MOJ
eISSN: 2475-5494


Research Article Volume 5 Issue 5
Department of Emergency, Guangzhou Women and Children's Medical Center, China
Correspondence: Qiuyan Peng, Department of Emergency, Guangzhou Women and Childrens Medical Center, Guangzhou 510623, China
Received: June 28, 2017 | Published: July 19, 2017
Citation: Peng Q, Zhu C. Changes of phagocytic function of monocytes and neutrophils in critically Ill children. MOJ Womens Health. 2017;5(5):277-280. DOI: 10.15406/mojwh.2017.05.00132
Objective: To explore the changes of phagocytosis of monocytes and neutrophils in critically ill children.
Study design: 26 health children from child healthcare clinics were randomized chosen as the control group including 19 boys, 7 girls, aged from 8 months to 13years old), similarly 41 critically ill children admitted into PICU according to the admission criteria were enrolled in the study group including 29 boys, 12 girls, aged from 3 months to 11 years old, among of them, 27 patients suffered from various infective diseases, 14 patients didn’t, 9 patients died and 32 patients were survival finally. The specimens of 1ml heparinized anti-coagulated peripheral blood were drawn when the children were in clinic for health checkup, or within 24 hours without administration of any antimicrobial and immune agents after the children were admitted into PICU. Measured the phagocytosis rate and mean fluorescence intensity (MFI) of monocytes and neutrophils with flow cytometry after they phagocytized the flurescein-labelled opsonized E.coli. The reagent was produced by Pharma company. The Student’s t test was used to determine the differences between two groups. A 2-tailed probability value of <0.05 was considered statistically significant.
Results: The pagocytic rate and MFI of monocytes were less than that of neutrophils in both groups. Compared to the control group, the MFI of monocytes and neutrophils in the study group were significantly higher. Additionally, in the study group, all of survival and demised children, children with infective diseases or without, the MFI of monocytes and neutrophils were significantly advanced compared to that of the control group. However, there was no significant difference between survival and demised children, children with infective diseases and without in the phagocytosis rate and MFI of monocytes and neutrophils.
Conclusion: The phagocytosis of monocytes and neutrophils are significantly increased in a certain stage of critically ill children.
Keywords: monocytes, neutrophils, phagocytosis, mean fluorescence intensity (MFI), flow cytometry
Any microorganism and the human being are able to survive on the planet mainly attribute to the immune system, which acts as a critical guard defense host against the external invasive microbes, as well as internal apoptotic debris and inflammatory damage. It consists of physical barriers, innate response system and adaptive component.1 Among of them, the innate immunity plays a crucial role because of its immediate response to pathogens within minutes working as a “rapid response team” although the anatomical barriers provide the first line of defense,2 additionally it facilitates and control the activation of adaptive immunity, which can’t respond to a pathogen only after it has been recognized by the innate immune system.3 Both of the hematopoietic and non-hematopoietic cells contribute to the innate protection. The former includes monocytes/macrophages, neutrophils, dendritic cells, mast cells, eosinophils, natural killer cells and natural killer T cells. In addition, well recognized components such as C-reactive protein, LPS binding protein, complement proteins and defensins account for augmentation of innate immunity. The healthy status of body is the result of balance of interaction between microbes and essential elements of immune system. Any failure in this equilibrium such as immunosupression or hyperinflammation will lead to disease.
Phagocytosis by polymorphonuclear neutrophils and monocytes is well known as an key arm of host defense against bacterial or fungal invasion. The study in the phagocytic ability of monocytes and neutrophils has been conducting extensively in single disease for decades. In adult patients with periodontal disease,4,5 HIV-1 infection,6 myelodysplastic disorders,7 Alzheimer's disease,8 chronic granulomatous disease,9 or SLE5 the phagocytic function by monocytes or neutrophils was impaired or down-regulated, however some reports were controversial, with increase of phagocytosis by neutrophils.10-12 For pediatric population in critical condition, which is coexistence of multifactory impacts, children are more susceptible to multiple infections and development of generalized inflammatory response, which is associated with immunologic derangements with excessive activation or hyporesponsiveness in turn ineffective clearance of pathogen, even death. However researches addressing the phagocytosis by monocytes or neutrophils are scarce in children with critical illness.13 Our purpose is to investigate the innate immune function represented by phagocytic function of monocytes and neutrophils in critically ill children.
Participants
67 children from Guangzhou Women and Children’s Medical Center in 2007 were allocated randomizedly to the control and study group. The control group included 26 health children from child healthcare clinics, 19 boys, 7 girls, aged from 8 months to 13 years old). 41 critically ill children admitted into PICU according to the admission criteria were enrolled in the study group including 29 boys, 12 girls, aged from 3 months to 11 years old, among of them, 27 patients suffered from various infective diseases, 14 patients didn’t, 9 patients died and 32 patients were survival finally. The study was conducted following the obtained approval and under the auspices of the Ethical Committee of Guangzhou Women and Children’s Medical Center, and an informed consent for blood drawing was obtained from the parents of children.
Phagocytosis assay
The principle of the test is that leucocytes in whole blood could ingest the fluorescein (FITC)-labelled E. Coli, which can generate a green fluorescence signal, then quantified by cytometer. To investigate the phagocytic ability of the peripheral blood from two groups in vitro, we took advantage of the Phagotest kit from ORPEGEN Pharma, Germany (Batch number: 10-0100). The test was performed according to the instructions of the product. The steps were as follows briefly, 100ul heparinized whole blood was incubated with FITC-labelled E. Coli at 37°C for 10 minutes in a water bath, a negative control sample remained on ice. The amount of bacteria added in each sample, was calculated in order the ratio of bacteria to leucocytes to be 25:1. Stopped phagocytosis by placing the samples on ice at the end of the incubation. Added 100ul of quenching solution to eliminate the fluorescence of non-phagocytized bacteria. The cells were washed twice with 3 ml washing solution (5 min, 250 x g, 4 °C). Cells then were re-suspended and incubated for 20 min in 2ml prewarmed (room temperature) lysing solution for remove of erythrocytes and fixation of the leucocytes, discarded the supernatant. After once more wash with 3 ml of washing solution (5 min, 250 x g, 4°C), the cells were re-suspended in 200ul of DNA staining solution, mix and incubate 10 min on ice, to exclude aggregation artifacts of bacteria or cells, and measured the cell suspension within 60 min by flow cytometry.
Flow cytometry analysis
Measured the phagocytic ability of monocytes and neutropils using BD-FACSCalibur cytometer (Becton Dickinson, USA), indicated by phagocytic rate and mean fluorescence intensity.
Statistical analyses
Values were expressed as mean with standard deviation. The Student’s t test was used to determine the differences between two groups. A 2-tailed probability value of <0.05 was considered statistically significant. All statistical analyses were processed with SPSS15.0 software.
Phagocytic fucntion of monocytes and neutrophils in total population
Figure 1 shows in the total participants, the phagocytic rate and MFI of monocytes were significantly lower than that of neutrophils (P<0.01) (Figure 1).
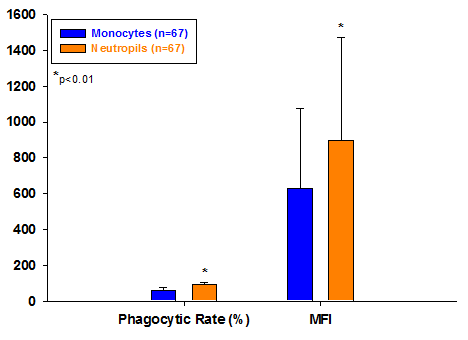
Comparison of phagocytic ability of monocytes and neutrophils in two groups
Compared to the control group, MFI of monocytes and neutrophils was higher in study group with significant difference (P<0.01), but not for phagocytic rate of neutrophils (P>0.05) (Figure 2).
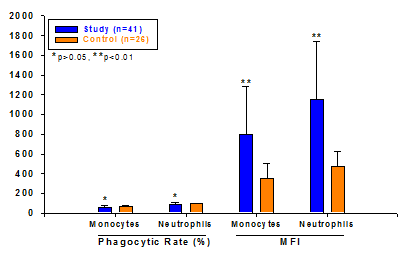
Comparison of phagocytosis of monocytes and neutrophils in demised and survival population to that of control group
We divided the study group into two subgroups, demised and survival group. MFI of monocytes and neutrophils in both subgroups was significantly greater than that of healthy children, there was no significant difference between two subgroups. However, the phagocytic rate of monocytes and neutrophils in two subgroups was lower without statistical significance (P>0.05) except for that of monocytes in survival group (P<0.05) compared with controls (Figure 3).
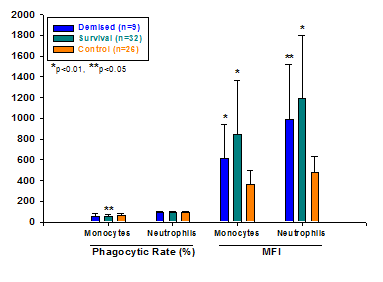
Comparison of phagocytosis of monocytes and neutrophils in infective and non-infective children to that of control group
Similarly, the study group was classified into infective and non-infective subgroups. Figure 4 shows MFI of both of monocytes and neutrophils was greater significantly compared to that of study groups (P<0.01). Both of the phagocytic rate and MFI of monocytes and neutrophils in infective group increased compared to non-infective group, but without significant difference (P>0.05).
In this study, we evaluated the phagocytic ability of monocytes and neutrophils in peripheral blood of critically ill children aged from 3 months to 13 years old. The study demonstrated the phagocytic function of monocytes and neutrophils was increased in children with critical illness compared to healthy group, and neutrophils had more powerful phagocytosis than monocytes. The recent data shows the evidence of immunosuppression or immunoparalysis in innate and adaptive immunity of population with sepsis or multiple organ failure,14-16 which is opposite to our result. Theoretically, it is reasonable that SIRS is the outcome of impairment of the immune system and disturbance between microbes and host. However, they share a common limitation with their exclusion criteria, we don’t know whether the population had been administrated by antimicrobial or immune agents prior to their test, and we are convinced that under the severe condition physicians had applied them. Besides, some patients involved in the study underwent blood or tissue sampling in the late stage of disease. In our study we obtained blood within 24 hours of ICU admission. As we know in early stage of sepsis prior to immunosuppression, there is a intense inflammatory response or “cytokine storm”,17,18 with early activation of immune cells followed by down-regulation with their activity,19 which is consistent with our result due to the probability of our test in early stage. The higher mortality with 22% (9 in 41) in our study maybe caused by the increased cytotoxic cell function20 even though we didn’t investigate the NK cells activity. Compared to the neutrophils which play a pivotal in the innate immunity, the monocytes had a weaker phagocytic ability due to their immaturity in peripheral blood before replacement for macrophages.
The phagocytic process mainly consists of chemotaxis, adhesion, ingestion and intracellular killing by oxygen dependent and oxygen-independent mechanism, there are amount of factors accounting for the innate immunity change. From the results with increase of MFI but not for phagocytic rate in study group, demonstrate ingestion of invasive microbes was not impaired, suggesting impact on mobilization of monocytes and neutrophils. Regarding to the comparison results of infective and non-infective population, the phagocytic rate was higher in infective group suggests monocytes and neutrophils are prone to be stimulated by external invasive microorganisms more than internal stress or inflammatory damage, but this remains to be defined with more sample size. Here some people will question about why the children with higher phagocytic ability suffered from critical illness and even death. Let us review the properties of innate and adaptive immunity. The cells responsible for innate immunity can only recognize the highly conserved structures present in a large group of microorganisms, however microbes are heterogeneous and can mutate at a very high rates, and patients can get nosocomial infection such as Staphylococcus aureus easily16 during hospitalization. Fortunately taking the advantage of stronger elimination of microbes, the adaptive component can localize the infection. But it requires 3 to 5 days for clonal expansion to produce sufficient effector cells, as well as the initiation and the recognization of innate immunity. Herein we can speculate there maybe some problems in intracellular killing by innate immunity and signaling or antigen presentation to adaptive immune system. Human leukocyte antigen (HLA)-DR is considered important for antigen presentation expressed on the monocytes surface, its reduction is associated with adverse outcome in adults.21
There are notable limitations in our study. Firstly, the effective phagocytosis relies on the sufficient effector cells, we didn’t test the leukocytes number and the individual percentage. Secondly, the immune response is variant with different stages of the disease, we just estimated it in the probable early stage. For the third one, the immune system is an integrity with the innate immune interplay of adaptive immune each other. It is not enough to detect phagocytic ability separately and isolatedly, it’s much better to monitor it consecutively and detect signaling molecules such as toll-like receptors engaged in interaction with adaptive immune system. All of the above limitations will be considered as our improvement in the further studies.
Despite these limitations, our study provides evidences showing the innate immunity is not initially decreased in critically ill children. Researches into continues change of innate immunity and the detail immunity defect should be preferable in future.
None.
The author declares no conflict of interest.

©2017 Peng, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.