MOJ
eISSN: 2577-8374


Research Article Volume 3 Issue 1
1Department of Mechanical Engineering, Graduate School of Engineering, Mie University, Japan
2Department of Chemistry for Materials, Graduate School of Engineering, Mie University, Japan
3School of Mechanical Engineering, The University of Adelaide, Australia
Correspondence: Akira Nishimura, Division of Mechanical Engineering, Graduate School of Engineering, Mie University, 1577 Kurimamachiya-cho, Tsu, Mie 514-8507, Japan, Tel +81 59 231 9747
Received: January 17, 2019 | Published: January 21, 2019
Citation: Nishimura A, Tatematsu D, Toyoda R, et al. Effect of overlapping layout of Fe/TiO2 on CO2 reduction with H2 and H2O. MOJ Solar Photoen Sys. 2019;3(1):1-8. DOI: 10.15406/mojsp.2019.03.00028
Fe-doped TiO2 (Fe/TiO2) film photocatalyst was prepared by sol-gel dip-coating, and pulse arc plasma process. The netlike glass fiber was used as a base material for the photocatalyst since it had a porous structure. The CO2 reduction performance with H2 and H2O into CO over the Fe/TiO2 photocatalyst was investigated. In addition, this study investigated overlapping two Fe/TiO2 coated on netlike glass fiber in order to utilize the light more effectively as well as increase the amount of photocatalyst for CO2 reduction. The characterization of prepared Fe/TiO2 film coated on netlike glass fiber was analyzed by SEM, EPMA, TEM, EDX and EELS. Furthermore, the CO2 reduction performance of the Fe/TiO2 film was tested under a Xe lamp with or without ultraviolet (UV) light, respectively. The results show that the CO2 reduction performance peaks under the condition of CO2/H2/H2O=1:1:0.5 in both cases with UV light and without UV light illumination. The highest concentration of CO with the Fe/TiO2 overlapped photocatalyst is 1.2 times of that with the single Fe/TiO2 photocatalyst. On the other hand, results also show the highest molar quantity of CO per weight of photocatalyst for Fe/TiO2 overlapped is almost half of single Fe/TiO2.
Keywords: photocatalyst, Fe-doped TiO2, CO2 reduction, reductants, overlapping effect
Due to mass consumption of fossil fuels, global warming and fossil fuels depletion have become serious global environmental problems in the world. After the industrial revolution, the averaged concentration of CO2 in the world has been increased from 278ppmV to 403ppmV by 2016.1 Therefore, it is necessary to develop a new CO2 reduction or utilization technology in order to recycle CO2. There are six vital CO2 conversions mechanisms: chemical conversions, electrochemical reductions, biological conversions, reforming, inorganic conversions, and photochemical reductions.2, 3 Recently, artificial photosynthesis or the photochemical reduction of CO2 to fuel (e.g. CO) has become an attractive route due to its economically and environmentally friendly behavior.2 TiO2 is the principle catalyst used for almost all types of photocatalysis reaction. It is well reported that CO2 can be reduced into fuels e.g. CO, CH4, CH3OH, and H2 etc. with TiO2 as the photocatalyst under ultraviolet (UV) light illumination.2,4−7 However, the CO2 reduction performance of TiO2 is still low since TiO2 has a large bandgap energy (~3.2 eV) and a rapid electron hole recombination rate.5 Recently, studies on CO2 photochemical reduction by TiO2 have been carried out from the viewpoint of performance promotion by extending absorption range towards visible region.8−15 N-doped reduced graphene oxide promotion,8 modified using Ce and La for TiO2 nanoparticles9, heterostructured g-C3N4/Ag-TiO2,10 noble metal doping such as Pt, Pd, Rh, Au, and Ag,11 nanocomposite CdS/TiO2 combining two different band gap photocatalysts,12 N2 modified TiO2,13 light harvesting complex of green plants assisted Rh-doped TiO2,14 and dye sensitized TiO2,15 have been attempted to overcome the shortcoming of the pure TiO2. They did improve the CO2 reduction performance, however, the amount of the products achieved in all the attempts were still unsatisfactory, ranging from 1mmol/g-cat to 350 mmol/g-cat.8−15 Therefore, a breakthrough in increasing the concentration level of products is necessary to advance the CO2 reduction technology in order to make the technology practically useful.
It was reported that doping transition metal was a useful technique for extending the absorbance of TiO2 into the visible region.16 For doping, various metal ions have been used, but among them, Fe3+ is considered as a strong candidate as it has a similar radius to Ti4+ (Fe3+ = 78.5pm, Ti4+ = 74.5pm)17 and can easily fit into the crystal lattice of TiO2.16,18,19 Moreover, the redox potential (energy differential) of Fe2+/Fe3+ is close to that of Ti3+/Ti4+, resulting in shifting its optical absorption into the visible region.16,18,19 The performance of Fe-doped TiO2 (Fe/TiO2) was reported for degradation of oxalic acid,20 rhodamine-B21 and methyl orange,22 resulting that the performance is superior to pure TiO2. Extending the absorbance of TiO2 into the visible region such as 600nm was also reported.18−20 However, the application of Fe/TiO2 for CO2 reduction is not investigated well compared to phtocatalytic degradation. Due to easy availability as well as above described characteristics, Fe is selected as the dopant in the present study.
Since a reductant is necessary for CO2 reduction to produce fuel, H2O and H2 are usually used as reductant according to the review papers.5,7 To promote the CO2 reduction performance, it is important to select the optimum reductant which provides the proton (H+) for the reduction reaction. The reaction scheme of CO2 reduction with H2O is as follows:23−25
<Photocatalytic reaction>
TiO2 + hn → h+ + e- (1)
<Oxidization>
H2O + h+ → ∙OH + H+ (2)
∙OH + H2O + h+ → O2 + 3H+ (3)
<Reduction>
CO2 + 2H+ + 2e- → CO + H2O (4)
The reaction scheme of CO2 reduction with H2 is as follows:26
<Photocatalytic reaction>
TiO2 + hn → h+ + e- (5)
<Oxidization>
H2 + 2h+ → 2H+ + 2e- (6)
<Reduction>
CO2 + e- → ∙CO2- (7)
∙CO2- + H+ + e- → HCOO- (8)
HCOO- + H+ → CO + H2O (9)
Though there are some reports on CO2 reduction with H2O or H2,5,7 the effect of combination of H2O and H2 is not investigated well. Though a few studies using pure TiO2 under CO2/H2/H2O condition were reported,26−28 the combined effect of ratio of CO2, H2 and H2O and metal doping on CO2 reduction performance were not investigated. Though Nishimura et al.29 reported the combined effect of ratio of CO2, H2 and H2O over Fe/TiO2 under the illumination condition with UV light, they did not investigate that under the illumination condition without UV light. To clarify the effect of doping Fe, it is important to examine the CO2 reduction performance under the illumination condition without UV light. In addition, the previous study29 did not investigate to promote the CO2 reduction performance for different ratios of CO2, H2 and H2O, e.g., by changing the amount of phototcatalyst or overlapping two photocatalysts. Therefore, the aim of the present study is to clarify the optimum ratio of CO2/H2/H2O for promoting the CO2 reduction performance with Fe/TiO2 photocatalyst.
In this study, TiO2 film is coated by sol-gel and dip-coating process on netlike glass fiber (SILIGLASS U, Nihonmuki Co.). The netlike glass fiber is a net composed of glass fiber whose diameter is about 10 mm. The fine glass fibers are knitted, resulting that the diameter of aggregate fiber is about 1mm. According to manufacturer’s specifications of netlike glass fiber, the porous diameter of glass fiber is about 1nm and the specific surface area is about 400m2/g. The netlike glass fiber consists of SiO2 whose purity is over 96 wt%. The aperture of net is about 2mm×2mm. Since the netlike glass fiber has a porous characteristic, it is believed that TiO2 film is captured by netlike glass fiber easily during sol-gel and dip-coating process. In addition, it can be expected a CO2 absorption performance of prepared photocatalyst is promoted due to the porous structure of netlike glass fiber.
Then, Fe is loaded on the TiO2 coated netlike glass fiber by pulse arc plasma method which can emit nanosized Fe particles uniformly by applying high electrical potential difference. The amount of loaded Fe can be controlled by pulse number. In this study, the pulse number is set at 100.
Since the netlike glass fiber has aperture of net, the light can pass through the netlike glass fiber. The present study has investigated overlapping two Fe/TiO2 coated on netlike glass fiber in order to utilize the light more effectively as well as increase the amount of photocatalyst for CO2 reduction. Though the effect of overlapping two Fe/TiO2 coated on netlike glass fiber on CO2 reduction performance with H2O was reported by Nishimura et al.,30 it was not investigated for CO2/H2/H2O. In addition, Nishimura et al.30 prepared Fe/TiO2 by sol-gel dip-coating process only. Since it is difficult to dope a metal on TiO2 uniformly by sol-gel dip-coating process,30 this study selects the pulse arc plasma process to dope Fe. It is believed that electron transfer between overlapping two Fe/TiO2 coated on netlike glass fiber is carried out.30 Therefore, it is expected that the CO2 reduction performance is promoted under many H+ production condition such as CO2/H2/H2O due to matching the electron according to Eqs. (1)-(9).
In this study, Fe/TiO2 prepared was characterized by Scanning Electron Microscope (SEM) and Electron Probe Micro Analyzer (EPMA), Transmission Electron Microscope (TEM), Energy Dispersive X-ray Spectrometry (EDX) and Electron Energy Loss Spectrum (EELS) analysis. The CO2 reduction performance with H2 and H2O using Fe/TiO2 coated on netlike glass fiber as photocatalyst under the condition of illuminating Xe lamp with or without UV light were investigated. The molar ratio of CO2/H2/H2O was set at 1:0.5:0.5, 1:1:0.5, 1:0.5:1, and 1:1:1 to determine the optimum ratio of CO2/H2/H2O. In addition, the effect of overlapping two Fe/TiO2 coated netlike glass fiber on CO2 reduction performance was investigated.
Preparation of Fe/TiO2 film
Sol-gel and dip-coating process was used for preparing TiO2 film. TiO2 sol solution was made by mixing [(CH3)2CHO]4Ti (purity of 95 wt%, Nacalai Tesque Co.) of 0.3mol, anhydrous C2H5OH (purity of 99.5 wt%, Nacalai Tesque Co.) of 2.4 mol, distilled water of 0.3 mol, and HCl (purity of 35 wt%, Nacalai Tesque Co.) of 0.07 mol. Netlike glass fiber was cut to disc, and its diameter and thickness were 50mm and 1mm, respectively. The netlike glass disc was dipped into TiO2 sol solution at the speed of 1.5mm/s and pulled up at the fixed speed of 0.22 mm/s. Then, it was dried out and fired under the controlled firing temperature (FT) and firing duration time (FD), resulting that TiO2 film was fastened on the base material. FT and FD were set at 623K and 180s, respectively. Fe was then loaded on TiO2 film by pulse arc plasma method. The pulse arc plasma gun device (ULVAC, Inc., ARL-300) having Fe electrode whose diameter was 10mm was applied for Fe loading. After the netlike glass fiber coated with TiO2 was set in the chamber of the pulse arc plasma gun device, where was vacuumed, the nanosized Fe particles were emitted from Fe electrode with applying the electrical potential difference of 200V. The pulse arc plasma gun can evaporate Fe particle over the target in the circle area whose diameter is 100 mm when the distance between Fe electrode and the target is 160mm. Since the distance between Fe electrode and TiO2 film was 150 mm, Fe particle can be evaporated over TiO2 film uniformly. The amount of loaded Fe was controlled by pulse number. In this study, the pulse number was set at 100. To promote the CO2 reduction performance, overlapping layout of two photoatalysts were investigated. The photocatalyst coated on both upper and lower surfaces of netlike glass disc was overlapped over the other photocatalyst coated on upper surface of netlike glass disc.
Characterization of Fe/TiO2 film
The structure and crystallization characteristics of Fe/TiO2 film were evaluated by SEM (JXA-8530F, JEOL Ltd.), EPMA (JXA-8530F, JEOL Ltd.), TEM (JEM-2100F/HK, JEOL Ltd.), EDX (JEM-2100F/HK, JEOL Ltd.) and EELS (JEM-ARM2007 Cold, JEOL Ltd.). Since these measuring instruments use electron for analysis, the sample should be an electron conductor. Since netlike glass disc is not an electron conductor, the carbon vapor deposition was conducted by the dedicated device (JEE-420, JEOL Ltd.) for Fe/TiO2 coated on netlike glass disc before analysis. The thickness of carbon deposited on samples was approximately 20-30nm.
The electron probe emits the electrons to the sample under the acceleration voltage of 15 kV and the current of 3.0×10-8 A, when the surface structure of sample is analyzed by SEM. The characteristic X-ray is detected by EPMA at the same time, resulting that the concentration of chemical element is analyzed according to the relationship between the characteristic X-ray energy and the atomic number. The spatial resolution of SEM and EPMA is 10mm. The EPMA analysis helps not only to understand the coating state of prepared photocatalyst but also to measure the amount of doped metal within TiO2 film on the base material.
The electron probe emits the electron to the sample under the acceleration voltage of 200 kV, when the inner structure of sample is analyzed by TEM. The size, thickness and structure of loaded Fe were evaluated. The characteristic X-ray is detected by EDX at the same time, resulting that the concentration distribution of chemical element toward thickness direction of the sample can be analyzed. In this study, the concentration distributions of Ti and Fe were analyzed.
EELS can be applied not only for detecting elements but also for determination of oxidation states of some transition metals. The EELS characterization was performed by JEM-ARM200F equipped with GIF Quantum having 2048ch. The dispersion of 0.5eV/ch can be achieved of the full width at half maximum of the zero loss peak.
CO2 reduction experiment
Figure 1 shows the experimental set-up of the reactor consisting of stainless pipe (100mm(H.)×50mm(I.D.)), a netlike glass disc coated with Fe/TiO2 film (50mm(D.)×1mm(t.)) which is located on the teflon cylinder (50mm (H.)×50mm (D.)), a quart glass disc (84mm(D.)×10mm(t.)), a sharp cut filter which cuts off the light of wavelength below 400nm (SCF–49.5C-42L, SIGMA KOKI CO. LTD.), a 150 W Xe lamp (L2175, Hamamatsu Photonics K. K.), mass flow controller, CO2 gas cylinder and H2 gas cylinder.
The reactor has volume of 1.25× 10-4m3, which is available for CO2. The light of Xe lamp, through the sharp cut filter and the quartz glass disc that are at the top of the stainless pipe, illuminates the netlike glass disc coated with Fe/TiO2 film, which is located inside the stainless pipe. The wavelength of light from Xe lamp is ranged from 185 nm to 2000nm. The Xe lamp can be fitted with a sharp cut filter to remove UV components of the light. With the filter, the wavelength of light from Xe lamp is ranged from 401nm to 2000nm. Figure 2 shows the light transmittance data of the sharp cut filter to prove the removal of the light whose wavelength is below 400nm. The average light intensity of Xe lamp on the photocatalyst without and with setting the sharp cut filter is 75.3 mW/cm2 and 68.2 mW/cm2, respectively.
In the CO2 reduction experiment with H2 and H2O, CO2 gas of 99.995 vol% purity and H2 gas of 99.99999 vol% purity were mixed in the buffer chamber, and introduced in the reactor which was pre-vacuumed by a vacuum pump. The mixing ratio of CO2 and H2 was controlled by a mass flow controller and confirmed by a TCD gas chromatogragh (Micro GC CP4900, GL Science) before introducing into the reactor. After confirming the mixing ratio of CO2 and H2, the distilled water was injected into the reactor through gas sampling tap and Xe lamp illumination was turned on at the same time. The molar ratio of CO2/H2/H2O was set at 1:0.5:0.5, 1:1:0.5, 1:0.5:1, and 1:1:1, respectively. The injected water vaporized completely in the reactor. Due to the heat of Xe lamp, the temperature in reactor was attained at 343K within an hour and kept at approximately 343K during the experiment.
The gas in the reactor was sampled every 24 hours during the experiment. The gas samples were analyzed by FID gas chromatograph (GC353B, GL Science) and methanizer (MT221, GD Science). Minimum resolution of FID gas chromatograph and methanizer is 1ppmV.
Characterization of Fe/TiO2 film
Figures 3 & 4 show EPMA images of TiO2 and Fe/TiO2 film, respectively. EPMA analysis was carried out for 1500 times magnification SEM images. In EPMA image, the concentrations of each element in observation area are indicated by the different colors. Light colors, for example, white, pink, and red indicate that the amount of element is large, while dark colors like black and blue indicate that the amount of element is small.
From these figures, it can be observed that TiO2 film with teeth like shape was coated on netlike glass fiber. It is also seen that TiO2 film builds a bridge among several glass fibers like reported also in.30 During firing process, the temperature profile of TiO2 solution adhered on the netlike glass disc was not even due to the different thermal conductivities of Ti and SiO2. Their thermal conductivities of Ti and SiO2 at 600 K are 19.4 W/(mK) and 1.82 W/(mK), respectively.31 Due to the thermal expansion and shrinkage around netlike glass fiber, thermal crack formed on TiO2 film. Therefore, TiO2 film on netlike glass fiber was teeth like. As to Fe, it is observed from Figure 3 that nanosized Fe particles loaded on TiO2 uniformly, resulting from that the pulse arc plasma method can emit nanosized Fe particles.
To evaluate the amount of loaded Fe within TiO2 film quantitatively, the observation area, which is the center of netlike glass disc, of diameter of 300 mm is analyzed by EPMA. The ratio of Fe to Ti in this observation area is counted by averaging the data obtained in this area. As a result, the weight percentages of elements Fe and Ti in the Fe/TiO2 film are 1.84 wt% and 98.16 wt%, respectively.
Figure 5 shows EDX images of Fe/TiO2. EDX analysis was carried out using 160000 times magnification TEM images. In these figures, the yellow circle indicates the existence of Fe. From this figure, it is observed that Fe particle is loaded on TiO2 film, and no Fe layer has formed. The average size of Fe particle for Fe/TiO2 loaded is approximately 28 nm where the longest length of Fe particle was used as a representative length.
Figure 6 shows EELS spectra of Fe in Fe/TiO2 film. From this figure, the peaks at around 708eV and 720eV can be observed. Compared to EELS data base,32 the EELS spectra of Fe2O3 (Hematite) matches with Figure 6. Therefore, Fe in Fe/TiO2 prepared presents as Fe3+ ion in Fe2O3. Since the doped Fe emits the electron during the photochemical reaction and the electron plays a part in the fuel production from CO2 under CO2/H2O condition23−25 and CO2/H2 condition,26 the ion number of Fe is important. For Fe/TiO2 photocatalyst, the incorporated Fe3+ ions can substitute the octahedrally coordinated Ti4+ ions in TiO2 lattice, which extends the photo-response of TiO2 to the visible light region and promotes the formation of electron-hole pair significantly.33 Since the Fe3+ ion acts as an electron-trapped agent, Fe/TiO2 exhibits a low recombination rate of electron-hole pairs and high separation efficiency.33
Effect of molar ratio of CO2, H2 and H2O on CO2 reduction characteristics of Fe/TiO2
Figures 7 and 8 show the concentration changes of CO produced and the molar quantity of CO per weight of photocatalyst in the reactor along the time under the Xe lamp with UV light on, respectively. The amount of Fe/TiO2 on the netlike glass disc is 0.10g. In this experiment, CO is the only fuel produced from the reactions. Before the experiments, a blank test, that was running the same experiment without illumination of Xe lamp, had been carried out to set up a reference case. No fuel was produced in the blank test as expected.

Figure 7 Comparison of concentration of CO among several moral ratios of CO2/H2/H2O under the illumination condition with UV light.
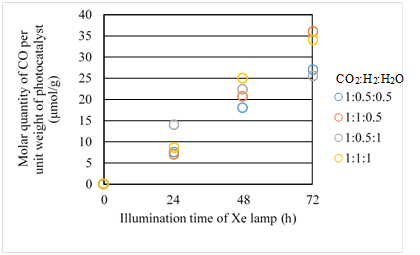
Figure 8 Comparison of molar quantity of CO per unit weight of photocatalyst among several moral ratios of CO2/H2/H2O under the illumination condition with UV light.
According to Figures 7 & 8, the CO2 reduction performance is the highest for the moral ratio of CO2/H2/H2O=1:1:0.5. Since the reaction scheme of CO2/H2/H2O is not clarified, this study refers to the reaction scheme of CO2/H2O and CO2/H2 as shown by Eqs. (1) – (9). It is known from Eqs. (1) – (9) that 2 H+ and 2 electrons are necessary for producing CO in the case of CO2/H2O as well as that of CO2/H2. In other words, the same number of H+ and electron is needed. According to Figure 6, Fe in Fe/TiO2 prepared is in the form of Fe3+ ion in Fe2O3. Therefore, it is believed that 3 electrons are emitted per Fe, resulting that the electron rich state is formed. According to Eqs. (2), (3) and (6), H+ is produced by the oxidization of reductant. Comparing H2O with H2, H2O is a weaker reductant,34 resulting that larger amount of H2 is necessary to match the electron rich state. On the other hand, the lowest CO2 reduction performance is obtained for the moral ratio of CO2/H2/H2O = 1:0.5:1 according to Figures 7 and 8. This result also shows that larger moral ratio of H2 compared to H2O is necessary for the better CO2 reduction performance of Fe/TiO2.
Figures 9 & 10 show the concentration changes of CO produced and the molar quantity of CO per weight of photocatalyst in the reactor along the time under the Xe lamp without UV light on, respectively. In this experiment, CO is the only fuel produced from the reactions.
According to Figures 9 & 10, the CO2 reduction performance is the best for the moral ratio of CO2/H2/H2O=1:1:0.5 in the case of illumination condition with UV light. It is considered that the same reaction mechanism as mentioned above is occurred. The order of CO2 reduction performance of Fe/TiO2 without UV light is the same as that with UV light. It is revealed that the wave length of light doesn’t influence on the CO2 reduction characteristics of Fe/TiO2 with H2 and H2O.
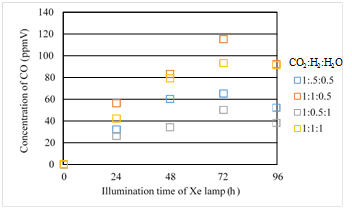
Figure 9 Comparison of concentration of CO among several moral ratios of CO2/H2/H2O under the illumination condition without UV light.

Figure 10 Comparison of molar quantity of CO per unit weight of photocatalyst among several moral ratios of CO2/H2/H2O under the illumination condition without UV light.
Effect of overlapping on CO2 reduction characteristics of Fe/TiO2
Figures 11 & 12 show the concentration changes of CO produced and the molar quantity of CO per weight of photocatalyst in the reactor along the time under the Xe lamp with UV light on, for Fe/TiO2 film coated on netlike glass disc overlapped, respectively. The total amount of Fe/TiO2 overlapped is 0.25g.
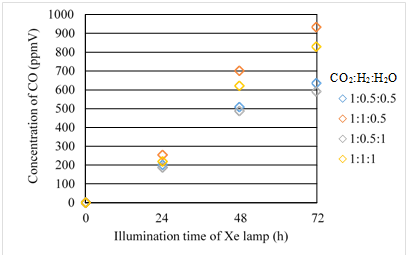
Figure 11 Comparison of concentration of CO for Fe/TiO2 overlapped among several moral ratios of CO2/H2/H2O under the illumination condition with UV light.
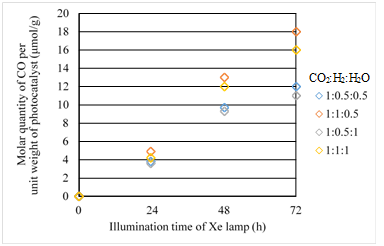
Figure 12 Comparison of molar quantity of CO per unit weight of photocatalyst for Fe/TiO2 overlapped among several moral ratios of CO2/H2/H2O under the illumination condition with UV light.
According to Figures 11 and 12, the moral ratio of CO2/H2/H2O = 1:1:0.5 generates the best CO2 reduction performance among various ratios, which is the like the case of the single Fe/TiO2. In addition, the order of CO2 reduction performance of Fe/TiO2 overlapped is the same as that of single Fe/TiO2. However, comparing Figure 11 with Figure 7, the concentration of CO for Fe/TiO2 overlapped is higher than that for single Fe/TiO2 under every molar ratio of CO2/H2/H2O. The highest concentration of CO for Fe/TiO2 overlapped is 993ppmV which is 1.2 times as large as that of single Fe/TiO2. When overlapped, the following things may occur:
However, comparing Figure 12 with Figure 8, the molar quantity of CO per weight of photocatalyst for Fe/TiO2 overlapped is lower than that for single Fe/TiO2 under every molar ratio of CO2/H2/H2O. The highest molar quantity of CO per weight of photocatalyst is 18mmol/g which is half of single Fe/TiO2. The reasons of this result are considered as follows:
From these results, the effective light utilization and mass transfer promotion are necessary for improvement of CO2 reduction performance of Fe/TiO2 overlapped.
Figures 14 &15 show the concentration changes of CO produced and the molar quantity of CO per weight of photocatalyst in the reactor along the time under the Xe lamp without UV light on, for Fe/TiO2 film coated on netlike glass disc overlapped, respectively.
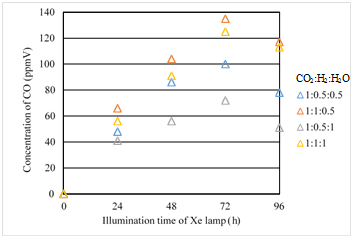
Figure 14 Comparison of concentration of CO for Fe/TiO2 overlapped among several moral ratios of CO2/H2/H2O under the illumination condition without UV light.
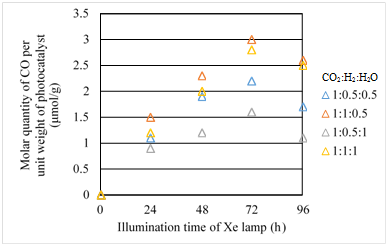
Figure 15 Comparison of molar quantity of CO per unit weight of photocatalyst for Fe/TiO2 overlapped among several moral ratios of CO2/H2/H2O under the illumination condition without UV light.
According to Figures 14 & 15, the moral ratio of CO2/H2/H2O=1:1:0.5 again generates the best CO2 reduction performance. The order of CO2 reduction performance of Fe/TiO2 overlapped is the same as that of single Fe/TiO2. However, comparing Figure 14 with Figure 9, the maximum concentration of CO for Fe/TiO2 overlapped is higher than that for single Fe/TiO2 under every molar ratio of CO2/H2/H2O. The highest concentration of CO for Fe/TiO2 overlapped is 135ppmV which is about 1.2times as large as that for single Fe/TiO2. The same reasons can explain this result as the illumination condition with UV light.
However, comparing Figure 15 with Figure 10, the molar quantity of CO per weight of photocatalyst for Fe/TiO2 overlapped is lower than that for single Fe/TiO2 under every molar ratio of CO2/H2/H2O. The highest molar quantity of CO per weight of photocatalyst for overlapped case is only 3.0mmol/g which is almost half of single Fe/TiO2.
In this study, the highest molar quantity of CO per weight of photocatalyst is 36mmol/g for single Fe/TiO2 under the illumination condition with UV light. Compared to the previous research on CO2 reduction with H2 and H2O over pure TiO2, the CO production performance of photocatalysts prepared in this study is approximately 120 times as large as that reported in references,26,28 which is owing to Fe doping. The CO production performance of Fe/TiO2 prepared in this study is approximately 36 times as large as that of ZrO2 reported in the reference.26 This is due to that the doped Fe provides the free electron preventing recombination of electron and hole produced as well as improving the light absorption effect. Another way to further promote the CO2 reduction performance may be using the netlike porous metal which has an appropriate area of aperture as a base material instead of netlike glass fiber. Since the netlike porous metal has good electrical conductivity, light permeability, and gas diffusivity, it can be expected that the CO2 reduction performance can be further promoted. In addition, the other dopant like Cu, which can absorb the longer wavelength light than Fe,19 should be used at lower positioned layers since the wavelength of penetrating light becomes long through higher positioned photocatalyst.30 This proposal is similar to the concept of the hybrid photocatalyst using two photocatalysts having different band gaps.35−37
Based on the investigation in this study, the following conclusions can be drawn.
The authors would like to gratefully thank from JSPS KAKENHI Grant Number 16K06970, and joint research program of the Institute of Materials and Systems for Sustainability, Nagoya University for the financial support of this work.
The authors declare that there is no conflicts of interest.

©2019 Nishimura, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.