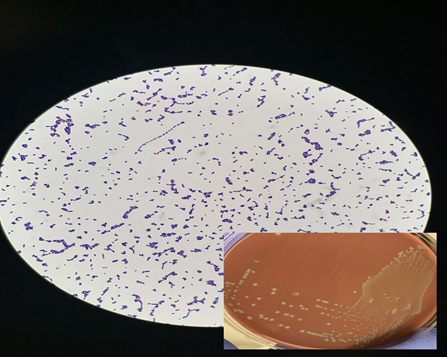
Figure 2 Microscopic images of Streptococcus bovis on blood agar.35
Pathogenesis of Streptococcus bovis
While Streptococcus bovis is considered a commensal in the gastrointestinal system of animals, it can also lead to various forms of infection. The complete mechanism of infection and the pathogenesis of the disease are not fully understood. However, it is believed that biofilm formation plays a critical role in the organism's growth and survival. Different virulence factors expressed by this organism assist in the pathogenesis process.36,37
Clinical symptoms of streptococcus bovis
Previously, S. bovis was commonly considered a low-grade pathogen associated with bacteremia and endocarditis. In addition to infective endocarditis, the possibility of S. bovis infections has been suggested in various areas such as osteomyelitis, discitis, and neck abscesses outside the colon. Depending on the geographical location of patients, other types of infections such as colorectal adenoma, cholecystitis, cholangitis, and biliary tract diseases have also been linked to S. bovis.38,39
S. bovis and colorectal cancer
S. bovis is a largely commensal species found in the intestines of animals, and it serves as a benign type that helps prevent the colonization of different pathogenic microorganisms. However, in individuals with suppressed immunity, it possesses various structures and proteins that support the colonization and invasion of host tissue surfaces. These structures not only aid the organism in entering the body and initiating invasion but also protect the organism from the host immune system.40
The S. bovis group bacteria have long been associated with colorectal cancer (Table 1). Meta-analyses conducted have revealed that patients infected with S. bovis biotype I have a significantly higher risk of developing colorectal cancer compared to those infected with biotype II. This analysis demonstrates that S. bovis is not just a bacterium, but it plays a role in the formation and progression of CRC and certain diseases. However, research has not yet determined whether S. bovis is a causative agent of colorectal cancer or if it aids in the growth and proliferation of pre-existing cancer within the lumen of the colon. Nonetheless, there is a clear established link between this group and colon adenomas/carcinomas based on studies conducted to date.41,42
|
Disease type |
Patient number |
Methods |
Results |
Reference |
|
CRC |
63 |
Facklam method |
% 58 positive |
43 |
|
CRC |
12 |
İmmunocapture mass spectrometry assay |
% 91 positive |
44 |
|
CRC |
64 |
Colonoscopy, Elisa and in situ hybridization experiment |
% 18-62 positive |
45 |
|
CRC |
82 |
Elisa |
% 60 positive |
46 |
|
Rat - colon cells |
- |
Elisa |
Increase in IL-8 production |
47 |
|
Rat - colon cells |
- |
Elisa, Western blot, Cell culture |
Induction of IL-8, PGE2 and COX-2 expression, promotion of neoplastic lesions |
48 |
|
Colorectal lesıons |
41 |
PCR |
% 8 positive |
49 |
|
CRC |
29 |
PCR |
% 7 positive |
50 |
|
CRC |
12 |
Tube dilution technique |
% 42 positive |
51 |
|
Colorectal lesıons |
34 |
Colonoscopy |
% 9 positive |
52 |
|
Colorectal and duodenal cancer |
16 |
- |
% 6 positive |
53 |
|
Colorectal |
14 |
Antibiotic susceptibility test |
% 29 positive |
54 |
|
neoplasia. |
||||
|
CRC |
21 |
Insulation |
% 5 positive |
55 |
|
CRC and Colorectal |
25 |
Serological methods |
% 8 positive (CRC) |
56 |
|
neoplasia. |
% 28 positive (Colorectal neoplasia) |
|||
|
CRC and Colorectal |
92 |
Serological methods |
% 17 positive (CRC) |
57 |
|
neoplasia. |
% 24 positive (Colorectal neoplasia) |
|||
|
CRC |
32 |
Serological methods |
% 9 positive |
58 |
|
CRC |
53 |
Colonoscopy and serological methods |
% 4 positive |
59 |
|
CRC |
20 |
Microbiological methods |
% 15 positive |
60 |
|
CRC |
20 |
Microbiological methods, Fisher's exact probability test and Mann-Whitney U test |
% 15 positive |
61 |
|
CRC |
40 |
Colonoscopy and serological methods |
% 10 positive |
62 |
|
CRC |
12 |
PCR |
% 17 positive |
63 |
|
Colorectal |
20 |
Colonoscopy and serological methods |
% 77 positive |
64 |
|
neoplasia. |
||||
|
CRC |
37 |
Microbiological methods |
% 11 positive |
65 |
|
CRC |
199 |
Microbiological methods |
% 46,7 positive |
66 |
|
CRC |
17 |
Microbiological methods and Fisher's exact probability test |
% 17 positive |
67 |
|
Colorectal lesions |
19 |
Agar dilution method |
% 47 positive |
68 |
|
CRC |
46 |
Microbiological methods |
% 13 positive |
69 |
|
CRC |
20 |
Microbiological methods |
% 45 positive |
70 |
|
CRC |
59 |
Microbiological methods |
% 18 positive |
71 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
72 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
73 |
|
CRC |
2 |
Microbiological methods |
% 100 positive |
74 |
|
CRC |
2 |
Microbiological methods |
% 100 positive |
75 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
76 |
|
CRC and Colorectal |
3 |
Microbiological methods |
% 100 positive |
77 |
|
neoplasia. |
||||
|
CRC |
4 |
Colonoscopy, barium enema and proctoscopic examination |
% 25 positive |
78 |
|
CRC |
6 |
Sigmoidoscopy, colonoscopy, barium enema |
% 83,3 positive |
79 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
80 |
|
CRC |
1 |
Colonofiberoscopy |
% 100 positive |
81 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
82 |
|
Colorectal lesions |
1 |
Microbiological methods |
% 100 positive |
83 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
84 |
|
Colorectal |
1 |
Microbiological methods |
% 100 positive |
85 |
|
neoplasia. |
||||
|
CRC |
1 |
Microbiological methods |
% 100 positive |
86 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
87 |
|
CRC |
3 |
Microbiological methods |
% 100 positive |
88 |
|
Colorectal |
1 |
Colonoscopy and microbiological methods |
% 100 positive |
89 |
|
neoplasia. |
||||
|
CRC |
1 |
Colonoscopy and microbiological methods |
% 100 positive |
90 |
|
CRC |
1 |
Colonoscopy, biopsy and microscopic examination |
% 100 positive |
91 |
|
CRC |
1 |
Colonoscopy and microbiological methods |
% 100 positive |
92 |
|
CRC |
1 |
Colonoscopy and microbiological methods |
% 100 positive |
93 |
|
CRC |
20 |
Retrospective research in hospital records |
% 67 positive |
94 |
|
CRC |
1 |
Microbiological methods |
% 100 positive |
95 |
|
CRC |
2 |
Clinical findings, standard radiographs and laboratory tests |
% 100 positive |
96 |
|
CRC |
15 |
Colonoscopy and microbiological methods |
% 20 positive |
97 |
|
CRC |
1 |
Colonoscopy, biopsy and microbiological methods |
% 100 positive |
98 |
|
Colorectal |
18 |
Colonoscopy and biopsy |
% 100 positive |
99 |
|
neoplasia. |
||||
|
CRC |
1 |
Computed tomography and microbiological methods |
% 100 positive |
100 |
|
CRC |
1 |
Colonoscopy and microbiological methods |
% 100 positive |
101 |
|
CRC and Colorectal |
25 |
PCR, colonoscopy and microbiological methods |
% 65 positive |
102 |
|
neoplasia. |
||||
|
Rat - colon cells |
- |
PCR |
Increase in IL-6, Scyb1, Ptgs2, IL-1b, TNF and Ccl2 levels |
103 |
|
CRC |
48 |
Colonoscopy and microbiological methods |
% 64,6 positive |
104 |
|
CRC |
20 |
Colonoscopy, biopsy and microbiological methods |
% 35 positive |
105 |
|
CRC |
1 |
Positron emission tomography and microbiological methods |
% 100 positive |
106 |
|
CRC |
1 |
Colonoscopy and microbiological methods |
% 100 positive |
107 |
|
CRC |
35 |
Microbiological methods, PCR and Elisa |
% 37,1 positive |
108 |
|
CRC |
30 |
Colonoscopy and microbiological methods |
% 47 positive |
109 |
Table 1 Results of the S. bovis bacterial group in patients with CRC
Streptococcus bovis is a part of the normal intestinal flora in humans and animals, but it is also responsible for infectious diseases (10-15% of all bacterial endocarditis cases). Various cases of bacteremia and metastatic abscesses involving the spleen, liver, soft tissues, bone, meninges, and endocardium have been reported to be associated with S. bovis, particularly related to colonic diseases, digestive system disorders, and specifically colonic neoplasms or chronic liver diseases.110
Endocarditis and/or bacteremia caused by Streptococcus bovis serve as early indicators of possible colorectal cancer presence. While further studies are needed to determine the exact pathophysiology, clinicians should be aware of this association. Rigorous investigation for colon cancer is advised in all patients presenting with S. bovis endocarditis/bacteremia; such patients may also present with liver disease or, rarely, extracolonic malignancies.111
In conclusion, there is strong epidemiological evidence that S. bovis can contribute to colorectal cancer. Moreover, its frequent detection in the majority of CRC cases, its greater and/or higher concentration in malignant tissues compared to corresponding healthy tissues, its localization in tissue at every stage of cancer development, and its support of cancer progression indicate that it should be included in the list of infectious agents by the IARC.112
Furthermore, cases of infective endocarditis related to the bovis group have shown an increase in recent years.113 Considering this information, any patient with bovis group bacteremia should undergo echocardiography and colonoscopy, and biliary tract imaging should also be considered. In light of the above data, we believe that clinical trials should be included to explore effective antibiotics against bovis group bacteria in patients with CRC, before resorting to heavy treatments like chemotherapy.
None.
The authors declare that there is no conflict of interest.

©2023 Şahin, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.