eISSN: 2378-3176


Case Report Volume 8 Issue 3
Pediatric Nephrology Unit of Santa Casa de Belo Horizonte Hospital, Minas Gerais, Brazil
Correspondence: Maria Goretti Moreira Guimarães Penido, Rua Piauí, 420 Santa Efigênia, Belo Horizonte, Minas Gerais, Brasil, CEP: 30150-320
Received: May 23, 2020 | Published: June 12, 2020
Citation: Antunes MMO, Penido MGMG. Use of eculizumab in atypical uremic hemolytic syndrome in childhood: case report. Urol Nephrol Open Access J. 2020;8(3):75-78. DOI: 10.15406/unoaj.2020.08.00280
Hemolytic-uremic syndrome is a form of thrombotic microangiopathy and is currently classified in childhood as typical and atypical. The atypical hemolytic-uremic syndrome encompasses genetic mutations in factors that regulate the complement cascade or antibodies against factor H. The typical form is caused by Shiga-toxin-producing Escherichia coli. There is also secondary urethral hemolytic syndrome due to an infectious condition such as pneumonia by S. pneumoniae, autoimmune diseases and use of medications with calcineurin inhibitors. The best knowledge of the pathophysiology of atypical hemolytic-uremic syndrome allowed the use of Eculizumab, a monoclonal antibody that acts in the final phase of the complement cascade. The objective of this study was to report a case of a patient with atypical hemolytic-uremic syndrome using Eculizumab, who was suspended for lack of medication and presented recurrence of the disease.
Keywords: atypical hemolytic-uremic syndrome, thrombotic microangiopathy, eculizumab
TMA, thrombotic microangiopathy; TTP, thrombotic thrombocytopenic purpura; HUS, hemolytic-uremic syndrome; DIC, disseminated intravascular coagulation; aHUS, atypical HUS; TMA, thrombotic microangiopathy; CFH, including factor; THBD, thrombomodulin
Thrombotic microangiopathy (TMA) is characterized by the presence of thrombi in the microvasculature associated with microangiopathic hemolytic anemia, thrombocytopenia and varying degrees of organ dysfunction.1,2 It results in three known clinical entities: thrombotic thrombocytopenic purpura (TTP), hemolytic-uremic syndrome (HUS) and disseminated intravascular coagulation (DIC).1–3 Histologically, MAT presents with thickening of the small vessel wall, intraluminal platelet thrombosis and luminal obstruction.4 In TTP the most commonly affected organ is the brain, with its pathophysiology deficient in the activity of ADAMTS13, a protease that cleaves the Von Willebrand factor.4 DIC is an acquired syndrome characterized by uncontrolled activation of the coagulation cascade with formation of fibrin thrombi. It is often a clinical complication of trauma, sepsis or adverse obstetric events.4–6 HUS most commonly affects the kidney and is characterized by non-immune microangiopathic hemolytic anemia (anemia, increased reticulocytes, increased LDH, indirect hyperbilirubinemia, peripheral blood schizocytes, low haptoglobin and direct negative coombs test), thrombocytopenia and acute kidney injury.2,3 Historically, HUS was classified as typical and atypical. The typical form occurs more commonly in children, responds well to supportive therapy, has a rare recurrence and is preceded by bloody bacterial diarrhea usually associated with Shiga-toxin-producing Escherichia Coli (strain OH-157: H7).3,4 Atypical HUS (aHUS) encompassed all other causes, being responsible for 5-10% of cases, affecting children and young adults and with a worse prognosis5 evidenced by high morbidity and mortality in the acute phase of the disease, and by the evolution to end-stage renal disease in about 50% of cases.2,3
The main pathogenesis of HUS involves hyper activation of the alternative pathway of the complement system, with excessive C3 cleavage and, subsequently, formation of C5b (Membrane attack complex - CAM), leading to damage to endothelial cells, platelet recruitment and thrombus formation in renal microvasculature.2,7,8 After a better understanding of the pathogenesis of HUS, it is currently preferred to classify HUS into primary and secondary. Primary HUS is caused by genetic (familial) abnormalities in the factors that regulate the complement system, documented in up to 70% of patients, or also caused by antibodies against complement regulatory factors (for example, anti-factor H-CFH antibody).2,3 More recently, the term aHUS has been used as a synonym for primary HUS. Secondary HUS can be attributed to the use of drugs (calcineurin inhibitors, sirolimus, clopidogrel), infectious agents (Escherichia coli, Streptococcus pneumoniae, HIV, H1N1 influenza A), pregnancy, cobalamin deficiency, neoplasms, transplantation and autoimmune diseases.3,5,9
Eculizumab is administered intravenously with doses based on the patient's weight. Treatment consists of an induction phase and a maintenance phase with doses of medication every 15 days.10,11 However, the biggest question today would be the duration of treatment for these patients. In addition, administration of Eculizumab reduces the response to infections such as Neisseria meningitis and the estimated risk of meningococcal infection in patients treated with Eculizumab is 0.5% per year and the relative risk is 5,000 compared to the general population.12–14 Vaccination against Neisseria serogroups ACWY and B is mandatory before the start of Eculizumab. The use of prophylactic antimicrobial is indicated for two or three months after using the medication.13,14 Regarding the discontinuation of treatment, there is no study to base the discontinuation of the use of Eculizumab. The expert recommendation is that discontinuation can be considered on a case-by-case basis after at least 6-12 months of treatment and with at least 3 months of normalization of renal function, excluding children under 3 years.12,14
To report a case of a patient who presented recurrence of aHUS after discontinuing use of Eculizumab.
Previously healthy patient (HAF), 4 months old, admitted to the Pediatric Nephrology Unit on 03/02/2016, with maternal report of mucous skin paleness, associated with prostration with evolution of one week. Initial pediatric evaluation showed severe anemia (Hb: 4.5mg/dL), and thrombocytopenia. The child's mother denied the occurrence of fever, bleeding, vomiting, diarrhea, use of medication or other symptoms. On admission, he received red blood cells and the tests performed showed worsening of renal function, hyperuricemia and elevated lactic dehydrogenase (HDL). That time, the intravenous hydration and allopurinol has been started. On 03/06/16 a myelogram was performed which excluded hematological neoplastic disease (marked erythrocytic series hyperplasia and presence of deitropoiesis. Absence of cells foreign to the bone marrow. He evolved with elevated blood pressure levels, progressive worsening of renal function and thrombocytopenia in addition to worsening of anemia, use of Nifedipine and Furosemide was started on 03/17/2016, evaluated by the nephrology team and indicated the beginning of Eculizumab (Day 03/17/16) due to the suspicion of HUS.
After the dose of the medication, the patient presented stabilization of hemoglobin values, significant increase in platelets as well as improved renal function. ADAMTS13 dosing ruled out diagnosis of PTT. He received the second dose of Eculizumab on 03/24/16 and was discharged in excellent clinical condition. Since then, he has been followed up on an outpatient basis with the Pediatric Nephrology Clinic using Eculizumab 300 mg IV, every 15 days. Clinical and laboratory evolution remained favorable. However, in November 2017 the patient did not receive the usual doses of Eculizumab due to its unavailability on the market. During this period, he was followed up with serial laboratory tests, maintaining clinical stability and without worsening the renal function. However, on 06/02/18 he developed hematuria and he was treated for urinary tract infection with ceftriaxone during 5 days, in his hometown. Unfortunately, the patient maintained his symptoms and on 06/04/18 he was referred to the Pediatric Nephrology Unit again. At that time, he had severe anemia, thrombocytopenia and worsening kidney function. He required transfusion of red blood cells on 06/07/18 and received a dose of Eculizumab (Figures 1–3). In few days the patient recovered renal function, the anemia was corrected as well as thrombocytopenia. He was referred again to outpatient control with maintenance of the use of Eculizumab (Figures 1–3).
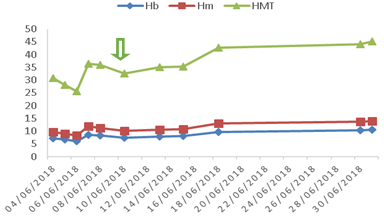
Figure 1 Recovery of hemoglobin (Hb), number of red blood cells (Hm) and hematocrit (HMT) after administration of Eculizumab. The arrow represents medication administration date.
HUS is characterized by nonimmune microangiopathic hemolytic anemia, thrombocytopenia and acute kidney injury.1,2 Its histological lesion is characterized by thrombotic microangiopathy (TMA), which preferentially affects renal vessels.3,7 The use of Eculizumab proved to be an effective therapy for remission of the disease, both in acute cases and in chronic patients with recovery of renal function. In the past decade, an increasing number of genetic abnormalities in components of the alternative complement pathway have been identified in patients with HUS, including factor H (CFH), factor I (CFI), membrane cofactor protein (MCP), factor B (CFB), C3, thrombomodulin (THBD) and CFH-related proteins 1 and 3 (CFHR1/CFHR3 del).1–3 One or more abnormalities in the complement system are found in 70% of patients with aHUS and 30% still remain without known changes.2 Unfortunately, for social and economic reasons, it was not possible to carry out this investigation in this patient. It is not necessary to identify the mutations for the diagnosis. It can be done with laboratory data of hemolytic anemia, thrombocytopenia (mild to moderate thrombocytopenia 30,000–150,000 mm3); elevated serum creatinine; ADAMTS13 activity >10%, as observed in the described case. Response monitoring should be performed with platelet counts, hemoglobin and LDH levels, which characterizes hematological remission. The haptoglobin levels generally remain decreased for long time and are not used to assess short-term response. It is recommended to continue treatment with Eculizumab indefinitely. The aforementioned patient keeps using Eculizumab in the recommended dose and is in good health. It can be suggested that the Eculizumab is able to maintain remission of the disease without the need for invasive procedures and should be maintained for long term.
HUSa is an odd disease. Its serious consequences reinforce the importance of early diagnosis and treatment. Clinical manifestations include the classic triad of microangiopathic anemia, thrombocytopenia and damage to other organs, where renal failure is the most common manifestation, often associated with other complications, such as neurological, cardiac and gastrointestinal complications. Mutations in the regulatory proteins of the complement system are recognized as the causes of this syndrome, however, they are not identified in all patients diagnosed with HUSa. Eculizumab (recombinant monoclonal antibody directed against fraction 5 of the complement) has been shown to significantly improve the prognosis and progression of the disease and is currently considered the first line of therapy. This medication can improve renal function and hematological changes and its use has changed the natural history of the disease, increasing both the success rate and the prognosis of this potentially fatal syndrome. According to this case report, the treatment with Eculizumab was able to control hematological changes, keeping the patient in remission. However, after the patient remaining for a long time without the medication, he evolved with recurrence of all signs and symptoms of the HUSa, demonstrating the need for chronic and prolonged use.
None.
The author declares there is no conflict of inter There are no conflicts of interest between the investigators and the patient, who authorized the reporting of his case by signing the free and informed consent form.
Mariana Marta de Oliveira Antunes and Maria Goretti Moreira Guimarães Penido were responsible for the research idea, study design, data acquisition, supervision and text writing.
None.

©2020 Antunes, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.